Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -casein micelles using neutron spectroscopy
-casein micelles using neutron spectroscopyNakagawa, Hiroshi; Appavou, M.-S.*; Wuttke, J.*; Zamponi, M.*; Holderer, O.*; Schrader, T.*; Richter, D.*; Doster, W.*
Joint Annual Report 2021 of the MLZ and FRM II, P. 55, 2022/00
Times Cited Count:0 Percentile:0.01(Biophysics) -casein undergoes a reversible endothermic self-association with increasing temperature, forming micelles of limited size. We characterize the structural flexibility in terms of nano-second molecular motions. We report on two relaxation processes on a nano-second and a sub-nano-second time scale for
-casein undergoes a reversible endothermic self-association with increasing temperature, forming micelles of limited size. We characterize the structural flexibility in terms of nano-second molecular motions. We report on two relaxation processes on a nano-second and a sub-nano-second time scale for  -casein in solution. Both processes are analyzed by Brownian Oscillator model, by which the spring constant can be defined in the isotropic parabolic potential. The slower process seems a characteristic feature of the unfolded structure, and dividing the relaxation time by the solvent viscosity removes most of the temperature dependence, indicating that indicates, the process involves density fluctuations of the solvent. The faster process has a smaller amplitude and requires hydration water. The flexibility of a
-casein in solution. Both processes are analyzed by Brownian Oscillator model, by which the spring constant can be defined in the isotropic parabolic potential. The slower process seems a characteristic feature of the unfolded structure, and dividing the relaxation time by the solvent viscosity removes most of the temperature dependence, indicating that indicates, the process involves density fluctuations of the solvent. The faster process has a smaller amplitude and requires hydration water. The flexibility of a  -casein monomer is preserved in the micelle.
-casein monomer is preserved in the micelle.
 -casein micelles by neutron spectroscopy
-casein micelles by neutron spectroscopyNakagawa, Hiroshi; Appavou, M.-S.*; Wuttke, J.*; Zamponi, M.*; Holderer, O.*; Schrader, T. E.*; Richter, D.*; Doster, W.*
Biophysical Journal, 120(23), p.5408 - 5420, 2021/12
Times Cited Count:1 Percentile:7.79(Biophysics)Casein proteins are characterized by its extended structural features and its dynamics, which are different from those of typical folded proteins, due to a large number of proline residues. We were able to describe the unique structural dynamics by Orenstein-Uhlenbeck Brownian oscillator model by several types of QENS experiments with wide time-scales. The characterized dynamics also allowed us to discuss the effective sequestration of calcium phosphate in terms of its unique dynamical structure. The biological role of the physical processes of the proteins on the nanosecond order on the biological functions is presented.
Hiromoto, Takeshi; Adachi, Motoyasu; Shibazaki, Chie; Schrader, T. E.*; Ostermann, A.*; Kuroki, Ryota
JPS Conference Proceedings (Internet), 8, p.033003_1 - 033003_6, 2015/09
T4 phage lysozyme (T4L) is an endoacetylmuramidase that degrades the murein of the bacterial cell wall by cleaving the  -1,4-glycosidic bond between
-1,4-glycosidic bond between  -acetylmuramic acid and
-acetylmuramic acid and  -acetylglucosamine. We previously reported that the substitution of the catalytic Thr26 with the nucleophilic His converts the wild-type (WT) T4L from an inverting to a retaining glycosidase, in which the
-acetylglucosamine. We previously reported that the substitution of the catalytic Thr26 with the nucleophilic His converts the wild-type (WT) T4L from an inverting to a retaining glycosidase, in which the  -configuration of the substrate is retained in the product. We also found that the Thr26His (T26H) mutant can catalyze a transglycosylation reaction more effectively than hydrolysis, although the WT-T4L has no transglycosidase activity. To clarify the role of the substituted His26 in transglycosylation and to investigate its relationship to the neighboring acidic residue Asp20 using neutron crystallography, a perdeuterated recombinant protein of the T26H mutant (d-T26H) was prepared for crystallization. The perdeuterated form was produced in
-configuration of the substrate is retained in the product. We also found that the Thr26His (T26H) mutant can catalyze a transglycosylation reaction more effectively than hydrolysis, although the WT-T4L has no transglycosidase activity. To clarify the role of the substituted His26 in transglycosylation and to investigate its relationship to the neighboring acidic residue Asp20 using neutron crystallography, a perdeuterated recombinant protein of the T26H mutant (d-T26H) was prepared for crystallization. The perdeuterated form was produced in 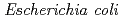 cells cultured in deuterated-rich media. After purification, the d-T26H mutant was crystallized under deuterated conditions and grown to a volume of 0.12 mm
cells cultured in deuterated-rich media. After purification, the d-T26H mutant was crystallized under deuterated conditions and grown to a volume of 0.12 mm using a macroseeding technique. A preliminary neutron-diffraction experiment at 100 K at the FRM II research reactor (Munich, Germany) gave diffraction spots of up to 2.8
using a macroseeding technique. A preliminary neutron-diffraction experiment at 100 K at the FRM II research reactor (Munich, Germany) gave diffraction spots of up to 2.8  resolution after a 1.5 h exposure.
resolution after a 1.5 h exposure.