Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tonna, Ryutaro*; Sasaki, Takayuki*; Okamoto, Yoshihiro; Kobayashi, Taishi*; Akiyama, Daisuke*; Kirishima, Akira*; Sato, Nobuaki*
Journal of Nuclear Materials, 589, p.154862_1 - 154862_10, 2024/02
Times Cited Count:0 Percentile:0.01(Materials Science, Multidisciplinary)The dissolution behavior of FeUO compounds formed by a high-temperature reaction of UO
compounds formed by a high-temperature reaction of UO with iron, a stainless-steel component of reactor structural materials, was investigated under atmospheric conditions. The compounds were prepared in an electric furnace using U
with iron, a stainless-steel component of reactor structural materials, was investigated under atmospheric conditions. The compounds were prepared in an electric furnace using U O
O and Fe
and Fe O
O as starting materials, and their solid states were analyzed using X-ray diffraction, scanning electron microscopy energy dispersive X-ray spectroscopy, and X-ray absorption fine structure spectroscopy. The concentration of nuclides dissolved in water was examined by performing static leaching tests of FeUO
as starting materials, and their solid states were analyzed using X-ray diffraction, scanning electron microscopy energy dispersive X-ray spectroscopy, and X-ray absorption fine structure spectroscopy. The concentration of nuclides dissolved in water was examined by performing static leaching tests of FeUO compounds for up to three months. A redox reaction was proposed to occur between trivalent Fe and pentavalent U ions in the early stage of FeUO
compounds for up to three months. A redox reaction was proposed to occur between trivalent Fe and pentavalent U ions in the early stage of FeUO dissolution. It was thermodynamically deduced that the reduced divalent Fe ion was finally oxidized into a trivalent ion in the presence of dissolved oxygen, and iron hydroxide limited the solubility of Fe. Meanwhile, the concentration of hexavalent U (i.e., uranyl ion) was limited owing to the presence of secondary minerals such as metaschoepite and sodium uranate and subsequently decreased, possibly owing to sorption on Fe oxides, for example. The concentrations of multivalent ions of fission products, such as Ru and Ce, also decreased, likely for the reason above. By contrast, the concentration of soluble Cs ions did not decrease. The validity of this interpretation was supported by comparing the results with the dissolution behavior of a reference sample (Fe-free U
dissolution. It was thermodynamically deduced that the reduced divalent Fe ion was finally oxidized into a trivalent ion in the presence of dissolved oxygen, and iron hydroxide limited the solubility of Fe. Meanwhile, the concentration of hexavalent U (i.e., uranyl ion) was limited owing to the presence of secondary minerals such as metaschoepite and sodium uranate and subsequently decreased, possibly owing to sorption on Fe oxides, for example. The concentrations of multivalent ions of fission products, such as Ru and Ce, also decreased, likely for the reason above. By contrast, the concentration of soluble Cs ions did not decrease. The validity of this interpretation was supported by comparing the results with the dissolution behavior of a reference sample (Fe-free U O
O ).
).
Okamoto, Yoshihiro; Shiwaku, Hideaki; Shimamura, Keisuke*; Kobayashi, Hidekazu; Nagai, Takayuki; Inose, Takehiko*; Sato, Seiichi*; Hatakeyama, Kiyoshi*
Journal of Nuclear Materials, 570, p.153962_1 - 153962_13, 2022/11
Simulated nuclear waste glass samples containing phosphorus, which increase the solubility of molybdenum, were prepared and analyzed using synchrotron X-ray Absorption Fine Structure (XAFS) analysis for some constituent elements and Raman spectroscopic analysis of their complex structure. Changes in local structure and chemical state due to different phosphorus additions and waste loading rates were systematically studied. Consequently, no crystalline phase due to the molybdate compound was observed even at a maximum waste content of 30 wt% (corresponding to 1.87 mol% MoO ). Oxidation proceeded when the waste-loading rate was increased, whereas the reduction proceeded when phosphorus was added. In some cases, the effects of oxidation and reduction were offset. The local structure around specific elements can be classified as follows; Zn that is affected mainly by the waste-loading rate, Ce that is affected by both the waste-loading rate and phosphorus addition, and Zr element that is not affected by either of them. From the comparison between the analytical results of Mo and other elements, it was considered that the added phosphorus exists as a free PO
). Oxidation proceeded when the waste-loading rate was increased, whereas the reduction proceeded when phosphorus was added. In some cases, the effects of oxidation and reduction were offset. The local structure around specific elements can be classified as follows; Zn that is affected mainly by the waste-loading rate, Ce that is affected by both the waste-loading rate and phosphorus addition, and Zr element that is not affected by either of them. From the comparison between the analytical results of Mo and other elements, it was considered that the added phosphorus exists as a free PO structural unit and may deprive the alkali metal coordinated to the molybdate ion.
structural unit and may deprive the alkali metal coordinated to the molybdate ion.
Ogawa, Shuichi*; Taga, Ryo*; Yoshigoe, Akitaka; Takakuwa, Yuji*
Journal of Vacuum Science and Technology A, 39(4), p.043207_1 - 043207_9, 2021/07
Times Cited Count:1 Percentile:7.45(Materials Science, Coatings & Films)Nickel (Ni) is used as a catalyst for nitric oxide decomposition and ammonia production but it is easily oxidized and deactivated. Clarification of the reduction process of oxidized Ni is essential to promote more efficient use of Ni catalysts. In this study, the reduction processes were investigated by in situ time-resolved photoelectron spectroscopy. We propose a two-step reduction reaction model. The rate-limiting process for the first step is surface precipitation of O atoms and that of the second step is dissociation of H molecules.
molecules.
Nakashima, Nobuaki*; Yatsuhashi, Tomoyuki*; Sakota, Kenji*; Iwakura, Izumi*; Hashimoto, Sena*; Yokoyama, Keiichi; Matsuda, Shohei
Chemical Physics Letters, 752, p.137570_1 - 137570_5, 2020/08
Times Cited Count:1 Percentile:4.85(Chemistry, Physical)Photo-redox reactions between Eu and Eu
and Eu ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu
ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu to form Eu
to form Eu . When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
. When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
Kato, Chiaki
Comprehensive Nuclear Materials, 2nd Edition, Vol.4, p.528 - 563, 2020/08
In spent fuel reprocessing plants, various nitric media are encountered throughout the PUREX process, used in the separation of fission products, uranium, and plutonium. The PUREX process is thus highly corrosive as it takes place at high temperatures under high concentrations of nitric acid solution containing oxidizing metal ions from spent fuel. In this review, the unique chemical properties of nitric acid are first described. Secondly, the process of oxidizing power generation in boiling nitric acid under heat transfer is described using the redox potential and a thermodynamic model of boiling nitric acid. Finally, the corrosion behavior and corrosion acceleration mechanism specific to the reprocessing environments are described from the perspective of solution chemistry.
Itoi, Hiroyuki*; Ninomiya, Takeru*; Hasegawa, Hideyuki*; Maki, Shintaro*; Sakakibara, Akihiro*; Suzuki, Ryutaro*; Kasai, Yuto*; Iwata, Hiroyuki*; Matsumura, Daiju; Owada, Mao*; et al.
Journal of Physical Chemistry C, 124(28), p.15205 - 15215, 2020/07
Times Cited Count:8 Percentile:39.3(Chemistry, Physical)Hayashida, Kazuki; Kato, Toshihiro*; Kubota, Mitsuru*; Murakami, Hiroaki; Amano, Yuki; Iwatsuki, Teruki
Chikyu Kagaku, 52(1), p.55 - 71, 2018/03
In this study, the simulated experimental drift was constructed in the granite of 500 m depth at Mizunami Underground Research Laboratory, and the hydrochemical process after the drift closure was observed. The groundwater chemistry around the drift changed with the change of the groundwater flow in the fractures when the gallery was constructed. The redox potential increased due to the infiltration of oxygen from the drift into the rock. After closing the drift, the redox potential of the groundwater plunged due to microbial activity, while the groundwater became alkalized conditon due to the influence of cement material such as shotcrete. The amount of cement material consumed for this alkalization was small, and it was considered that its influence would last long in accordance with the amount of cement used.
Okamoto, Yoshihiro; Shiwaku, Hideaki; Nakada, Masami; Komamine, Satoshi*; Ochi, Eiji*; Akabori, Mitsuo
Journal of Nuclear Materials, 471, p.110 - 115, 2016/02
Times Cited Count:6 Percentile:49.05(Materials Science, Multidisciplinary)Extended X-ray Absorption Fine Structure (EXAFS) analyses were performed to evaluate REDOX (REDuction and OXidation) state of platinoid elements in simulated high-level nuclear waste glass samples prepared under different conditions of temperature and atmosphere. At first, EXAFS functions were compared with those of standard materials such as RuO . Then structural parameters were obtained from a curve fitting analysis. In addition, a fitting analysis used a linear combination of the two standard EXAFS functions of a given elements metal and oxide was applied to determine ratio of metal/oxide in the simulated glass. The redox state of Ru was successfully evaluated from the linear combination fitting results of EXAFS functions. The ratio of metal increased at more reducing atmosphere and at higher temperatures. Chemical form of rhodium oxide in the simulated glass samples was RhO
. Then structural parameters were obtained from a curve fitting analysis. In addition, a fitting analysis used a linear combination of the two standard EXAFS functions of a given elements metal and oxide was applied to determine ratio of metal/oxide in the simulated glass. The redox state of Ru was successfully evaluated from the linear combination fitting results of EXAFS functions. The ratio of metal increased at more reducing atmosphere and at higher temperatures. Chemical form of rhodium oxide in the simulated glass samples was RhO unlike expected Rh
unlike expected Rh O
O . It can be estimated rhodium behaves according with ruthenium when the chemical form is oxide.
. It can be estimated rhodium behaves according with ruthenium when the chemical form is oxide.
Shirai, Osamu*; Yamana, Hajimu*; Arai, Yasuo
Journal of Alloys and Compounds, 408-412, p.1267 - 1273, 2006/02
Times Cited Count:41 Percentile:84.4(Chemistry, Physical)no abstracts in English
Onuki, Toshihiko; Aoyagi, Hisao*; Kitatsuji, Yoshihiro; Samadfam, M.; Kimura, Yasuhiko; Purvis, O. W.*
Journal of Environmental Radioactivity, 77(3), p.339 - 353, 2004/08
Times Cited Count:16 Percentile:33.95(Environmental Sciences)no abstracts in English
Kato, Chiaki; Yano, Masaya*; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Corrosion Engineering, 52(1), p.53 - 67, 2003/01
The effects of heat-transfer on the corrosion of zirconium was examined in boiling nitric acid solutions with various concentrations. Corrosion mass losses and electrochemical polarization curves were measured on the heat-transfer and isothermal surfaces in the solutions. It was found that the corrosion rate of zirconium was higher on the heat-transfer surface than that on the isothermal surface. The rate increased with increasing nitric acid concentration and solution temperature. The increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles. The redox potential of 12 mol/dm nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.
nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.
Kato, Chiaki; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Corrosion Engineering, 52(1), p.69 - 85, 2003/01
It is necessary to know the generation mechanism of high equilibrium potential in the solutions. Existing nitrogen oxides in nitric acid solutions were first analyzed by Raman spectroscopy and then existing amount of nitrogen oxides were examined by thermodynamic calculation using the SOLGASMIX software. The Raman spectroscopic analysis showed that the existing amount of un-dissociated HNO increased with increasing nitric acid concentration and solution temperature. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO
increased with increasing nitric acid concentration and solution temperature. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO , NO
, NO
 , HNO
, HNO , NO
, NO , and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO
, and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO /HNO
/HNO equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
Kato, Chiaki; Yano, Masaya*; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Zairyo To Kankyo, 52(1), p.35 - 43, 2003/01
The effects of heat-transfer on the corrosion of zirconium was examined in boiling nitric acid solutions with various concentrations. Corrosion mass losses and electrochemical polarization curves were measured on the heat-transfer and isothermal surfaces in the solutions. It was found that the corrosion rate of zirconium was higher on the heat-transfer surface than that on the isothermal surface. The rate increased with increasing nitric acid concentration and solution temperature. The increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition on the surface and the removal of the decomposition product from solution by boiling bubbles. The redox potential of 12 mol/dm3 nitric acid on a boiling heat-transfer surface was very close to the breakdown potential of primary passivity of zirconium. This suggests the initiation of SCC on a boiling heat-transfer surface in a nuclear fuel reprocessing.
Kato, Chiaki; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Zairyo To Kankyo, 52(1), p.44 - 52, 2003/01
In order to understand corrosion of metals in nitric acid solutions, it is necessary to know the generation mechanism of high equilibrium potential in the solutions, especially under boiling conditions. The Raman spectroscopic analysis showed that the existing amount of un-dissociated HNO increased with increasing nitric acid concentration and solution temperature. The existing amount of NO
increased with increasing nitric acid concentration and solution temperature. The existing amount of NO also increased by thermal decomposition. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO
also increased by thermal decomposition. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO , NO
, NO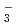 , HNO
, HNO , NO
, NO , and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO
, and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO /HNO
/HNO equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
Mine, Tatsuya*; Mihara, Morihiro;
JNC TN8430 2000-009, 35 Pages, 2000/07
In the geological disposal system of TRU wastes, nitrogen generation by denitrifying bacteria could provide significant impact on the assessment of this system, because nitrate contained in process concentrated liquid waste might be electron acceptor for denitrifying bacteria. In this study, the activities and tolerance of denitrifying bacteria under disposal condition were investigated. pseudomonas denitrificans as denitrifying bacteria was used. The results showed that Pseudomonas denitrificans had activity under reducing condition, but under high pH condition (PH 9.5), the activity of Pseudomonas denitrificans was not detected. It is possible that the activity of Pseudomonas denitrificans would be low under disposal condition.
9.5), the activity of Pseudomonas denitrificans was not detected. It is possible that the activity of Pseudomonas denitrificans would be low under disposal condition.
Iwase, Masanori*
JNC TJ8400 2000-063, 78 Pages, 2000/03
This study is aimed at controlling oxidation reaction of molten metal by ash in incineration systems, and at positively utilizing the oxidation reaction for decontamination of slag. In this year, in order to investigate physico-chemical properties of mixed fused salt containing alkali sulfates, with special focus on the behaviour of oxygen anion in the melts, Cu / Cu
/ Cu redox equilibrium experiments were carried out. Among the effect of various parameters on Cu
redox equilibrium experiments were carried out. Among the effect of various parameters on Cu / Cu
/ Cu ratio in binary and ternary alkali sulfate melts, the effect of partial pressures of oxygen and SO
ratio in binary and ternary alkali sulfate melts, the effect of partial pressures of oxygen and SO was mainly investigated in the study. Variation in Cu
was mainly investigated in the study. Variation in Cu / Cu
/ Cu ratio were presented as the function of partial pressures of oxygen and SO
ratio were presented as the function of partial pressures of oxygen and SO , respectively. Possible thermodynamic interpretation were made on the experimental results. In addition, the dissolution of Cr
, respectively. Possible thermodynamic interpretation were made on the experimental results. In addition, the dissolution of Cr O
O in mixed alkali sulfates were also investigated as a first step to elucidate the mechanism of hot corrosion. With this investigation, an important finding was obtained that the solubility of Cr
in mixed alkali sulfates were also investigated as a first step to elucidate the mechanism of hot corrosion. With this investigation, an important finding was obtained that the solubility of Cr O
O for melts with same average ionic radius, in other words, oxygen ion activity, were essentially identical under constant temperature and atmosphere.
for melts with same average ionic radius, in other words, oxygen ion activity, were essentially identical under constant temperature and atmosphere.
J. A. BERRY*; M. BROWNSWORD*; D. J. ILETT*; Linklater, C. M.*; Mason, C.*; TWEED, C. J.*
JNC TJ8400 2000-060, 60 Pages, 2000/02
Batch sorption experiments have been carried out to investigate the sorption behaviour of plutonium onto basalt and sandstone from the appropriate rock-equilibrated waters under different redox eonditions. Redox Potentials in solution were controlled by the addition of two reducing agents and one oxidising agent. Thermodynamic chemical modelling was undertaken to interpret the results. The sorption models were based on iron oxide. They adequately reproduced the data for sorption of plutonium onto sandstone, but tended to underpredict sorption onto basalt.
Miki, Takahito*; Sasamoto, Hiroshi; Chiba, Tamotsu*; Inagaki, Manabu*; Yui, Mikazu
JNC TN8400 2000-007, 32 Pages, 2000/01
This report presents a summary of literature survey about geochemical reactions which are important to evaluate the redox conditions in the near field rock mass and buffer. The results of literature survey are summarized as follows; (1)Minerals including ferrous iron and organic materials in the rock mass are important reductants. Initial stage after closure of repository, oxygen will be consumed by pyrite, because the reaction rate between pyrite and oxygen is relatively fast. (2)It is possible to estimate the redox capacity for reductants by rock (mineral)-water iteraction experiment in a laboratory. And it is expected that the ferrous iron-rich rock and higher porosity rock may have bigger redox capacity. (3)It is possible to estimate the oxygen consumption rate by reductants such as minerals including ferrous iron. The rate law and rate constant for the oxidation reaction of ferrous iron in the solution are also determined. As a conclusion, it seems that we can evaluate kinetically the evolution of geochemical conditions in the near field rock mass and buffer by excavation of drifts, based on data derived from these existing literatures.
Yamaguchi, Tetsuji
Genankyo Dayori, (173), p.11 - 14, 2000/01
no abstracts in English
Yamaguchi, Tetsuji
Genshiryoku Bakkuendo Kenkyu, 6(1), p.147 - 149, 1999/12
no abstracts in English