Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Chiera, N. M.*; Sato, Tetsuya; Eichler, R.*; Tomitsuka, Tomohiro; Asai, Masato; Adachi, Sadia*; Dressler, R.*; Hirose, Kentaro; Inoue, Hiroki*; Ito, Yuta; et al.
Angewandte Chemie; International Edition, 60(33), p.17871 - 17874, 2021/08
Times Cited Count:3 Percentile:14.19(Chemistry, Multidisciplinary)The formation and the chemical characterization of single atoms of dubnium (Db, element 105), in the form of its volatile oxychloride, was investigated using the on-line gas phase chromatography technique, in the temperature range 350 - 600  C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl
C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl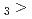 TaOCl
TaOCl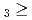 DbOCl
DbOCl . From the obtained experimental results, thermochemical data for DbOCl
. From the obtained experimental results, thermochemical data for DbOCl were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:72.98(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 Be(
Be(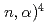 He reaction and the cosmological lithium problem; Measurement of the cross section in a wide energy range at n_TOF at CERN
He reaction and the cosmological lithium problem; Measurement of the cross section in a wide energy range at n_TOF at CERNBarbagallo, M.*; Musumarra, A.*; Cosentino, L.*; Maugeri, E.*; Heinitz, S.*; Mengoni, A.*; Dressler, R.*; Schumann, D.*; K ppeler, F.*; Harada, Hideo; et al.
ppeler, F.*; Harada, Hideo; et al.
Physical Review Letters, 117(15), p.152701_1 - 152701_7, 2016/10
Times Cited Count:85 Percentile:94.8(Physics, Multidisciplinary) at the single-atom level
at the single-atom levelSteinegger, P.*; Asai, Masato; Dressler, R.*; Eichler, R.*; Kaneya, Yusuke*; Mitsukai, Akina*; Nagame, Yuichiro; Piguet, D.*; Sato, Tetsuya; Sch del, M.; et al.
del, M.; et al.
Journal of Physical Chemistry C, 120(13), p.7122 - 7132, 2016/04
Times Cited Count:23 Percentile:60.41(Chemistry, Physical)A new experimental method "vacuum chromatography" has been developed to measure adsorption enthalpy of superheavy elements, and its feasibility has been examined using short-lived thallium isotopes. The short-lived thallium isotopes were produced at the JAEA tandem accelerator. The thallium ion beam prepared with an on-line isotope separator which ionized and mass-separated the thallium isotopes was injected into an isothermal vacuum chromatography apparatus. A temperature-dependent adsorption property of thallium atom on SiO surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
 Ca+
Ca+ U
U 
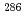 112
112 studied at the GSI-SHIP
studied at the GSI-SHIPHofmann, S.*; Ackermann, D.*; Antalic, S.*; Burkhard, H. G.*; Comas, V. F.*; Dressler, R.*; Gan, Z.*; Heinz, S.*; Heredia, J. A.*; He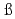 berger, F. P.*; et al.
berger, F. P.*; et al.
European Physical Journal A, 32(3), p.251 - 260, 2007/06
Times Cited Count:262 Percentile:99.7(Physics, Nuclear)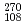 Hs
Hs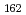
Dvorak, J.*; Br chle, W.*; Chelnokov, M.*; Dressler, R.*; D
chle, W.*; Chelnokov, M.*; Dressler, R.*; D llmann, Ch. E.*; Eberhardt, K.*; Gorshkov, V.*; J
llmann, Ch. E.*; Eberhardt, K.*; Gorshkov, V.*; J ger, E.*; Kr
ger, E.*; Kr cken, R.*; Kuznetsov, A.*; et al.
cken, R.*; Kuznetsov, A.*; et al.
Physical Review Letters, 97(24), p.242501_1 - 242501_4, 2006/12
Times Cited Count:152 Percentile:96.01(Physics, Multidisciplinary)no abstracts in English