In situ electrochemical, electrochemical quartz crystal microbalance, scanning tunneling microscopy, and surface X-ray scattering studies on Ag/AgCl reaction at the underpotentially deposited Ag bilayer on the Au(111) electrode surface
Au(111)電極上でのアンダーポテンシャル電析反応におけるAg/AgCl反応に関する研究
魚崎 浩平*; 森田 潤*; 勝崎 友子*; 高草木 達*; 田村 和久 

 ; 高橋 正光; 水木 純一郎; 近藤 敏啓*
; 高橋 正光; 水木 純一郎; 近藤 敏啓*
Uosaki, Kohei*; Morita, Jun*; Katsuzaki, Tomoko*; Takakusagi, Satoru*; Tamura, Kazuhisa; Takahashi, Masamitsu; Mizuki, Junichiro; Kondo, Toshihiro*
UPD反応により作成したAgバイレーヤー上でのAg/AgCl反応について、EQCM, STM, SXS及び電気化学測定を用いて追跡した。その結果、最初Cl-が(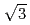

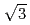 )構造でAg上に吸着し、次に(
)構造でAg上に吸着し、次に(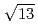

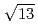 )R13.9
)R13.9 の構造をもつAgCl層を形成することがわかった。さらにAgCl層は(4
の構造をもつAgCl層を形成することがわかった。さらにAgCl層は(4 4)構造に変化した。
4)構造に変化した。
Ag/AgCl reaction at the Ag bilayer, which was underpotentially prepared on a Au(111) surface, was investigated using electrochemical quartz crystal microbalance (EQCM), scanning tunneling microscopy (STM), surface X-ray scattering (SXS), and electrochemical techniques. When the potential was scanned positively from -200 mV, the Cl ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl
ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl ion layer from random orientation to (
ion layer from random orientation to (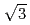

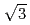 ) structure took place at around +130 mV. The Ag bilayer and Cl
) structure took place at around +130 mV. The Ag bilayer and Cl ions were oxidatively reacted to form the AgCl monolayer with (
ions were oxidatively reacted to form the AgCl monolayer with (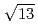

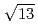 )
) 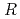 13.9
13.9 structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (
structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (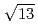

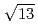 )R13.9
)R13.9 structure to (4
structure to (4 4) structure around +500 mV.
4) structure around +500 mV.