Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Uno, Koichiro*; Okumura, Masahiko; Nakao, Atsushi*; Yamaguchi, Akiko; Yanai, Junta*
Science of the Total Environment, 949, p.175012_1 - 175012_8, 2024/11
Times Cited Count:3 Percentile:36.82(Environmental Sciences)Frayed edge sites (FES), formed by the partial weathering of mica minerals, selectively adsorb Cs ions. However, the detailed mechanism of this adsorption has not been fully clarified. In this study, cation extraction and Cs adsorption experiments were conducted on mica. It was found that potassium-adsorbed mica adsorbed more Cs than rubidium-adsorbed mica. To elucidate the cause of this, the stability of Cs, rubidium, and potassium adsorbed on FES was evaluated using first-principles calculations. It was determined that the presence of potassium as the cation species prior to Cs adsorption is important for the stability of Cs on the FES.

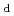 values calculated from radiocesium interception potential
values calculated from radiocesium interception potentialUno, Koichiro*; Nakao, Atsushi*; Okumura, Masahiko; Yamaguchi, Akiko; Kogure, Toshihiro*; Yanai, Junta*
Nihon Dojo Hiryo Gaku Zasshi, 94(5), p.376 - 384, 2023/10
Radiocesium interception potential (RIP) has been widely used as a quantitative indicator of cesium (Cs) adsorption capacity of soil, but it has been found that RIP does not always correlate with the distribution coefficient (
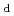 ) of Cs in the actual environment. In order to clarify the cause of this discrepancy, we measured Kd using more realistic solutions, compared it with RIP, and evaluated the mineral structure. As a result, it was found that the concentration of competing cations, such as potassium and ammonium ions, and the structural change of the mineral itself are important.
) of Cs in the actual environment. In order to clarify the cause of this discrepancy, we measured Kd using more realistic solutions, compared it with RIP, and evaluated the mineral structure. As a result, it was found that the concentration of competing cations, such as potassium and ammonium ions, and the structural change of the mineral itself are important.
Uno, Koichiro*; Nakao, Atsushi*; Okumura, Masahiko; Kogure, Toshihiro*; Wagai, Rota*; Yamaguchi, Akiko; Yanai, Junta*
no journal, ,
no abstracts in English
Uno, Koichiro*; Nakao, Atsushi*; Okumura, Masahiko; Kogure, Toshihiro*; Yamaguchi, Akiko; Takeda, Hikaru*; Maruyama, Hayato*; Shinano, Takuro*; Yanai, Junta*
no journal, ,
Biotite inhibits radiocesium (RCs) transfer from soil to plants by two functions of selective adsorption of radiocesium (Cs) between layers (Cs adsorption ability) and K supply from between layers (K supply ability). However, it was difficult to distinguish between the two. In this study, the authors succeeded in preparing biotite in which both functions are variously different by preparing weathered biotite in which interlayer pottasium (K) was replaced with rubidium (Rb) to lose K supply ability. By systematically comparing these biotites, the authors clarified the effects of Cs adsorption ability and K supply ability on Cs transfer to plants.