Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 (001) surface with supersonic seeded oxygen molecular beam
(001) surface with supersonic seeded oxygen molecular beamKatsube, Daiki*; Ono, Shinya*; Inami, Eiichi*; Yoshigoe, Akitaka; Abe, Masayuki*
Vacuum and Surface Science, 65(11), p.526 - 530, 2022/11
The oxidation of oxygen vacancies at the surface of anatase TiO (001) was investigated by synchrotron radiation photoelectron spectroscopy and supersonic O
(001) was investigated by synchrotron radiation photoelectron spectroscopy and supersonic O beam (SSMB). The oxygen vacancies at the top surface and subsurface could be eliminated by the supply of hyperthermal oxygen molecules. Oxygen vacancies are present on the surface of anatase TiO
beam (SSMB). The oxygen vacancies at the top surface and subsurface could be eliminated by the supply of hyperthermal oxygen molecules. Oxygen vacancies are present on the surface of anatase TiO (001) when it is untreated before transfer to a vacuum chamber. These vacancies, which are stable in the ambient condition, could also be effectively eliminated by using oxygen SSMB. This result is promising as a surface processing for various functional oxides.
(001) when it is untreated before transfer to a vacuum chamber. These vacancies, which are stable in the ambient condition, could also be effectively eliminated by using oxygen SSMB. This result is promising as a surface processing for various functional oxides.
 (001) surface using supersonic seeded oxygen molecular beam
(001) surface using supersonic seeded oxygen molecular beamKatsube, Daiki*; Ono, Shinya*; Takayanagi, Shuhei*; Ojima, Shoki*; Maeda, Motoyasu*; Origuchi, Naoki*; Ogawa, Arata*; Ikeda, Natsuki*; Aoyagi, Yoshihide*; Kabutoya, Yuito*; et al.
Langmuir, 37(42), p.12313 - 12317, 2021/10
Times Cited Count:1 Percentile:6.39(Chemistry, Multidisciplinary)We investigated the oxidation of oxygen vacancies at the surface of anatase TiO (001) using supersonic seeded molecular beam (SSMB) of oxygen. The oxygen vacancies at the top-surface and sub-surface could be eliminated by the supply of oxygen using an SSMB. These results indicate that the interstitial vacancies can be mostly assigned to oxygen vacancies, which can be effectively eliminated by using an oxygen SSMB. Oxygen vacancies are present on the surface of anatase TiO
(001) using supersonic seeded molecular beam (SSMB) of oxygen. The oxygen vacancies at the top-surface and sub-surface could be eliminated by the supply of oxygen using an SSMB. These results indicate that the interstitial vacancies can be mostly assigned to oxygen vacancies, which can be effectively eliminated by using an oxygen SSMB. Oxygen vacancies are present on the surface of anatase TiO (001) when it is untreated before transfer to a vacuum chamber. These vacancies, which are stable in the as-grown condition, could also be effectively eliminated using the oxygen SSMB.
(001) when it is untreated before transfer to a vacuum chamber. These vacancies, which are stable in the as-grown condition, could also be effectively eliminated using the oxygen SSMB.
Abe, Hiroshi; Tokuhira, Shinnosuke*; Uchida, Hirohisa*; Oshima, Takeshi
Nuclear Instruments and Methods in Physics Research B, 365(Part A), p.214 - 217, 2015/12
no abstracts in English
Abe, Hiroshi; Shimomura, Takuya; Tokuhira, Shinnosuke*; Shimada, Yukihiro*; Takenaka, Yusuke*; Furuyama, Yuta*; Nishimura, Akihiko; Uchida, Hirohisa*; Daido, Hiroyuki; Oshima, Takeshi
Proceedings of 7th International Congress on Laser Advanced Materials Processing (LAMP 2015) (Internet), 4 Pages, 2015/08
A short pulse laser (the nanosecond and femtosecond) was applied to hydrogen absorbing alloys surface layer, and a surface modification experiment was put into effective to aim at improvement of hydrogen adsorption functionally. It was investigated about correlation between an initial hydrogen absorption reaction rate of hydrogen alloys and a laser irradiation in this research. The laser irradiation condition was done with pulse width 100 fsec and energy 0.2 - 3.4 mJ/pulse. It blazed down on hydrogen absorbing alloys (LaNi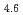 Al
Al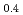 ) and changed local order in the surface. As a result, the initial hydrogen absorption reaction rate was 1.5 - 3.0 times as fast as a irradiated sample, and the result and laser irradiated sample found out that a hydrogen absorption function improves. A laser irradiation can conclude to be effective in surface modification of the hydrogen storage materials.
) and changed local order in the surface. As a result, the initial hydrogen absorption reaction rate was 1.5 - 3.0 times as fast as a irradiated sample, and the result and laser irradiated sample found out that a hydrogen absorption function improves. A laser irradiation can conclude to be effective in surface modification of the hydrogen storage materials.
Tokuhira, Shinnosuke*; Moriyama, Kazuhiro*; Abe, Hiroshi; Uchida, Hirohisa*
no journal, ,
no abstracts in English
Abe, Hiroshi; Moriyama, Kazuhiro*; Tokuhira, Shinnosuke*; Uchida, Hirohisa*; Oshima, Takeshi
no journal, ,
no abstracts in English
Tokuhira, Shinnosuke*; Ohata, Yuki*; Abe, Hiroshi; Hiraki, Takeshi*; Uchida, Hirohisa*
no journal, ,
no abstracts in English
Tokuhira, Shinnosuke*; Onuki, Shun*; Uchida, Hirohisa*; Matsumura, Yoshihito*; Abe, Hiroshi
no journal, ,
no abstracts in English
Tokuhira, Shinnosuke*; Uchida, Hirohisa*; Abe, Hiroshi
no journal, ,
no abstracts in English
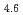 Al
Al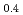
Ono, Taiga*; Tokuhira, Shinnosuke*; Abe, Hiroshi; Hiraki, Takeshi*; Uchida, Hirohisa*
no journal, ,
no abstracts in English
Abe, Hiroshi; Tokuhira, Shinnosuke*; Shimomura, Takuya; Shimada, Yukihiro*; Takenaka, Yusuke*; Furuyama, Yuta*; Nishimura, Akihiko; Daido, Hiroyuki; Uchida, Hirohisa*; Oshima, Takeshi
no journal, ,
no abstracts in English
Daido, Hiroyuki; Abe, Hiroshi; Shobu, Takahisa; Shimomura, Takuya; Tokuhira, Shinnosuke*; Takenaka, Yusuke*; Furuyama, Takehiro*; Nishimura, Akihiko; Uchida, Hirohisa*; Oshima, Takeshi
no journal, ,
no abstracts in English
Abe, Hiroshi; Shimomura, Takuya; Shobu, Takahisa; Tokuhira, Shinnosuke*; Miyashita, Atsumi; Nishimura, Akihiko; Daido, Hiroyuki; Uchida, Hirohisa*
no journal, ,
no abstracts in English
Katsube, Daiki*; Ono, Shinya*; Takayanagi, Shuhei*; Ojima, Shoki*; Maeda, Motoyasu*; Yoshida, Hikaru*; Nishi, Shizuka*; Yoshigoe, Akitaka; Abe, Masayuki*
no journal, ,
TiO has been extensively studied because of high photocatalytic activity. However, the basic reaction process of photocatalysis has not been understood yet. In this study, the difference in water adsorption reaction between rutile and anatase was clarified using synchrotron radiation soft X-ray photoelectron spectroscopy (XPS). The OH component observed at the higher binding energy side was clearly confirmed in the rutile type TiOTiO
has been extensively studied because of high photocatalytic activity. However, the basic reaction process of photocatalysis has not been understood yet. In this study, the difference in water adsorption reaction between rutile and anatase was clarified using synchrotron radiation soft X-ray photoelectron spectroscopy (XPS). The OH component observed at the higher binding energy side was clearly confirmed in the rutile type TiOTiO , suggesting that the rutile type is more reactive to water adsorption.
, suggesting that the rutile type is more reactive to water adsorption.
 (001) surface
(001) surfaceKatsube, Daiki*; Ono, Shinya*; Kim, K.*; Tsuda, Yasutaka; Inami, Eiichi*; Yoshigoe, Akitaka; Abe, Masayuki*
no journal, ,
NOx-based gases emitted from engines and plants are an important issue from an environmental perspective. For the detoxification of NO, it is important to understand its reactivity and reaction mechanisms. The reactivity of anatase TiO (001) which is a highly active photocatalyst was studied by using NO supersonic molecular beams and synchrotron radiation XPS. It was found that the N1s peak was observed, indicating that the surface without surface cleaning reacts with NO molecules.
(001) which is a highly active photocatalyst was studied by using NO supersonic molecular beams and synchrotron radiation XPS. It was found that the N1s peak was observed, indicating that the surface without surface cleaning reacts with NO molecules.
吉越 章隆
阿部 真之*; 勝部 大樹*; 大野 真也*
【課題】 効率的なN元素ドープTIO2の製造方法を提供する。 【解決手段】本発明に係るN元素ドープTIO2の製造方法は、ターゲットとなるTIO2を真空チャンバ内の所定位置に配置するステップと、真空チャンバを真空引きするステップと、ノズルを所定温度に加熱するステップと、ノズルを所定温度に維持しつつ、ノズルからNO分子の超音速分子線をターゲットに照射するステップとを備える。