On-line isothermal gas chromatographic behavior of oxychlorides of group-5 elements, Nb, Ta, and Db
Sato, Tetsuya 

 ; Chiera, N. M.*; Tomitsuka, Tomohiro; Tokoi, Katsuyuki*; Suzuki, Hayato*; Ito, Yuta
; Chiera, N. M.*; Tomitsuka, Tomohiro; Tokoi, Katsuyuki*; Suzuki, Hayato*; Ito, Yuta 

 ; Asai, Masato
; Asai, Masato 
 ; Shirai, Kaori*; Inoue, Hiroki*; Adachi, Sadia*; Kashihara, Ayuna*; Makii, Hiroyuki
; Shirai, Kaori*; Inoue, Hiroki*; Adachi, Sadia*; Kashihara, Ayuna*; Makii, Hiroyuki 

 ; Hirose, Kentaro
; Hirose, Kentaro 
 ; Nishio, Katsuhisa
; Nishio, Katsuhisa 

 ; Nagame, Yuichiro*
; Nagame, Yuichiro*  ; Watanabe, Eisuke*; Sakama, Minoru*; Goto, Shinichi*
; Watanabe, Eisuke*; Sakama, Minoru*; Goto, Shinichi*
The influence of strong relativistic effects on chemical properties has been interesting in the superheavy element region. Their chemical properties, however, have not been investigated sufficiently because of experimental difficulties owing to their low production rates and short half-lives. In order to elucidate the chemical properties of dubnium (Db, Z = 105), we have conducted on-line isothermal gas chromatographic experiments of oxychloride of group-5 elements. We confirmed the formation of volatile oxychlorides of Db and its lighter homologs Nb and Ta by using  Db (half-life,
Db (half-life, 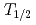 = 33.8 s),
= 33.8 s),  Nb (
Nb (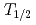 = 14.5 min.), and
= 14.5 min.), and 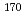 Ta (
Ta (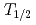 = 6.76 min.), respectively. We successfully determined the adsorption enthalpies of the oxychlorides of each element on the quartz surface from their isothermal gas chromatographic behavior. The obtained volatility sequence of the group-5 elements is found to be Nb
= 6.76 min.), respectively. We successfully determined the adsorption enthalpies of the oxychlorides of each element on the quartz surface from their isothermal gas chromatographic behavior. The obtained volatility sequence of the group-5 elements is found to be Nb  Ta
Ta  Db.
Db.