Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 bus FP results and single effect testing
bus FP results and single effect testingKnebel, K.*; Jokiniemi, J.*; Bottomley, D.
Journal of Nuclear Science and Technology, 56(9-10), p.772 - 789, 2019/09
Times Cited Count:7 Percentile:57.84(Nuclear Science & Technology)Revaporisation of the fission products deposited in the primary circuit of a reactor was identified as a possible late source of fission product release during a severe accident: eg. loss of coolant accident (LOCA). Subsequent testing has shown that revaporisation is very likely to occur given a breach of the reactor and is an important contributor for the source term release to the containment and biosphere. The first part reviews the revaporisation mechanisms of Cs and other volatile or semi-volatile fission products transported in the primary circuit that were derived from the Phebus FP and associated programmes. The second part examines the separate effects testing to determine the high temperature chemistry ofvolatile and semi-volatile fission products (I, Mo, Ru) and structural materials (Ag, B) as well as atmospheric effects which substantially affect the source term. Finally, it examines Cs data from reactor accident sites that is providing additional knowledge of longer-term fission product chemistry. The results have been summarised in the form of a table and schematic diagram. This accumulated knowledge and experience has important applications to minimising contamination during decommissioning and site remediation techniques, as well as improving SA simulation codes and raising nuclear safety.
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Ito, Yuta; Shirai, Kaori*; Suzuki, Hayato; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; et al.
Journal of Radioanalytical and Nuclear Chemistry, 320(3), p.633 - 642, 2019/06
Times Cited Count:2 Percentile:20.94(Chemistry, Analytical)An isothermal gas-chromatographic (IGC) device has been developed and tested for on-line gas phase studies of volatile oxychlorides of short-lived group-5 transition metals. Radioisotopes of niobium and tantalum, produced in nuclear fusion evaporation reactions, are directly flushed into the IGC setup by an inert gas-jet. Oxychloride compounds are formed by the addition of SOCl and O
and O . Parameters influencing the formation and transport of NbOCl
. Parameters influencing the formation and transport of NbOCl and TaOCl
and TaOCl are investigated. For nuclides with half-lives (
are investigated. For nuclides with half-lives (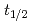 ) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of
) of about 30 s, an overall efficiency of 7% is obtained, rendering the IGC setup suitable for the chemical exploration of  Db(
Db(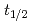 = 34s).
= 34s).
Chiera, N. M.; Sato, Tetsuya; Tomitsuka, Tomohiro; Asai, Masato; Suzuki, Hayato*; Tokoi, Katsuyuki; Toyoshima, Atsushi; Tsukada, Kazuaki; Nagame, Yuichiro
Inorganica Chimica Acta, 486, p.361 - 366, 2019/02
Times Cited Count:4 Percentile:24.55(Chemistry, Inorganic & Nuclear)The formation of NbOCl and TaOCl
and TaOCl and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values (
and their adsorption behavior on quartz surfaces was explored by applying an isothermal gas-chromatographic method. Trace amounts of short-lived Nb and Ta isotopes were used. Adsorption enthalpy values ( ) at zero surface coverage of -
) at zero surface coverage of - (NbOCl
(NbOCl ) = 102
) = 102  4 kJ/mol and -
4 kJ/mol and - (TaOCl
(TaOCl ) = 128
) = 128  5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental
5 kJ/mol were determined by analyzing the chromatographic behavior of the Nb andTa complexes with a Monte-Carlo simulation method based on an adsorption-desorption kinetic model.By applying an empirical correlation, the experimental  values were successively related to the macroscopic standard sublimation enthalpy,
values were successively related to the macroscopic standard sublimation enthalpy, 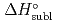 , as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between
, as a measure of the volatility of each substance. The inferred sublimation enthalpies are in agreement with tabulated thermochemical values. Thus, the linear empirical correlation between  and
and 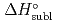 for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted
for metal-oxychlorides was updated with the inclusion of the present data. According to the predicted 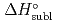 (DbOCl
(DbOCl ), a
), a  (DbOCl
(DbOCl ) value of 135
) value of 135  2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl
2 kJ/mol was extrapolated. The future accomplishment of comparative studies with DbOCl under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
under the same experimental conditions will provide valuable information on the volatility trend in Group-5 elements, together with an indication on the magnitude of relativistic effects on the electronic structure of dubnium.
Kojima, Takuji
Hoshasen, 29(2), p.77 - 85, 2003/04
The radiation technologies for environment conservation are useful for purification of pollutants contained in flue gas or wastewater at very low concentration which is difficult to perform by conventional methods: removal using fine filter or charcoals and decomposition using catalysis at high temperature, etc. This paper reviews some examples of radiation application to removal of SO and NO
and NO from coal-combustion flue gases, decomposition of dioxin in gas emitted through the incinerator, decomposition of gaseous toxic volatile organic compounds in off gas, reuse of agricultural wastes.
from coal-combustion flue gases, decomposition of dioxin in gas emitted through the incinerator, decomposition of gaseous toxic volatile organic compounds in off gas, reuse of agricultural wastes.
Hakoda, Teruyuki; Hashimoto, Shoji; Kojima, Takuji
Bulletin of the Chemical Society of Japan, 75(10), p.2177 - 2183, 2002/10
Times Cited Count:8 Percentile:37.12(Chemistry, Multidisciplinary)no abstracts in English
Kojima, Takuji
JETI, 50(13), p.17 - 21, 2002/10
no abstracts in English
Sunaga, Hiromi
Denki Gakkai Gijutsu Hokoku, (895), p.61 - 62, 2002/09
no abstracts in English
Kojima, Takuji
Enerugi Rebyu, 22(4), p.27 - 29, 2002/04
no abstracts in English
Hirota, Koichi
Hoshasen To Sangyo, (92), p.76 - 79, 2001/12
The research on electron-beam treatment of VOCs (volatile organic compounds) was presented at 10th international conference of Air Pollution 2001 in Italy. The presentation referred to the decomposition products and active species that oxidize the aromatics and aliphatics. Also, the test facility for EB treatment of PCDD/F from MSWI was presented.
Hakoda, Teruyuki; Arai, Hidehiko; Hashimoto, Shoji
Journal of Chemical Engineering of Japan, 34(10), p.1300 - 1308, 2001/10
Times Cited Count:2 Percentile:25.21(Engineering, Chemical)The continuous gas monitoring system using the mass filter (CGM-MS) was developed for the measurement of gaseous substances in air under atmospheric pressure. A capillary tube introduces the gases under atmospheric pressure into a mass filter installed in a vacuum chamber. The CGM-MS detected gaseous substances, such as sulfur dioxide, benzene and chlorobenzene, with detectable sensitivity of 0.7-1 ppmv. The monitoring system was also applied for the analysis of the products formed in electron beam irradiation of trichloroethylene (TCE) and air mixture. Dichloroacetyl chloride, carbonyl chloride (COCl ), and chlorine (Cl
), and chlorine (Cl ) were quantitatively analyzed. Trichloroethylene and the products were oxidized and completely converted into carbon dioxide, Cl
) were quantitatively analyzed. Trichloroethylene and the products were oxidized and completely converted into carbon dioxide, Cl , and hydrochloric acid at 15 kGy. Carbonyl chloride is dissolved in an alkaline solution to be automatically oxidized into CO
, and hydrochloric acid at 15 kGy. Carbonyl chloride is dissolved in an alkaline solution to be automatically oxidized into CO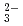 and Cl
and Cl . The combination of the irradiation and the dissolution of the irradiated gas decreased to 7 from 15 kGy for the complete oxidation of TCE and the products.
. The combination of the irradiation and the dissolution of the irradiated gas decreased to 7 from 15 kGy for the complete oxidation of TCE and the products.
Sun, Y.*; Hakoda, Teruyuki; Chmielewski, A. G.*; Hashimoto, Shoji; Zimek, Z.*; Bulka, S.*; Ostapczuk, A.*; Nichipor, H.*
Radiation Physics and Chemistry, 62(4), p.353 - 360, 2001/10
Times Cited Count:22 Percentile:81.29(Chemistry, Physical)no abstracts in English
Hakoda, Teruyuki; Hirota, Koichi; Hashimoto, Shoji
Proceedings of 5th International Symposium & Exhibition on Environmental Contamination in Central & Eastern Europe (CD-ROM), 7 Pages, 2001/09
no abstracts in English
 P) radicals, thermal electrons, and O
P) radicals, thermal electrons, and O in electron beam irradiation of 1,2-dichloroethylenes and air mixtures
in electron beam irradiation of 1,2-dichloroethylenes and air mixturesHakoda, Teruyuki; Zhang, G.*; Hashimoto, Shoji
Radiation Physics and Chemistry, 62(2-3), p.243 - 252, 2001/09
Times Cited Count:13 Percentile:66.85(Chemistry, Physical)no abstracts in English
Hidaka, Akihide; Nakamura, Takehiko; Kudo, Tamotsu; Hayashida, Retsu*; Nakamura, Jinichi; Otomo, Takashi; Uetsuka, Hiroshi
JAERI-Conf 2000-015, p.193 - 200, 2000/11
no abstracts in English
Hashimoto, Shoji; Arai, Hidehiko
Denki Gakkai Gijutsu Hokoku, (810), p.46 - 50, 2000/10
no abstracts in English
Hidaka, Akihide; Nakamura, Takehiko; Kudo, Tamotsu
Genshiryoku eye, 46(3), p.79 - 83, 2000/03
no abstracts in English
Hakoda, Teruyuki; Zhang, G.; Hashimoto, Shoji
JAERI-Conf 2000-001, p.211 - 214, 2000/03
no abstracts in English
Hashimoto, Shoji; Hakoda, Teruyuki; Hirota, Koichi; Arai, Hidehiko
Radiation Physics and Chemistry, 57(3-6), p.485 - 488, 2000/03
Times Cited Count:26 Percentile:82.52(Chemistry, Physical)no abstracts in English
Hakoda, Teruyuki; Hashimoto, Shoji; Fujiyama, Yuichi*; Mizuno, Akira*
Journal of Physical Chemistry A, 104(1), p.59 - 66, 2000/01
Times Cited Count:32 Percentile:68.49(Chemistry, Physical)no abstracts in English
Sun, Y.*; Hakoda, Teruyuki; Chmielewski, A. G.*; Hashimoto, Shoji; Zimek, Z.*; Bulka, S.*; Ostapczuk, A.*; Nichipor, H.*
Proceedings of 5th International Symposium & Exhibition on Environmental Contamination in Central & Eastern Europe (CD-ROM), 6 Pages, 2000/00
no abstracts in English