Effects of overpack corrosion on redox potential of bentonite pore water under geological disposal environment; Important parameter acquisition and a preliminary 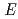 h analysis (Contract research)
h analysis (Contract research)
Otsuka, Ichiro; Taki, Hiroshi*; Yamaguchi, Tetsuji  ; Iida, Yoshihisa
; Iida, Yoshihisa  ; Yamada, Fumika; Inada, Daisuke*; Tanaka, Tadao
; Yamada, Fumika; Inada, Daisuke*; Tanaka, Tadao 
The influence of carbon steel overpack corrosion on redox potential (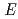 h) of bentonite pore water under geological disposal environment was investigated. The thermodynamics data of corrosion products, the corrosion rate of carbon steel, and the information on cathode reactions were acquired by experiments and literature survey. We conducted preliminary analysis of
h) of bentonite pore water under geological disposal environment was investigated. The thermodynamics data of corrosion products, the corrosion rate of carbon steel, and the information on cathode reactions were acquired by experiments and literature survey. We conducted preliminary analysis of 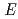 h, ascertained the validity of Phreeq C and identified important points on the analysis. Results were summarized as follows. (1) Thermodynamic data of Fe
h, ascertained the validity of Phreeq C and identified important points on the analysis. Results were summarized as follows. (1) Thermodynamic data of Fe , FeOH
, FeOH , Fe(OH)
, Fe(OH) (aq), Fe(OH)
(aq), Fe(OH)
 , Fe(OH)
, Fe(OH)
 , Fe
, Fe , FeS
, FeS (pyrite), FeCO
(pyrite), FeCO (siderite),Fe(OH)
(siderite),Fe(OH) (s), Fe
(s), Fe O
O (magnetite), Fe(cr) were determined by literature survey. The solubility product of Fe
(magnetite), Fe(cr) were determined by literature survey. The solubility product of Fe CO
CO (OH)
(OH) was determined experimentally, and thermodynamic data were estimated. (2) The corrosion rate of carbon steel was obtained as a function of pH and sulfide ion concentration. (3) After corrosion tests of carbon steel, no CH
was determined experimentally, and thermodynamic data were estimated. (2) The corrosion rate of carbon steel was obtained as a function of pH and sulfide ion concentration. (3) After corrosion tests of carbon steel, no CH , HS
, HS and H
and H S, the reduction product of CO
S, the reduction product of CO
 and SO
and SO
 ,were not detected in liquid and gas phases. (4) Preliminary analysis showed that the redox couple changed as HS
,were not detected in liquid and gas phases. (4) Preliminary analysis showed that the redox couple changed as HS (aq)/SO
(aq)/SO
 , CH
, CH (aq)/CO
(aq)/CO
 , H
, H (aq)/H
(aq)/H O during the evaluation period. After 1000 years,
O during the evaluation period. After 1000 years, 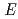 h attained about -500 to -600 mV (vs. NHE) or -750 mV controlled by CH
h attained about -500 to -600 mV (vs. NHE) or -750 mV controlled by CH (aq)/CO
(aq)/CO
 ,or H
,or H (aq)/H
(aq)/H O, respectively.
O, respectively.