Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Osawa, Naoki*; Kim, S.-Y.*; Kubota, Masahiko*; Wu, H.*; Watanabe, So; Ito, Tatsuya; Nagaishi, Ryuji
Nuclear Engineering and Technology, 56(3), p.812 - 818, 2024/03
Times Cited Count:0 -hexaoctyl nitrilotriacetamide (HONTA) using mixer-settler extractors in a hot cell
-hexaoctyl nitrilotriacetamide (HONTA) using mixer-settler extractors in a hot cellBan, Yasutoshi; Suzuki, Hideya*; Hotoku, Shinobu; Tsubata, Yasuhiro
Solvent Extraction Research and Development, Japan, 31(1), p.1 - 11, 2024/00
A demonstration test was performed to separate minor actinides (MA; Am and Cm) by  -hexaoctyl nitrilotriacetamide (HONTA) as an extractant using mixer-settler extractors installed in a hot cell. A high-level liquid waste containing MA, and rare earths (RE; Y, La, Nd, and Eu) was used as the feed. HONTA diluted to 0.05 mol/dm
-hexaoctyl nitrilotriacetamide (HONTA) as an extractant using mixer-settler extractors installed in a hot cell. A high-level liquid waste containing MA, and rare earths (RE; Y, La, Nd, and Eu) was used as the feed. HONTA diluted to 0.05 mol/dm in
in  -dodecane was fed as the organic phase, and a part of the organic phase was reused without solvent regeneration. HONTA effectively extracted MA, whereas RE were less extractable. Consequently, the Y, La, Nd, and Eu ratios distributed to a RE fraction were
-dodecane was fed as the organic phase, and a part of the organic phase was reused without solvent regeneration. HONTA effectively extracted MA, whereas RE were less extractable. Consequently, the Y, La, Nd, and Eu ratios distributed to a RE fraction were  99.9%, 99.2%, 61.8%, and 81.4%, respectively. The Am and Cm ratios distributed to an MA fraction were 86.8% and 74.7%, respectively, and a substantial amount of MA (0.12 g) was recovered in the MA fraction by the end of the cumulative duration of 40 h.
99.9%, 99.2%, 61.8%, and 81.4%, respectively. The Am and Cm ratios distributed to an MA fraction were 86.8% and 74.7%, respectively, and a substantial amount of MA (0.12 g) was recovered in the MA fraction by the end of the cumulative duration of 40 h.
Suzuki, Hideya*; Ban, Yasutoshi
Analytical Sciences, 39(8), p.1341 - 1348, 2023/08
Times Cited Count:2 Percentile:71.78(Chemistry, Analytical)The Japan Atomic Energy Agency (JAEA) has proposed the Solvent Extraction from Liquid waste using Extractants of CHON-type for Transmutation (SELECT) process by solvent extraction as a new separation technology to recover minor actinides (MA) from high-level liquid waste (HLLW) produced by spent fuel reprocessing. The MA separation in the SELECT process comprises the batch recovery of MA and rare earths (RE) from HLLW, MA/RE separation, and Am/Cm separation. Three highly practical extractants are used in the MA separation. Furthermore, this flow configuration facilitates the preparation of nitric acid concentrations in the aqueous phase. However, the separation factor between Cm and Nd in the MA/RE separation is small (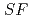
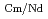 = 2.5), requiring many extraction stages for continuous extraction in a mixer-settler. Therefore, this study investigated the separation of only Am from an aqueous nitric acid solution containing MA (Am and Cm) and RE using an organic phase mixed with two extractants alkyl diamideamine with 2-ethylhexyl alkyl chains (ADAAM(EH)) and hexa-n-octylnitrilotriacetamide (HONTA) used in the SELECT process. Under high-concentration nitric acid conditions, Am and La, Ce, Pr, Nd (light lanthanides) were extracted in the ADAAM(EH) + HONTA mixed solvent, whereas Cm, medium, and heavy lanthanides, and Y were partitioned in the aqueous phase. Subsequently, only light lanthanides could be back extracted from the ADAAM(EH) + HONTA mixture solvent containing Am and light lanthanides in low nitric acid concentrations. Furthermore, Am could be easily stripped with 0.2 M or 5 M nitric acid. This method does not require the mutual separation of Cm and Nd, which have low separation factors. Am can be efficiently separated by one extraction and two back-extractions, reducing the number of steps in the SELECT process.
= 2.5), requiring many extraction stages for continuous extraction in a mixer-settler. Therefore, this study investigated the separation of only Am from an aqueous nitric acid solution containing MA (Am and Cm) and RE using an organic phase mixed with two extractants alkyl diamideamine with 2-ethylhexyl alkyl chains (ADAAM(EH)) and hexa-n-octylnitrilotriacetamide (HONTA) used in the SELECT process. Under high-concentration nitric acid conditions, Am and La, Ce, Pr, Nd (light lanthanides) were extracted in the ADAAM(EH) + HONTA mixed solvent, whereas Cm, medium, and heavy lanthanides, and Y were partitioned in the aqueous phase. Subsequently, only light lanthanides could be back extracted from the ADAAM(EH) + HONTA mixture solvent containing Am and light lanthanides in low nitric acid concentrations. Furthermore, Am could be easily stripped with 0.2 M or 5 M nitric acid. This method does not require the mutual separation of Cm and Nd, which have low separation factors. Am can be efficiently separated by one extraction and two back-extractions, reducing the number of steps in the SELECT process.
 -MoO
-MoO whiskers in
whiskers in  Mo/
Mo/ Tc radioisotope production and
Tc radioisotope production and  Mo/
Mo/ Tc extraction using hot atoms
Tc extraction using hot atomsNgo, M. C.*; Fujita, Yoshitaka; Suzuki, Tatsuya*; Do, T. M. D.*; Seki, Misaki; Nakayama, Tadachika*; Niihara, Koichi*; Suematsu, Hisayuki*
Inorganic Chemistry, 62(32), p.13140 - 13147, 2023/08
Times Cited Count:0 Percentile:0.01(Chemistry, Inorganic & Nuclear)Technetium-99m ( Tc) is one of the most important radioisotopes for diagnostic radio-imaging applications.
Tc) is one of the most important radioisotopes for diagnostic radio-imaging applications.  Tc is a daughter product of the
Tc is a daughter product of the  Mo isotope. There are two methods used to produce
Mo isotope. There are two methods used to produce  Mo/
Mo/ Tc: the nuclear fission (n,f) and the neutron capture (n,
Tc: the nuclear fission (n,f) and the neutron capture (n, ) methods. Between them, the (n,f) method is the main route, used for approximately 90% of the world's production. However, the (n,f) method faces numerous problems, including the use of highly enriched uranium, the release of highly radioactive waste, and nonproliferation problems. Therefore, the (n,
) methods. Between them, the (n,f) method is the main route, used for approximately 90% of the world's production. However, the (n,f) method faces numerous problems, including the use of highly enriched uranium, the release of highly radioactive waste, and nonproliferation problems. Therefore, the (n, ) method is being developed as a future replacement for the (n,f) method. In this work,
) method is being developed as a future replacement for the (n,f) method. In this work,  -MoO
-MoO whiskers prepared by the thermal evaporation method and
whiskers prepared by the thermal evaporation method and  -MoO
-MoO particles were irradiated in a nuclear reactor to produce
particles were irradiated in a nuclear reactor to produce  Mo/
Mo/ Tc via neutron capture. The irradiated targets were dispersed into water to extract the
Tc via neutron capture. The irradiated targets were dispersed into water to extract the  Mo/
Mo/ Tc. As a result,
Tc. As a result,  -MoO
-MoO whisker yielded higher
whisker yielded higher  Mo extraction rate than that from
Mo extraction rate than that from  -MoO
-MoO . In addition, by comparing the dissolved
. In addition, by comparing the dissolved 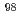 Mo concentrations in water, we clarified a prominent hot-atom of
Mo concentrations in water, we clarified a prominent hot-atom of  -MoO
-MoO whiskers. This research is the first demonstration of
whiskers. This research is the first demonstration of  -MoO
-MoO being used as an irradiation target in the neutron capture method. On the basis of the results,
being used as an irradiation target in the neutron capture method. On the basis of the results,  -MoO
-MoO is considered a promising irradiation target for producing
is considered a promising irradiation target for producing  Mo/
Mo/ Tc by neutron capture and using water for the radioisotope extraction process in the future.
Tc by neutron capture and using water for the radioisotope extraction process in the future.
 Tc separation/concentration technology from
Tc separation/concentration technology from  Mo by (n,
Mo by (n,  ) method
) methodFujita, Yoshitaka; Hu, X.*; Takeuchi, Tomoaki; Takeda, Ryoma; Fujihara, Yasuyuki*; Yoshinaga, Hisao*; Hori, Junichi*; Suzuki, Tatsuya*; Suematsu, Hisayuki*; Ide, Hiroshi
KURNS Progress Report 2022, P. 110, 2023/07
no abstracts in English
Watanabe, So; Takahatake, Yoko; Hasegawa, Kenta; Goto, Ichiro*; Miyazaki, Yasunori; Watanabe, Masayuki; Sano, Yuichi; Takeuchi, Masayuki
Proceedings of 30th International Conference on Nuclear Engineering (ICONE30) (Internet), 6 Pages, 2023/05
Hasegawa, Kenta; Goto, Ichiro*; Miyazaki, Yasunori; Ambai, Hiromu; Watanabe, So; Watanabe, Masayuki; Sano, Yuichi; Takeuchi, Masayuki
Proceedings of 30th International Conference on Nuclear Engineering (ICONE30) (Internet), 5 Pages, 2023/05
Massey, D.*; Williams, C. D.*; Mu, J.*; Masters, A. J.*; Motokawa, Ryuhei; Aoyagi, Noboru; Ueda, Yuki; Antonio, M. R.*
Journal of Physical Chemistry B, 127(9), p.2052 - 2065, 2023/03
Times Cited Count:0 Percentile:0(Chemistry, Physical)Nuclear Science and Engineering Center; Fuel Cycle Design Office; Plutonium Fuel Development Center; Nuclear Plant Innovation Promotion Office; Fast Reactor Cycle System Research and Development Center; J-PARC Center
JAEA-Review 2022-052, 342 Pages, 2023/02
This report summarizes the current status and future plans of research and development (R&D) on partitioning and transmutation technology in Japan Atomic Energy Agency, focusing on the results during the 3rd Medium- to Long-term Plan period (FY 2015-2021). Regarding the partitioning technology, R&D of the solvent extraction method and the extraction chromatography method are described, and regarding the minor actinide containing fuel technology, R&D of the oxide fuel production using the simplified pellet method, the nitride fuel production using the external gelation method, and pyrochemical reprocessing of the nitride fuel were summarized. Regarding transmutation technology, R&D of technology using fast reactors and accelerator drive systems were summarized. Finally, the new facilities necessary for the future R&D were mentioned.
Micheau, C.; Ueda, Yuki; Akutsu, Kazuhiro*; Bourgeois, D.*; Motokawa, Ryuhei
Solvent Extraction and Ion Exchange, 41(2), p.221 - 240, 2023/02
Times Cited Count:1 Percentile:44.81(Chemistry, Multidisciplinary)Sasaki, Yuji; Nakase, Masahiko*; Kaneko, Masashi; Kobayashi, Toru; Takeshita, Kenji*; Matsumiya, Masahiko*
Analytical Sciences, 5 Pages, 2023/00
Times Cited Count:0 Percentile:0(Chemistry, Analytical)We conducted three field researches on Ru-extraction, XANES, and DFT-calculation. The order of the distribution ratio, D(Ru), from acid, HCl  H
H SO
SO
 HNO
HNO
 HClO
HClO , by MIDOA is studied by XANES spectra, which indicates the valency change of Ru in HCl media and supports the ion pairing extraction of anionic Ru ion and cationic MIDOA. The same extractant trend, NTAamide
, by MIDOA is studied by XANES spectra, which indicates the valency change of Ru in HCl media and supports the ion pairing extraction of anionic Ru ion and cationic MIDOA. The same extractant trend, NTAamide  MIDOA
MIDOA  IDOA, due to D values as the energy gap of HOMO and LUMO could be found by DFT calculation, which suggests that the reaction heat has a positive correlation with extractability for extractant.
IDOA, due to D values as the energy gap of HOMO and LUMO could be found by DFT calculation, which suggests that the reaction heat has a positive correlation with extractability for extractant.
Uchino, Seiko*; Narita, Hirokazu*; Kita, Keisuke*; Suzuki, Hideya*; Matsumura, Tatsuro; Naganawa, Hirochika*; Sakaguchi, Koichi*; Oto, Keisuke*
Solvent Extraction Research and Development, Japan, 30(1), p.39 - 46, 2023/00
The extraction of trivalent rare earth ions (RE ) from HNO
) from HNO solution using a triamide amine, tris(N,N-di-2-ethylhexyl-ethylamide)amine (DEHTAA), was conducted, and the extraction mechanism was estimated from extraction behavior of HNO
solution using a triamide amine, tris(N,N-di-2-ethylhexyl-ethylamide)amine (DEHTAA), was conducted, and the extraction mechanism was estimated from extraction behavior of HNO and RE
and RE and the relationship between atomic number and extraction percentages (E%) for RE
and the relationship between atomic number and extraction percentages (E%) for RE . A DEHTAA molecule dominantly formed a DEHTAA HNO
. A DEHTAA molecule dominantly formed a DEHTAA HNO at 1.0 M HNO
at 1.0 M HNO and a DEHTAA(HNO
and a DEHTAA(HNO )
) at 6.0 M HNO
at 6.0 M HNO in the acid-equilibrated organic phase. This would provide the unique dependence of E% for the light RE
in the acid-equilibrated organic phase. This would provide the unique dependence of E% for the light RE on the HNO
on the HNO concentration, in which the E% value had a minimum and maximum at
concentration, in which the E% value had a minimum and maximum at  0.5 M and
0.5 M and  2 M HNO
2 M HNO , respectively. The results of the slope analyses for the distribution ratios for RE
, respectively. The results of the slope analyses for the distribution ratios for RE suggested that the dominant RE
suggested that the dominant RE complex was RE(NO
complex was RE(NO )
) DEHTAA(DEHTAA HNO
DEHTAA(DEHTAA HNO ) at 1.0 M HNO
) at 1.0 M HNO . The E% for RE
. The E% for RE decreased from La
decreased from La to Lu
to Lu at 1.0 M HNO
at 1.0 M HNO ; on the other hand, those increased from La
; on the other hand, those increased from La to Nd
to Nd at 0.25 M and from La
at 0.25 M and from La to Sm
to Sm and 6.0 M HNO
and 6.0 M HNO .
.
Akuzawa, Tadashi*; Kim, S.-Y.*; Kubota, Masahiko*; Wu, H.*; Watanabe, So; Sano, Yuichi; Takeuchi, Masayuki; Arai, Tsuyoshi*
Journal of Radioanalytical and Nuclear Chemistry, 331(12), p.5851 - 5858, 2022/12
Times Cited Count:5 Percentile:66.21(Chemistry, Analytical)Nguyen, T. H.*; Le Ba, T.*; Tran, C. T.*; Nguyen, T. T.*; Doan, T. T. T.*; Do, V. K.; Watanabe, Masayuki; Pham, Q. M.*; Hoang, S. T.*; Nguyen, D. V.*; et al.
Hydrometallurgy, 213, p.105933_1 - 105933_11, 2022/08
Times Cited Count:9 Percentile:81.6(Metallurgy & Metallurgical Engineering)A continuous counter-current extraction for the selective recovery of thorium (Th) and uranium (U) from the Yen Phu (Vietnam) rare earth concentrate leach solutions was systematically studied. The primary amine N1923 was used as an extractant which was prepared in the isoparaffin IP-2028 diluent. Thorium and uranium were selectively recovered in a hydrometallurgical circuit established by continuous mixer-settler extraction, scrubbing, and back-extraction at the laboratory scale. The desired purity of Th and U can be achieved by managing the volume ratio of organic to aqueous phase (O/A ratio) in the corresponding steps. Highly pure Th and U were recovered from the pregnant back-extraction liquor and the raffinate, respectively, which have satisfactory properties for further processing of the subsequent nuclear materials.
Kusaka, Ryoji; Watanabe, Masayuki
Journal of Physical Chemistry Letters (Internet), 13(30), p.7065 - 7071, 2022/08
Times Cited Count:5 Percentile:67.52(Chemistry, Physical) aqueous solution system revealed by fluorescence microspectroscopy
aqueous solution system revealed by fluorescence microspectroscopyMiyagawa, Akihisa*; Kusano, Yuka*; Nagatomo, Shigenori*; Sano, Yuichi; Nakatani, Kiyoharu*
Analytical Sciences, 38(7), p.955 - 961, 2022/07
Times Cited Count:0 Percentile:0(Chemistry, Analytical)In this study, we reveal an Eu(III) extraction mechanism at the interface between HNO and tributyl phosphate (TBP) solutions using fluorescence microspectroscopy. The mass transfer rate constant at the interface is obtained from the analysis of fluorescence intensity changes during the forward and backward extractions at various HNO
and tributyl phosphate (TBP) solutions using fluorescence microspectroscopy. The mass transfer rate constant at the interface is obtained from the analysis of fluorescence intensity changes during the forward and backward extractions at various HNO and TBP concentrations to investigate the reaction mechanism. This result indicates that one nitrate ion reacts with Eu(III) at the interface, whereas TBP molecules are not involved in the interfacial reaction, which is different from the results obtained using the NaNO
and TBP concentrations to investigate the reaction mechanism. This result indicates that one nitrate ion reacts with Eu(III) at the interface, whereas TBP molecules are not involved in the interfacial reaction, which is different from the results obtained using the NaNO solution in our previous study. We demonstrate that the chemical species of Eu(III) complex with nitrate ion and TBP in the aqueous solution play an important role for the extraction mechanism.
solution in our previous study. We demonstrate that the chemical species of Eu(III) complex with nitrate ion and TBP in the aqueous solution play an important role for the extraction mechanism.
Sano, Yuichi; Sakamoto, Atsushi; Miyazaki, Yasunori; Watanabe, So; Morita, Keisuke; Emori, Tatsuya; Ban, Yasutoshi; Arai, Tsuyoshi*; Nakatani, Kiyoharu*; Matsuura, Haruaki*; et al.
Proceedings of International Conference on Nuclear Fuel Cycle; Sustainable Energy Beyond the Pandemic (GLOBAL 2022) (Internet), 4 Pages, 2022/07
We developed a hybrid MA(III) recovery process combining MA(III)+Ln(III) co-recovery flowsheet by solvent extraction with TBP and MA(III)/Ln(III) separation flowsheet by simulated moving bed chromatography using HONTA impregnated adsorbents with large particle size porous silica support.
Ueda, Yuki; Eguchi, Ayano; Tokunaga, Kohei; Kikuchi, Kei*; Sugita, Tsuyoshi; Okamura, Hiroyuki; Naganawa, Hirochika
Industrial & Engineering Chemistry Research, 61(19), p.6640 - 6649, 2022/05
Times Cited Count:1 Percentile:11.64(Engineering, Chemical)no abstracts in English
Toigawa, Tomohiro; Kumagai, Yuta; Yamashita, Shinichi*; Ban, Yasutoshi; Matsumura, Tatsuro
UTNL-R-0502 (Internet), 2 Pages, 2022/04
This report summarizes the results obtained in FY2020 at the Electron Linac Facility of the University of Tokyo. The radiolysis process of  -hexaoctyl nitrilotriacetamide (HONTA), which is expected to be used as an extractant in a separation process for minor actinides, diluted in dodecane was investigated by pulse radiolysis experiments. The radical cation and the triplet-excited state of HONTA were observed in the nanosecond time region. The transition from the radical cation to the triplet excited state was slowed down by adding electron scavengers, and further, the reactivity of the triplet excited state was also suppressed.
-hexaoctyl nitrilotriacetamide (HONTA), which is expected to be used as an extractant in a separation process for minor actinides, diluted in dodecane was investigated by pulse radiolysis experiments. The radical cation and the triplet-excited state of HONTA were observed in the nanosecond time region. The transition from the radical cation to the triplet excited state was slowed down by adding electron scavengers, and further, the reactivity of the triplet excited state was also suppressed.
Nomizu, Daiki; Sasaki, Yuji; Kaneko, Masashi; Matsumiya, Masahiko*; Katsuta, Shoichi*
Journal of Radioanalytical and Nuclear Chemistry, 331(3), p.1483 - 1493, 2022/03
Times Cited Count:4 Percentile:76.47(Chemistry, Analytical)We studied the successive formation of water soluble DGA (diglycolamide) and DOODA (dioxaoctanediamide) for the mutual separation of Ln in this extraction system. TODGA (tetraoctyl-diglycolamide) and DOODA(C8) (tetraoctyl-dioxaoctanediamide) have the opposite trend to extract light and heavy Ln through Ln-patterns. Metal-complexes of two folding Ln ions with water-soluble DOODA and three folding with DGA are found and their observed formation constants are calculated. The suitable separation condition (aqueous phase: 30 mM DOODA(C2) in 1 M nitric acid, organic phase: 0.1 M TODGA in n-dodecane) of multi-stage extraction (10  10) is conducted. From the present work, it is clear that La, Pr and Nd are mainly present in aqueous phase, instead Sm-Dy exist in the organic phase.
10) is conducted. From the present work, it is clear that La, Pr and Nd are mainly present in aqueous phase, instead Sm-Dy exist in the organic phase.