Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
岡村 浩之; 坂江 広基*; 木谷 径治*; 平山 直紀*; 青柳 登; 斎藤 拓巳*; 下条 晃司郎; 長縄 弘親; 井村 久則*
Polyhedron, 31(1), p.748 - 753, 2012/01
被引用回数:28 パーセンタイル:87.66(Chemistry, Inorganic & Nuclear)The extraction constant and the two-phase stability constant of tris(2-thenoyltrifluoroacetonato) europium(III) between 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]) as an ionic liquid and an aqueous phase were determined by considering the extraction equilibria. Specific solute-solvent interactions between the neutral Eu(III) chelate and [C
N]) as an ionic liquid and an aqueous phase were determined by considering the extraction equilibria. Specific solute-solvent interactions between the neutral Eu(III) chelate and [C mim][Tf
mim][Tf N] molecules were revealed from the relationships between the distribution constant of the enol form of 2-thenoyltrifluoroacetone (Htta) and the distribution constant (
N] molecules were revealed from the relationships between the distribution constant of the enol form of 2-thenoyltrifluoroacetone (Htta) and the distribution constant (
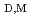 ) of the neutral Eu(III) chelate. The coordination environment of Eu
) of the neutral Eu(III) chelate. The coordination environment of Eu in the neutral Eu(III) chelate in [C
in the neutral Eu(III) chelate in [C mim][Tf
mim][Tf N] was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both methods consistently indicated that not only the Eu(III)chelate extracted but also Eu(tta)
N] was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both methods consistently indicated that not only the Eu(III)chelate extracted but also Eu(tta) (H
(H O)
O) synthesized as a solid crystal were almost completely dehydrated in [C
synthesized as a solid crystal were almost completely dehydrated in [C mim][Tf
mim][Tf N] saturated with water. Consequently, the higher
N] saturated with water. Consequently, the higher 
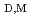 or extractability of the neutral Eu(III) chelate in the [C
or extractability of the neutral Eu(III) chelate in the [C mim][Tf
mim][Tf N] system can be ascribed to the dehydration of the Eu(III) chelate, which is caused by the specific solvation with [C
N] system can be ascribed to the dehydration of the Eu(III) chelate, which is caused by the specific solvation with [C mim][Tf
mim][Tf N] molecules.
N] molecules.
岡村 浩之; 坂江 広基*; 木谷 径治*; 平山 直紀*; 下条 晃司郎; 長縄 弘親; 井村 久則*
no journal, ,
Solvation of a central Eu(III) ion in the tris(2-thenoyltrifluoroacetonato)europium(III) (Eu(tta) ) chelate in 1-alkyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C
) chelate in 1-alkyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N];
N];  = 2-10) was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both spectroscopic studies consistently indicated that Eu(tta)
= 2-10) was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both spectroscopic studies consistently indicated that Eu(tta) extracted or synthesized was almost completely dehydrated in [C
extracted or synthesized was almost completely dehydrated in [C mim][Tf
mim][Tf N] saturated with water. Furthermore, it was found that a small amount of [C
N] saturated with water. Furthermore, it was found that a small amount of [C mim][Tf
mim][Tf N] caused the dehydration of the Eu(III) chelate in 1,2-dichloroethane. The release of the water molecules from the Eu(III) inner coordination sphere of Eu(tta)
N] caused the dehydration of the Eu(III) chelate in 1,2-dichloroethane. The release of the water molecules from the Eu(III) inner coordination sphere of Eu(tta) can be ascribed to the specific solvation of [C
can be ascribed to the specific solvation of [C mim][Tf
mim][Tf N], i.e., coordination of Tf
N], i.e., coordination of Tf N
N molecules.
molecules.