Production of highly purified no-carrier-added  Lu for radioimmunotherapy
Lu for radioimmunotherapy
Watanabe, Satoshi; Hashimoto, Kazuyuki; Watanabe, Shigeki; Iida, Yasuhiko*; Hanaoka, Hirofumi*; Endo, Keigo*; Ishioka, Noriko
No-carrier-added  Lu produced via the
Lu produced via the 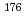 Yb(n,
Yb(n,  )
)  Yb
Yb 
 Lu process was separated from the macroscopic amounts of the Yb target using reversed-phase ion-pair liquid chromatography. To produce a highly purified
Lu process was separated from the macroscopic amounts of the Yb target using reversed-phase ion-pair liquid chromatography. To produce a highly purified  Lu solution capable of labeling antibodies, the metallic impurities were removed using cation, chelating ion, and anion exchange columns. After the elimination of metallic impurities, the concentrations of Ca, Fe, and Zn were reduced from 87, 340, and 77 ppb to 13, 18, and 9 ppb, respectively. Consequently, the labeling yield of the
Lu solution capable of labeling antibodies, the metallic impurities were removed using cation, chelating ion, and anion exchange columns. After the elimination of metallic impurities, the concentrations of Ca, Fe, and Zn were reduced from 87, 340, and 77 ppb to 13, 18, and 9 ppb, respectively. Consequently, the labeling yield of the  Lu -labeled antibody increased from less than 5% to 88%.
Lu -labeled antibody increased from less than 5% to 88%.