Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 (101) surface
(101) surface長塚 直樹*; 福谷 克之; 他2名*
Physical Review B, 105(4), p.045424_1 - 045424_6, 2022/01
被引用回数:3 パーセンタイル:43.71(Materials Science, Multidisciplinary)Hydrogen is involved in various important chemical processes on metal-oxide surfaces. Due to its electron-donating character, hydrogen tends to form polaronic states, where the electron is spatially separated from the proton, which exerts significant impact on the electronic properties of metal-oxide surfaces. By individually probing the proton with nuclear reaction analysis and the electron with ultraviolet photoelectron spectroscopy and in combination with density functional theory calculations, we clarify the electronic state of hydrogen on the anatase TiO (101) surface. Hydrogen is found to adsorb with a saturation coverage of 0.48
(101) surface. Hydrogen is found to adsorb with a saturation coverage of 0.48 0.12 monolayers, donating an excess electron to the substrate without forming a midgap state, which is in contrast to the behavior of oxygen vacancies.
0.12 monolayers, donating an excess electron to the substrate without forming a midgap state, which is in contrast to the behavior of oxygen vacancies.
藤本 将秀*; 松本 益明*; 長塚 直樹*; 福谷 克之
RSC Advances (Internet), 11(7), p.4270 - 4275, 2021/01
被引用回数:0 パーセンタイル:0(Chemistry, Multidisciplinary)A fast blackening process of titanium dioxide nanoparticles by exposing to atomic hydrogen was studied by estimating the color of the nanoparticles. The whiteness of TiO decreased exponentially with time, which suggests a first-order reaction between atomic H and surface oxygen, whose rate constant is proportional to the ambient pressure of H
decreased exponentially with time, which suggests a first-order reaction between atomic H and surface oxygen, whose rate constant is proportional to the ambient pressure of H . The rate constant increases as the temperature of nanoparticles at exposing to atomic hydrogen. The structure and size of nanoparticles were estimated by the X-ray diffraction (XRD), which shows that a part of anatase transferred to rutile and the crystal sizes of both anatase and rutile increased by hydrogenation above 600 K. The blackening of TiO
. The rate constant increases as the temperature of nanoparticles at exposing to atomic hydrogen. The structure and size of nanoparticles were estimated by the X-ray diffraction (XRD), which shows that a part of anatase transferred to rutile and the crystal sizes of both anatase and rutile increased by hydrogenation above 600 K. The blackening of TiO halfway stopped under the condition of the similar partial pressure of water with hydrogen. This suggests the presence of reverse reaction between H
halfway stopped under the condition of the similar partial pressure of water with hydrogen. This suggests the presence of reverse reaction between H O and oxygen vacancy, whose reaction rate constant is proportional to the partial pressure of H
O and oxygen vacancy, whose reaction rate constant is proportional to the partial pressure of H O.
O.
 (101) surface
(101) surface長塚 直樹*; Wilde, M.*; 福谷 克之
Journal of Chemical Physics, 152(7), p.074708_1 - 074708_6, 2020/02
被引用回数:10 パーセンタイル:58.2(Chemistry, Physical)Hydrogenation of TiO enhances its visible photoabsorption, leading to efficient photocatalytic activity. However, the role of hydrogen has not been fully understood. The anatase TiO
enhances its visible photoabsorption, leading to efficient photocatalytic activity. However, the role of hydrogen has not been fully understood. The anatase TiO (101) surface treated by hydrogen ion irradiation at 500 eV was investigated by photoemission spectroscopy and nuclear reaction analysis. Hydrogen irradiation induces an in-gap state 1-1.6 eV below the Fermi level and a downward band bending of 0.27 eV. The H depth profile at 300 K shows a surface peak with an H amount of (2.9
(101) surface treated by hydrogen ion irradiation at 500 eV was investigated by photoemission spectroscopy and nuclear reaction analysis. Hydrogen irradiation induces an in-gap state 1-1.6 eV below the Fermi level and a downward band bending of 0.27 eV. The H depth profile at 300 K shows a surface peak with an H amount of (2.9 0.3)
0.3) 10
10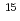 cm
cm with little concentration in a deeper region. At 200 K, on the other hand, the H depth profile shows a maximum at about 1 nm below the surface corresponding to an H amount of (6.1
with little concentration in a deeper region. At 200 K, on the other hand, the H depth profile shows a maximum at about 1 nm below the surface corresponding to an H amount of (6.1 0.3)
0.3) 10
10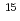 cm
cm along with a broad distribution extending to 50 nm at an average concentration of 0.8 at. %. These results show that H diffusion in anatase TiO
along with a broad distribution extending to 50 nm at an average concentration of 0.8 at. %. These results show that H diffusion in anatase TiO is much faster than in rutile TiO
is much faster than in rutile TiO [Y. Ohashi, J. Phys. Chem. C 123, 10319-10324 (2019)]. The H diffusion coefficient at 200 K is determined to be 2.7
[Y. Ohashi, J. Phys. Chem. C 123, 10319-10324 (2019)]. The H diffusion coefficient at 200 K is determined to be 2.7 0.1
0.1 10
10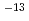 m
m s
s .
.
 (110) hydrogenated with low-energy hydrogen ions
(110) hydrogenated with low-energy hydrogen ions大橋 由季*; 長塚 直樹*; 小倉 正平*; 福谷 克之
Journal of Physical Chemistry C, 123(16), p.10319 - 10324, 2019/04
被引用回数:12 パーセンタイル:47.28(Chemistry, Physical)The rutile TiO surface irradiated by hydrogen ions at 0.5 keV was investigated by ultraviolet photoemission spectroscopy, X-ray photoemission spectroscopy, and nuclear reaction analysis. With hydrogen irradiation, an in-gap state (IGS) was formed at
surface irradiated by hydrogen ions at 0.5 keV was investigated by ultraviolet photoemission spectroscopy, X-ray photoemission spectroscopy, and nuclear reaction analysis. With hydrogen irradiation, an in-gap state (IGS) was formed at  0.8 eV below the Fermi level, and the bands were downward bent by 0.5 eV. The H depth profile showed a maximum at about 1 nm, extending to
0.8 eV below the Fermi level, and the bands were downward bent by 0.5 eV. The H depth profile showed a maximum at about 1 nm, extending to  30 nm with an average concentration of 5.6 at. %. Upon annealing at 673 K, the IGS intensity was reduced and H with a coverage of 1.4 monolayer remained in the near-surface region, suggesting stable H occupation of subsurface sites. After an O
30 nm with an average concentration of 5.6 at. %. Upon annealing at 673 K, the IGS intensity was reduced and H with a coverage of 1.4 monolayer remained in the near-surface region, suggesting stable H occupation of subsurface sites. After an O dose to the H-irradiated surface, although the IGS intensity was substantially reduced, the hydrogen distribution was found to be unchanged.
dose to the H-irradiated surface, although the IGS intensity was substantially reduced, the hydrogen distribution was found to be unchanged.