Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Toyomori, Yuka*; Tsuji, Satoru*; Mitsuda, Shinobu*; Okayama, Yoichi*; Ashida, Shiomi*; Mori, Atsunori*; Kobayashi, Toru; Miyazaki, Yuji; Yaita, Tsuyoshi; Arae, Sachie*; et al.
Bulletin of the Chemical Society of Japan, 89(12), p.1480 - 1486, 2016/09
Times Cited Count:9 Percentile:29.91(Chemistry, Multidisciplinary)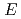 -alkene bearing bisazole component formed by sequential C-H homocoupling and ring-closing metathesis
-alkene bearing bisazole component formed by sequential C-H homocoupling and ring-closing metathesisNishio, Shotaro*; Somete, Takashi*; Sugie, Atsushi*; Kobayashi, Toru; Yaita, Tsuyoshi; Mori, Atsunori*
Organic Letters, 14(10), p.2476 - 2479, 2012/05
Times Cited Count:26 Percentile:62.83(Chemistry, Organic)Inaba, Yusuke*; Tsumagari, Takayuki*; Kida, Tatsuya*; Watanabe, Wataru*; Nakajima, Yasutaka*; Fukuoka, Sachio*; Mori, Atsunori*; Matsumura, Tatsuro; Nakano, Yoshio*; Takeshita, Kenji*
Polymer Journal, 43(7), p.630 - 634, 2011/07
Times Cited Count:11 Percentile:34.06(Polymer Science) -(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing a polymerizable double bond in the substituent structure of the pyridine ring are synthesized and subjected to copolymerization with
-(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing a polymerizable double bond in the substituent structure of the pyridine ring are synthesized and subjected to copolymerization with  -isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA) gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing C3, C4, C10 and branched C3 spacer chains are synthesized and temperature-dependent extraction behavior of cadmium ion is compared. These gels extract Cd(II) ion efficiently from the aqueous solution in the swelling state at 5
-isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA) gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing C3, C4, C10 and branched C3 spacer chains are synthesized and temperature-dependent extraction behavior of cadmium ion is compared. These gels extract Cd(II) ion efficiently from the aqueous solution in the swelling state at 5 C, while little extraction is observed at 45
C, while little extraction is observed at 45 C with shrinking. It is found that poly(TPEN-NIPA) gel of branched C3 spacer (C3b) shows the excellent thermoresponsive extraction performance.
C with shrinking. It is found that poly(TPEN-NIPA) gel of branched C3 spacer (C3b) shows the excellent thermoresponsive extraction performance.
Matsumura, Tatsuro; Inaba, Yusuke*; Mori, Atsunori*; Takeshita, Kenji*
Journal of Nuclear Science and Technology, 47(2), p.123 - 126, 2010/02
Times Cited Count:13 Percentile:63.45(Nuclear Science & Technology)We are developing a new MA/Ln separation process with TPEN and its derivatives for P&T technology. TPEN is a hexadentate soft-donor ligand that has six soft-donor sites as a kind of podand type molecule and can encapsulate a metal ion. TPEN has good selectivity of Am(III) from Ln(III) and has potential to establish partitioning of MA. From the viewpoint of practical application, the hydrophobicity of TPEN must be improved. Because the dissolution of a slight amount of TPEN (about 10 mol/l) to water will restrict decontamination of Ln in extraction process. In this study, the hydrophobicity of TPEN was improved by introducing alkyl groups and the effect of the introduction of alkyl groups on the separation of Am(III) and Eu(III) was examined. One of them, N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine, TBPEN, showed good selectivity and the maximum separation factor, SF
mol/l) to water will restrict decontamination of Ln in extraction process. In this study, the hydrophobicity of TPEN was improved by introducing alkyl groups and the effect of the introduction of alkyl groups on the separation of Am(III) and Eu(III) was examined. One of them, N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine, TBPEN, showed good selectivity and the maximum separation factor, SF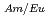 , was 91 at pH 3.02. The hydrophobicity of TBPEN is checked by the distribution data between water and chloroform, and improved successfully. The approach for development of hydrophobic derivatives of TPEN was effective.
, was 91 at pH 3.02. The hydrophobicity of TBPEN is checked by the distribution data between water and chloroform, and improved successfully. The approach for development of hydrophobic derivatives of TPEN was effective.
Fukuoka, Sachio*; Kida, Tatsuya*; Nakajima, Yasutaka*; Tsumagari, Takayuki*; Watanabe, Wataru*; Inaba, Yusuke*; Mori, Atsunori*; Matsumura, Tatsuro; Nakano, Yoshio*; Takeshita, Kenji*
Tetrahedron, 66(9), p.1721 - 1727, 2010/02
Times Cited Count:18 Percentile:47.28(Chemistry, Organic)N,N,N',N'-(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing the different number (1-4) of a double bond moiety on the pyridine ring are synthesized and subjected to copolymerization with N-isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA)gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing three or four double bonds exhibit temperature-dependent change of swelling and shrinking in water. These gels extract CdII ion efficiently from the aqueous solution in the swelling state at 5 C, while little extraction was observed at 45
C, while little extraction was observed at 45 C with shrinking.
C with shrinking.
Matsumura, Tatsuro; Takeshita, Kenji*; Inaba, Yusuke*; Mori, Atsunori*
Proceedings of 10th OECD/NEA Information Exchange Meeting on Actinide and Fission Product Partitioning and Transmutation (CD-ROM), 6 Pages, 2010/00
no abstracts in English
 -tetrakis((5-alkoxypyridin-2-yl)methyl)ethylenediamine, TRPEN, effective ligands for the separation of trivalent minor actinides from lanthanides
-tetrakis((5-alkoxypyridin-2-yl)methyl)ethylenediamine, TRPEN, effective ligands for the separation of trivalent minor actinides from lanthanidesMatsumura, Tatsuro; Inaba, Yusuke*; Koma, Yoshikazu; Morita, Yasuji; Mori, Atsunori*; Takeshita, Kenji*
Proceedings of International Conference on Advanced Nuclear Fuel Cycle; Sustainable Options & Industrial Perspectives (Global 2009) (CD-ROM), p.1107 - 1112, 2009/09
no abstracts in English
Matsumura, Tatsuro; Inaba, Yusuke*; Takeshita, Kenji*; Mori, Atsunori*
Proceedings of International Solvent Extraction Conference "Solvent Extraction-Fundamentals to Industrial Applications" (ISEC 2008), 4 Pages, 2008/09
We are developing a new MA/Ln separation process with derivatives of TPEN (N,N,N',N'-tetrakis(2-pyridylmethyl)ethylenediamine) for P&T technology. TPEN has good selectivity of Am(III) from Ln(III). However, TPEN is soluble slightly (about 10 mol/l) to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize several new hydrophobic derivative of TPEN. One of the ligands, TDdPEN (N,N,N',N'-tetrakis((5-dodecyloxypyridin-2-yl)-methyl)ethylenediamine) showed high selectivity. The maximum separation factor, SF
mol/l) to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize several new hydrophobic derivative of TPEN. One of the ligands, TDdPEN (N,N,N',N'-tetrakis((5-dodecyloxypyridin-2-yl)-methyl)ethylenediamine) showed high selectivity. The maximum separation factor, SF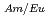 , was over 800. Hydrophobic derivatives of TPEN that have potential of application to the MA/Ln separation process was synthesized successfully.
, was over 800. Hydrophobic derivatives of TPEN that have potential of application to the MA/Ln separation process was synthesized successfully.
Takeshita, Kenji*; Nakano, Yoshio*; Matsumura, Tatsuro; Mori, Atsunori*
Proceedings of 3rd International ATALANTE Conference (ATALANTE 2008) (CD-ROM), 8 Pages, 2008/05
A thermal-swing chromatographic process using a thermosensitive gel copolymerized with NIPA (N-isopropylacrylamide) and TPPEN (N,N,N',N'-tetrakis(4-propenyloxy-2-pyridylmethyl)ethylenediamine) was studied for the separation of Am(III) from Eu(III). Firstly, the radiolysis of the TPPEN-NIPA gel was tested by the  -ray irradiation and the
-ray irradiation and the  nuclide adsorption. The extraction separation of Am(III) was not influenced in the radioactive environment of the proposed process. Next, the TPPEN-NIPA gel was immobilized in porous silica particles and the applicability of the gel-immobilized silica to the proposed process was tested. Am(III) was extracted selectively in the gel-immobilized silica at 5
nuclide adsorption. The extraction separation of Am(III) was not influenced in the radioactive environment of the proposed process. Next, the TPPEN-NIPA gel was immobilized in porous silica particles and the applicability of the gel-immobilized silica to the proposed process was tested. Am(III) was extracted selectively in the gel-immobilized silica at 5 C and the separation factor of Am(III) over Eu(III) was evaluated to be 3.7. The distribution ratio of Am(III) was reduced to less than 1/20 by increasing temperature from 5
C and the separation factor of Am(III) over Eu(III) was evaluated to be 3.7. The distribution ratio of Am(III) was reduced to less than 1/20 by increasing temperature from 5 C to 40
C to 40 C. These results indicate that the TPPEN-NIPA gel is applicable to the thermal-swing chromatographic process for the MA recovery.
C. These results indicate that the TPPEN-NIPA gel is applicable to the thermal-swing chromatographic process for the MA recovery.
Matsumura, Tatsuro; Takeshita, Kenji*; Mori, Atsunori*
Proceedings of 3rd International ATALANTE Conference (ATALANTE 2008) (CD-ROM), 4 Pages, 2008/05
We are developing a new MA/Ln separation process with derivatives of TPEN (N,N,N',N'-tetrakis(2-pyridylmethyl)ethylenediamine) for P&T technology. TPEN is a hexadentate ligand, which has good selectivity of Am(III) from Ln(III). However, there is a serious problem for the practical application. This is to the dissolution of a slight amount of TPEN (about 10 mol/l) to water. High enrichment of Am(III) will be restricted by the dissolution of TPEN to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize a new hydrophobic derivative of TPEN, TBPEN (N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine. It showed good selectivity and the maximum separation factor, SF
mol/l) to water. High enrichment of Am(III) will be restricted by the dissolution of TPEN to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize a new hydrophobic derivative of TPEN, TBPEN (N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine. It showed good selectivity and the maximum separation factor, SF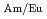 , was 91 at pH 3.0. A hydrophobic derivative of TPEN that has potential of application to the MA/Ln separation process was synthesized successfully.
, was 91 at pH 3.0. A hydrophobic derivative of TPEN that has potential of application to the MA/Ln separation process was synthesized successfully.
Takeshita, Kenji*; Fugate, G.*; Matsumura, Tatsuro; Nakano, Yoshio*; Mori, Atsunori*; Fukuoka, Sachio*
Proceedings of International Conference on Advanced Nuclear Fuel Cycles and Systems (Global 2007) (CD-ROM), p.120 - 125, 2007/09
Extraction separation of Am(III) and Eu(III) was examined by the thermal-swing extraction technique using a thermosensitive gel, poly- -isopropylacrylamide (NIPA) copolymerized with a TPEN derivative,
-isopropylacrylamide (NIPA) copolymerized with a TPEN derivative, 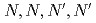 -tetrakis(4-propenyloxy-2-pyridylmethyl)ethylenediamine (TPPEN). The separation of Am(III) from Eu(III) was observed in the swollen state of gel (5
-tetrakis(4-propenyloxy-2-pyridylmethyl)ethylenediamine (TPPEN). The separation of Am(III) from Eu(III) was observed in the swollen state of gel (5 C) and the separation factor of Am(III) was evaluated as about 18 at pH5.2. More than 90% of Am(III) extracted into the gel was released by the volume phase transition of gel from the swollen state (5
C) and the separation factor of Am(III) was evaluated as about 18 at pH5.2. More than 90% of Am(III) extracted into the gel was released by the volume phase transition of gel from the swollen state (5 C) to the shrunken one (40
C) to the shrunken one (40 C). The repetition test for the thermal swing extraction of a soft metal ion, Cd(II), which was used as a substitute of Am(III), was carried out and the extraction and release of Cd(II) were repeated three times stably under the thermal-swing operation between 5
C). The repetition test for the thermal swing extraction of a soft metal ion, Cd(II), which was used as a substitute of Am(III), was carried out and the extraction and release of Cd(II) were repeated three times stably under the thermal-swing operation between 5 C and 40
C and 40 C. The radiation effect of gel on the extraction of Am and Eu was tested by the irradiation of
C. The radiation effect of gel on the extraction of Am and Eu was tested by the irradiation of  -ray (10 kGy) and the long-term adsorption of
-ray (10 kGy) and the long-term adsorption of  -emitter (
-emitter ( Cm). The TPPEN-NIPA gel sustained no damage by these radiation tests. These results suggest that the thermal-swing extraction technique is applicable to the MA partitioning process indispensable for the establishment of P&T technology.
Cm). The TPPEN-NIPA gel sustained no damage by these radiation tests. These results suggest that the thermal-swing extraction technique is applicable to the MA partitioning process indispensable for the establishment of P&T technology.
Matsumura, Tatsuro; Inaba, Yusuke*; Mori, Atsunori*; Takeshita, Kenji*
no journal, ,
no abstracts in English
Suzuki, Shinichi; Akutsu, Kazuhiro; Yaita, Tsuyoshi; Okamoto, Yoshihiro; Ogata, Tsuyoshi*; Takeshita, Kenji*; Inaba, Yusuke*; Ikeda, Atsushi; Kobayashi, Toru; Oaki, Hiroshi*; et al.
no journal, ,
no abstracts in English
Yaita, Tsuyoshi; Kobayashi, Toru; Shuh, D.*; Asano, Towa*; Mori, Atsunori*
no journal, ,
Ogata, Takeshi*; Takeshita, Kenji*; Oaki, Hiroshi*; Inaba, Yusuke*; Mori, Atsunori*; Matsumura, Tatsuro; Nakano, Yoshio*
no journal, ,
N,N,N',N'-tetrakis(2-pyridylmethyl)ethylenediamine (TPEN) is one of the podand-type ligands with six nitrogen-donors and recognize the slight difference in the softness between Am(III) and Eu(III). We have tried to synthesize a novel extraction chromatographic agent, which exploit such feature of TPEN. The use of the polymer gel as a support of the extractant can be expect to suppress the elution of the extractant and keep the flexibility of extractant. N,N,N',N'-tetrakis-(4-propenyloxy-2-pyridylmethyl)ethylenediamine (TPPEN) which is TPEN analog introducing polymerizable group to pyridine ring was used as a cross-linker of N-isopropylacrylamide (NIPA). Furthermore, the TPPEN-NIPA gel on highly porous silica was prepared to improve the mechanical strength of the gel. These extraction chromatographic agents have been tested for the separation of Am(III) and Eu(III). Characterization and extraction performance of the extraction chromatographic agents will be described.
Suzuki, Shinichi; Akutsu, Kazuhiro; Yaita, Tsuyoshi; Shiwaku, Hideaki; Kobayashi, Toru; Takeshita, Kenji*; Ogata, Takeshi*; Inaba, Yusuke*; Oaki, Hiroshi*; Mori, Atsunori*
no journal, ,
no abstracts in English
Asano, Towa*; Kobayashi, Toru; Shimojo, Kojiro; Yamaoka, Seiha*; Torii, Ren*; Okano, Kentaro*; Yaita, Tsuyoshi; Mori, Atsunori*
no journal, ,
no abstracts in English
Matsumura, Tatsuro; Takeshita, Kenji*; Mori, Atsunori*
no journal, ,
no abstracts in English
Suzuki, Shinichi; Kobayashi, Toru; Shiwaku, Hideaki; Yaita, Tsuyoshi; Takeshita, Kenji*; Oaki, Hiroshi*; Mori, Atsunori*
no journal, ,
no abstracts in English
Takeshita, Kenji*; Ogata, Tsuyoshi*; Nakano, Yoshio*; Matsumura, Tatsuro; Mori, Atsunori*
no journal, ,
Extraction separation of Am(III) and Eu(III) was examined by the thermal-swing extraction technique using a thermosensitive gel, NIPA (poly-N-isopropylacrylamide) copolymerized with an encapsulating ligand, TPEN derivative called TPPEN (N,N,N',N'-tetrakis(4-propenyloxy-2-pyridylmethyl)ethylenediamine). The separation of Am(III) from Eu(III) was observed in the swollen state of gel (5 C) and the separation factor of Am(III) was evaluated as about 18 at pH5.2. More than 90% of Am(III) extracted into the gel was released by the volume phase transition of gel from the swollen state (5
C) and the separation factor of Am(III) was evaluated as about 18 at pH5.2. More than 90% of Am(III) extracted into the gel was released by the volume phase transition of gel from the swollen state (5 C) to the shrunken one (40
C) to the shrunken one (40 C). The radiation effect of gel on the extraction of Am and Eu was tested by the irradiation of
C). The radiation effect of gel on the extraction of Am and Eu was tested by the irradiation of  -ray (10 kGy) and the long-term adsorption of
-ray (10 kGy) and the long-term adsorption of  -emitter (
-emitter ( Cm). The TPPEN-NIPA gel sustained no damage by these radiation tests. These results suggest that the thermal-swing extraction technique is applicable to the MA partitioning process indispensable for the establishment of P&T technology.
Cm). The TPPEN-NIPA gel sustained no damage by these radiation tests. These results suggest that the thermal-swing extraction technique is applicable to the MA partitioning process indispensable for the establishment of P&T technology.