Atomically dispersed Pt on the surface of Ni particles; Synthesis and catalytic function in hydrogen generation from aqueous ammonia-borane
ニッケル粒子上に原子分散したプラチナ; 合成法およびアンモニアボランからの水素生成触媒能
Li, Z.*; He, T.*; 松村 大樹 

 ; Miao, S.*; Wu, A.*; Liu, L.*; Wu, G.*; Chen, P.*
; Miao, S.*; Wu, A.*; Liu, L.*; Wu, G.*; Chen, P.*
Li, Z.*; He, T.*; Matsumura, Daiju; Miao, S.*; Wu, A.*; Liu, L.*; Wu, G.*; Chen, P.*
We report herein that the single-atom alloy (SAA) made of atomically dispersed Pt on the surface of Ni particles (Pt is surrounded by Ni atoms) exhibits improved catalytic activity on the hydrolytic dehydrogenation of ammonia-borane, a promising hydrogen storage method for onboard applications. Specifically, an addition of 160 ppm of Pt leads to ca. 3-fold activity improvement in comparison to that of pristine Ni/CNT catalyst. The turnover frequency based on the isolated Pt is 12000 mol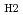 mol
mol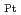
 min
min , which is about 21 times the value of the best Pt-based catalyst ever reported. Our simulation results indicate that the high activity achieved stems from the synergistic effect between Pt and Ni, where the negatively charged Pt (Pt
, which is about 21 times the value of the best Pt-based catalyst ever reported. Our simulation results indicate that the high activity achieved stems from the synergistic effect between Pt and Ni, where the negatively charged Pt (Pt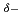 ) and positively charged Ni (Ni
) and positively charged Ni (Ni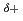 ) in the Pt-Ni alloy are prone to interact with H and OH of H
) in the Pt-Ni alloy are prone to interact with H and OH of H O molecules, respectively, leading to an energetically favorable reaction pathway.
O molecules, respectively, leading to an energetically favorable reaction pathway.