Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 ,
, -dioctyldiglycolamic acid
-dioctyldiglycolamic acid下条 晃司郎; 中井 綾香*; 岡村 浩之; 斎藤 拓巳*; 大橋 朗*; 長縄 弘親
Analytical Sciences, 30(4), p.513 - 517, 2014/04
被引用回数:15 パーセンタイル:48.38(Chemistry, Analytical)We report on the acid dissociation constants (
 ) of diglycolamic acid-type ligands together with comprehensive data on the extraction performance of
) of diglycolamic acid-type ligands together with comprehensive data on the extraction performance of  ,
, -dioctyldiglycolamic acid (DODGAA) for 54 metal ions. The p
-dioctyldiglycolamic acid (DODGAA) for 54 metal ions. The p
 of the diglycolamic acid framework was determined to be 3.54 in water by potentiometric titration, indicating that DODGAA is strongly acidic compared with carboxylic acid. DODGAA can quantitatively transfer various metal ions among the 54 metal ions through a proton-exchange reaction, and provides excellent extraction performance and separation ability for rare-earth metal ions, In(III), Fe(III), Hg(II), and Pb(II) among the 54 metal ions.
of the diglycolamic acid framework was determined to be 3.54 in water by potentiometric titration, indicating that DODGAA is strongly acidic compared with carboxylic acid. DODGAA can quantitatively transfer various metal ions among the 54 metal ions through a proton-exchange reaction, and provides excellent extraction performance and separation ability for rare-earth metal ions, In(III), Fe(III), Hg(II), and Pb(II) among the 54 metal ions.
下条 晃司郎; 青柳 登; 斎藤 拓巳*; 岡村 浩之; 久保田 富生子*; 後藤 雅宏*; 長縄 弘親
Analytical Sciences, 30(2), p.263 - 269, 2014/02
被引用回数:49 パーセンタイル:86.53(Chemistry, Analytical) ,
, -dioctyldiglycolamic acid (DODGAA) enables the quantitative partitioning of all Ln
-dioctyldiglycolamic acid (DODGAA) enables the quantitative partitioning of all Ln ions from moderately acidic solutions, while showing selectivity for heavier lanthanides, and provides remarkably high extraction separation performance for Ln
ions from moderately acidic solutions, while showing selectivity for heavier lanthanides, and provides remarkably high extraction separation performance for Ln compared with typical carboxylic acid extractants. Furthermore, the mutual separation abilities of DODGAA for light lanthanides are higher than those of organophosphorus extractants. Slope analysis, loading tests, and electrospray ionization mass spectrometry measurements demonstrated that the transfer of Ln
compared with typical carboxylic acid extractants. Furthermore, the mutual separation abilities of DODGAA for light lanthanides are higher than those of organophosphorus extractants. Slope analysis, loading tests, and electrospray ionization mass spectrometry measurements demonstrated that the transfer of Ln with DODGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, Ln(DODGAA)
with DODGAA proceeded through a proton-exchange reaction, forming a 1:3 complex, Ln(DODGAA) . The stripping of Ln
. The stripping of Ln from the extracting phase was successfully achieved under acidic conditions. Time-resolved laser-induced fluorescence spectroscopy revealed that the extracted Eu
from the extracting phase was successfully achieved under acidic conditions. Time-resolved laser-induced fluorescence spectroscopy revealed that the extracted Eu ions were completely dehydrated by complexation with DODGAA.
ions were completely dehydrated by complexation with DODGAA.
岡村 浩之; 坂江 広基*; 木谷 径治*; 平山 直紀*; 青柳 登; 斎藤 拓巳*; 下条 晃司郎; 長縄 弘親; 井村 久則*
Polyhedron, 31(1), p.748 - 753, 2012/01
被引用回数:29 パーセンタイル:87.57(Chemistry, Inorganic & Nuclear)The extraction constant and the two-phase stability constant of tris(2-thenoyltrifluoroacetonato) europium(III) between 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]) as an ionic liquid and an aqueous phase were determined by considering the extraction equilibria. Specific solute-solvent interactions between the neutral Eu(III) chelate and [C
N]) as an ionic liquid and an aqueous phase were determined by considering the extraction equilibria. Specific solute-solvent interactions between the neutral Eu(III) chelate and [C mim][Tf
mim][Tf N] molecules were revealed from the relationships between the distribution constant of the enol form of 2-thenoyltrifluoroacetone (Htta) and the distribution constant (
N] molecules were revealed from the relationships between the distribution constant of the enol form of 2-thenoyltrifluoroacetone (Htta) and the distribution constant (
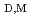 ) of the neutral Eu(III) chelate. The coordination environment of Eu
) of the neutral Eu(III) chelate. The coordination environment of Eu in the neutral Eu(III) chelate in [C
in the neutral Eu(III) chelate in [C mim][Tf
mim][Tf N] was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both methods consistently indicated that not only the Eu(III)chelate extracted but also Eu(tta)
N] was investigated by time-resolved laser-induced fluorescence spectroscopy and infrared absorption spectroscopy. Both methods consistently indicated that not only the Eu(III)chelate extracted but also Eu(tta) (H
(H O)
O) synthesized as a solid crystal were almost completely dehydrated in [C
synthesized as a solid crystal were almost completely dehydrated in [C mim][Tf
mim][Tf N] saturated with water. Consequently, the higher
N] saturated with water. Consequently, the higher 
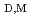 or extractability of the neutral Eu(III) chelate in the [C
or extractability of the neutral Eu(III) chelate in the [C mim][Tf
mim][Tf N] system can be ascribed to the dehydration of the Eu(III) chelate, which is caused by the specific solvation with [C
N] system can be ascribed to the dehydration of the Eu(III) chelate, which is caused by the specific solvation with [C mim][Tf
mim][Tf N] molecules.
N] molecules.
斎藤 親; 佐々木 幸男; 小倉 照雄; 田村 勝裕; 野村 勇; 国谷 安二; 古田 敏城; 吉田 菊夫
JAERI-M 5084, 60 Pages, 1973/01
JPDR-Iは1963年8月22日の初臨界後、1969年9月にJPDR-II改造工事のため長期停止に入った。本報告書は約6年間にわたる自然循環BWRとしての運転が終了したのを機会に、その間に蓄積された貴重な放射線管理データを整理し、検討を加えたものである。
岡村 浩之; 下条 晃司郎; 池田 篤史*; 斎藤 拓巳*; 青柳 登; 平山 直紀*; 梅谷 重夫*; 井村 久則*; 長縄 弘親
no journal, ,
We recently reported that diaza-18-crown-6 bearing two  -diketone fragments (H2
-diketone fragments (H2 DA18C6) provides a considerably high extractability of Sr
DA18C6) provides a considerably high extractability of Sr due to a cooperative intramolecular interaction in the ionic liquid (IL) extraction system. In the present study, we have investigated the extraction capability of H2
due to a cooperative intramolecular interaction in the ionic liquid (IL) extraction system. In the present study, we have investigated the extraction capability of H2 DA18C6 for Sr
DA18C6 for Sr into 1-ethyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C
into 1-ethyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([C mim][Tf
mim][Tf N]) and chloroform in detail. From the p
N]) and chloroform in detail. From the p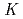
 measurements and the slope analysis, the transfer of Sr
measurements and the slope analysis, the transfer of Sr with H2
with H2 DA18C6 into [C
DA18C6 into [C mim][Tf
mim][Tf N] proceeds via a proton exchange reaction, which is identical to the extraction mechanism in the chloroform system. The superior extraction performance in the IL system is not attributed to the difference in the extraction mechanism between IL and chloroform systems. Extended X-ray absorption fine structure measurements showed that the Sr coordination environment of the Sr
N] proceeds via a proton exchange reaction, which is identical to the extraction mechanism in the chloroform system. The superior extraction performance in the IL system is not attributed to the difference in the extraction mechanism between IL and chloroform systems. Extended X-ray absorption fine structure measurements showed that the Sr coordination environment of the Sr -
- DA18C6 complex in [C
DA18C6 complex in [C mim][Tf
mim][Tf N] is similar to that in chloroform. However, the distance between Sr
N] is similar to that in chloroform. However, the distance between Sr and O atom in the Sr
and O atom in the Sr -
- DA18C6 complex in [C
DA18C6 complex in [C mim][Tf
mim][Tf N] was shorter than that in chloroform. This result reveals that the shortening of the distance causes remarkably high extractability of Sr
N] was shorter than that in chloroform. This result reveals that the shortening of the distance causes remarkably high extractability of Sr , and is ascribable to the cooperative intramolecular interaction of this ligand in IL.
, and is ascribable to the cooperative intramolecular interaction of this ligand in IL.