Crystal structure of a cold-adapted cellulase
Tamada, Taro; Arimori, Takao*; Fukuhara, Hiroaki*; Ito, Akihiro*; Ueda, Mitsuhiro*
The saccharification process is essential for bioethanol production from woody biomass including celluloses. Cold-adapted cellulase, which has sufficient activity at low temperature ( 293 K), is capable of reducing heating costs during the saccharification process and is suitable for simultaneous saccharification and fermentation. Endo-1,4-
293 K), is capable of reducing heating costs during the saccharification process and is suitable for simultaneous saccharification and fermentation. Endo-1,4- -glucanase from the earthworm Eisenia fetida (EF-EG2) belonging to glycoside hydrolase family 9 has been shown to have the highest activity at 313 K, and also retained a comparatively high activity at 283 K. The recombinant EF-EG2, expressed in
-glucanase from the earthworm Eisenia fetida (EF-EG2) belonging to glycoside hydrolase family 9 has been shown to have the highest activity at 313 K, and also retained a comparatively high activity at 283 K. The recombinant EF-EG2, expressed in 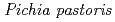 , was purified and then grew needle-shaped crystals with dimensions of 0.02
, was purified and then grew needle-shaped crystals with dimensions of 0.02  0.02
0.02  1 mm. The final model of EF-EG2, including 435 residues, two ions, seven crystallization reagents and 696 waters, was refined to a crystallographic
1 mm. The final model of EF-EG2, including 435 residues, two ions, seven crystallization reagents and 696 waters, was refined to a crystallographic 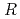 -factor of 14.7% (free
-factor of 14.7% (free 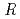 -factor of 16.8%) to 1.5
-factor of 16.8%) to 1.5  resolution. The overall structure of EF-EG2 has an(
resolution. The overall structure of EF-EG2 has an( /
/ )
) barrel fold which contains a putative active-site cleft and a negatively charged surface. Furthermore, in the structure of the E431Q mutant of EF-EG2 complexed with cellotriose (CTR), electron densities corresponding to CTR are observed well at -4 to -2 subsites. This structural information helps us understand the catalytic and cold adaptation mechanisms of EF-EG2.
barrel fold which contains a putative active-site cleft and a negatively charged surface. Furthermore, in the structure of the E431Q mutant of EF-EG2 complexed with cellotriose (CTR), electron densities corresponding to CTR are observed well at -4 to -2 subsites. This structural information helps us understand the catalytic and cold adaptation mechanisms of EF-EG2.