Experiments on the release of gaseous iodine from  -irradiated aqueous CsI solution and influence of oxygen and Methyl Isobutyl Ketone (MIBK)
-irradiated aqueous CsI solution and influence of oxygen and Methyl Isobutyl Ketone (MIBK)
Moriyama, Kiyofumi; Tashiro, Shinsuke  ; Chiba, Noriaki; Hirayama, Fumio*; Maruyama, Yu
; Chiba, Noriaki; Hirayama, Fumio*; Maruyama, Yu  ; Nakamura, Hideo
; Nakamura, Hideo 
 ; Watanabe, Atsushi*
; Watanabe, Atsushi*
The volatile iodine production due to radiation chemistry is an important uncertainty source in the source term evaluation for LWRs. The gaseous release of molecular iodine and organic iodine from  -irradiated (
-irradiated (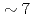 kGy/h, 2h) cesium iodide aqueous solution (1E-4M) containing methyl-isobuthyl-ketone (MIBK) was measured. The solution was buffered at pH
kGy/h, 2h) cesium iodide aqueous solution (1E-4M) containing methyl-isobuthyl-ketone (MIBK) was measured. The solution was buffered at pH 7. The concentration of MIBK (up to 1E-3M) and oxygen were changed as parameters. The total iodine release fraction and the fraction released as organic iodine were 2-47% and 0.02-1.5%, respectively, at the end of the irradiation. With the same cover gas condition, the total iodine release decreased and the organic iodine release increased when the MIBK concentration increased. This behavior can be explained by branching of the reaction path of organic degradation depending on availability of dissolved oxygen and competition between iodine and organic compounds on the consumption of radicals.
7. The concentration of MIBK (up to 1E-3M) and oxygen were changed as parameters. The total iodine release fraction and the fraction released as organic iodine were 2-47% and 0.02-1.5%, respectively, at the end of the irradiation. With the same cover gas condition, the total iodine release decreased and the organic iodine release increased when the MIBK concentration increased. This behavior can be explained by branching of the reaction path of organic degradation depending on availability of dissolved oxygen and competition between iodine and organic compounds on the consumption of radicals.