Synthesis of radiohalogen-labeled peptides with high affinity to HER2/neu receptor
Sasaki, Ichiro; Yamada, Keiichi*; Watanabe, Shigeki; Hanaoka, Hirofumi*; Sugo, Yumi; Oku, Hiroyuki*; Ishioka, Noriko
Radioisotope (RI)-labeled peptides which have the high affinity to receptors on surface of the tumor cell are promising for diagnostic radiography (PET and SPECT) and radiotherapy. MARSGL (H-Met -Ala
-Ala -Arg
-Arg -Ser
-Ser -Gly
-Gly -Leu
-Leu -OH), a linear hexapeptide consisting of six amino acids, has the high affinity to HER2/neu receptor overexpressing in various cancer cells. Thus, radiolabeled MARSGL has potential for the above mentioned purposes. Radiohalogens have various useful radionuclides, which could be introduced to aromatic compounds via tin-halogen exchange reaction. In this study, stannylated peptide Boc-F(
-OH), a linear hexapeptide consisting of six amino acids, has the high affinity to HER2/neu receptor overexpressing in various cancer cells. Thus, radiolabeled MARSGL has potential for the above mentioned purposes. Radiohalogens have various useful radionuclides, which could be introduced to aromatic compounds via tin-halogen exchange reaction. In this study, stannylated peptide Boc-F(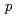 -SnBu
-SnBu )MAR(Pbf)S(
)MAR(Pbf)S(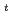 Bu)GL-OH was synthesized to prepare radiohalogen (
Bu)GL-OH was synthesized to prepare radiohalogen (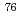 Br or
Br or  I)-labeled FMARSGL derivatives. First, Boc-Phe(
I)-labeled FMARSGL derivatives. First, Boc-Phe(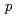 -SnBu
-SnBu )-OH was prepared from Boc-Phe(
)-OH was prepared from Boc-Phe(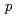 -I)-OMe via Pd(0)-catalyzed coupling reaction with (Bu
-I)-OMe via Pd(0)-catalyzed coupling reaction with (Bu Sn)
Sn) and was successfully introduced into
and was successfully introduced into  -terminal of H-MARSGL-Trt(2-Cl) resin synthesized from H-Leu-Trt(2-Cl) resin by Fmoc-SPPS. After cleavage reaction of Boc-F(
-terminal of H-MARSGL-Trt(2-Cl) resin synthesized from H-Leu-Trt(2-Cl) resin by Fmoc-SPPS. After cleavage reaction of Boc-F(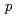 -SnBu
-SnBu )MAR(Pbf)S(
)MAR(Pbf)S(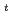 Bu)GL-Trt(2-Cl) resin with 25% 1,1,1,3,3,3-hexafluoro-2-propanol in CH
Bu)GL-Trt(2-Cl) resin with 25% 1,1,1,3,3,3-hexafluoro-2-propanol in CH Cl
Cl , Boc-F(
, Boc-F(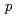 -SnBu
-SnBu )MAR(Pbf)S(
)MAR(Pbf)S(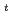 Bu)GL-OH was obtained and identified by using ESI-MS and NMR. Synthesis of
Bu)GL-OH was obtained and identified by using ESI-MS and NMR. Synthesis of  I-labeled FMARSGL is due to report in this presentation.
I-labeled FMARSGL is due to report in this presentation.