Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
岸 浩史*; 坂本 友和*; 朝澤 浩一郎*; 山口 進*; 加藤 豪士*; Zulevi, B.*; Serov, A.*; Artyushkova, K.*; Atanassov, P.*; 松村 大樹; et al.
Nanomaterials (Internet), 8(12), p.965_1 - 965_13, 2018/12
被引用回数:11 パーセンタイル:49.2(Chemistry, Multidisciplinary)Platinum group metal-free catalysts based on transition metal-nitrogen-carbon nanomaterials have been studied by a combination of in situ X-ray spectroscopy techniques, high-resolution transmission electron microscope, M ssbauer spectroscopy, electrochemical methods and density functional theory. Fe-N-C oxygen reduction reaction electrocatalysts were synthesized by varying several synthetic parameters to obtain nanomaterials with different composition and morphology. Associated with Fe-N
ssbauer spectroscopy, electrochemical methods and density functional theory. Fe-N-C oxygen reduction reaction electrocatalysts were synthesized by varying several synthetic parameters to obtain nanomaterials with different composition and morphology. Associated with Fe-N motive and the presence of Fe metallic particles in the electrocatalysts showed the clear differences in the variation of composition; processing and treatment conditions of sacrificial support method. From the results of material characterization; catalytic activity and theoretical studies; Fe metallic particles (coated with carbon) are main contributors into the HO
motive and the presence of Fe metallic particles in the electrocatalysts showed the clear differences in the variation of composition; processing and treatment conditions of sacrificial support method. From the results of material characterization; catalytic activity and theoretical studies; Fe metallic particles (coated with carbon) are main contributors into the HO
 generation.
generation.
 O
O /C hydrazine electrooxidation catalysts for anion exchange membrane fuel cells
/C hydrazine electrooxidation catalysts for anion exchange membrane fuel cells坂本 友和*; 増田 晃之*; 吉本 光児*; 岸 浩史*; 山口 進*; 松村 大樹; 田村 和久; 堀 彰宏*; 堀内 洋輔*; Serov, A.*; et al.
Journal of the Electrochemical Society, 164(4), p.F229 - F234, 2017/01
被引用回数:13 パーセンタイル:43.47(Electrochemistry)NiO/ Nb O
O /C (8:1), (4:1), (2:1), NiO/C, and Ni/C catalysts for hydrazine electrooxidation were synthesized by an evaporation drying method followed by thermal annealing. Prepared catalysts were characterized by X-ray diffraction (XRD), high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM), energy dispersive X-ray spectrometry (EDS), and X-ray absorption fine structure (XAFS). The highest catalytic activity in mentioned above reactionwas found for Ni/C, followed by: NiO/Nb
/C (8:1), (4:1), (2:1), NiO/C, and Ni/C catalysts for hydrazine electrooxidation were synthesized by an evaporation drying method followed by thermal annealing. Prepared catalysts were characterized by X-ray diffraction (XRD), high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM), energy dispersive X-ray spectrometry (EDS), and X-ray absorption fine structure (XAFS). The highest catalytic activity in mentioned above reactionwas found for Ni/C, followed by: NiO/Nb O
O /C (8:1), NiO/Nb
/C (8:1), NiO/Nb O
O /C (4:1). NiO/Nb
/C (4:1). NiO/Nb O
O /C (2:1) whiles NiO/C has almost no activity for hydrazine oxidation. It was explained by oxygen defect of NiO in NiO/ Nb
/C (2:1) whiles NiO/C has almost no activity for hydrazine oxidation. It was explained by oxygen defect of NiO in NiO/ Nb O
O /C from XAFS analysis. The selectivity hydrazine electrooxidation as measured by ammonia production resulted in observation that metallic Ni surface facilitates N-N bond breaking of hydrazine, which was confirmed by density functional theory (DFT) calculations.
/C from XAFS analysis. The selectivity hydrazine electrooxidation as measured by ammonia production resulted in observation that metallic Ni surface facilitates N-N bond breaking of hydrazine, which was confirmed by density functional theory (DFT) calculations.
坂本 友和*; 岸 浩史*; 山口 進*; 松村 大樹; 田村 和久; 堀 彰宏*; 堀内 洋輔*; Serov, A.*; Artyushkova, K.*; Atanassov, P.*; et al.
Journal of the Electrochemical Society, 163(10), p.H951 - H957, 2016/08
被引用回数:30 パーセンタイル:76.46(Electrochemistry)The catalytic process takes place on nickel oxide surface of a Ni oxide nano-particle decorated carbon support (NiO/C). In-situ X-ray absorption fine structure (XAFS) spectroscopy was used to investigate the reaction mechanism for hydrazine electrooxidation on NiO surface. The spectra of X-ray absorption near-edge structure (XANES) of Ni K-edge indicated that adsorption of OH on Ni site during the hydrazine electrooxidation reaction. Density functional theory (DFT) calculations were used to elucidate and suggest the mechanism of the electrooxidation and specifically propose the localization of electron density from OH
on Ni site during the hydrazine electrooxidation reaction. Density functional theory (DFT) calculations were used to elucidate and suggest the mechanism of the electrooxidation and specifically propose the localization of electron density from OH to 3d orbital of Ni in NiO. It is found that the accessibility of Ni atomic sites in NiO structure is critical for hydrazine electrooxidation. Based on this study, we propose a possible reaction mechanism for selective hydrazine electrooxidation to water and nitrogen taking place on NiO surface as it is applicable to direct hydrazine alkaline membrane fuel cells.
to 3d orbital of Ni in NiO. It is found that the accessibility of Ni atomic sites in NiO structure is critical for hydrazine electrooxidation. Based on this study, we propose a possible reaction mechanism for selective hydrazine electrooxidation to water and nitrogen taking place on NiO surface as it is applicable to direct hydrazine alkaline membrane fuel cells.
坂本 友和*; 松村 大樹; 朝澤 浩一郎*; Martinez, U.*; Serov, A.*; Artyushkova, K.*; Atanassov, P.*; 田村 和久; 西畑 保雄; 田中 裕久*
Electrochimica Acta, 163, p.116 - 122, 2015/05
被引用回数:57 パーセンタイル:82.8(Electrochemistry)Carbon supported Ni, Ni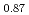 Zn
Zn , and Co hydrazine electrooxidation catalysts were synthesized by an impregnation/freeze-drying procedure followed by thermal annealing for use as anode catalyst of direct hydrazine hydrate fuel cells (DHFCs). The cell performance of DHFCs changed significantly when different catalysts were used as anode. Ammonia generation from anode outlet at open circuit voltage (OCV) condition was higher for Co/C than for Ni-based catalysts. To better understand the cause of different performance and selectivity of each anode catalyst, extensive ex-situ and operando characterization was carried out. Operando XAFS measurement of Ni-K and Co-
, and Co hydrazine electrooxidation catalysts were synthesized by an impregnation/freeze-drying procedure followed by thermal annealing for use as anode catalyst of direct hydrazine hydrate fuel cells (DHFCs). The cell performance of DHFCs changed significantly when different catalysts were used as anode. Ammonia generation from anode outlet at open circuit voltage (OCV) condition was higher for Co/C than for Ni-based catalysts. To better understand the cause of different performance and selectivity of each anode catalyst, extensive ex-situ and operando characterization was carried out. Operando XAFS measurement of Ni-K and Co- edge shows the potential dependence of atomic structure of Ni/C, Ni
edge shows the potential dependence of atomic structure of Ni/C, Ni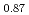 Zn
Zn /C, and Co/C during hydrazine electrooxidation reaction.
/C, and Co/C during hydrazine electrooxidation reaction.
朝澤 浩一郎*; 岸 浩史*; 田中 裕久*; 松村 大樹; 田村 和久; 西畑 保雄; Saputro, A. G.*; 中西 寛*; 笠井 秀明*; Artyushkova, K.*; et al.
Journal of Physical Chemistry C, 118(44), p.25480 - 25486, 2014/11
被引用回数:15 パーセンタイル:44.91(Chemistry, Physical)Non-noble metal electrocatalysts not only are a solution to limited resources but also achieve higher efficiency for fuel cells, especially in alkaline media such as alkaline membrane fuel cells. Co-polypyrrole-based electrocatalysts provide high oxygen reduction reaction (ORR) reactivity, but the active sites and reaction mechanism have yet to be elucidated fully. In this study, ex situ and in situ synchrotron characterization and theoretical study have been combined to evaluate the ORR mechanism on two possible active sites consisting of Co coordinated with pyrrolic nitrogen and Co coordinated with pyridinic nitrogen.
坂本 友和*; 朝澤 浩一郎*; Martinez, U.*; Halevi, B.*; 鈴木 敏行*; 荒井 重勇*; 松村 大樹; 西畑 保雄; Atanassov, P.*; 田中 裕久*
Journal of Power Sources, 234, p.252 - 259, 2013/07
被引用回数:68 パーセンタイル:87.04(Chemistry, Physical)Carbon supported Ni, La, and Ni La
La (0.1
(0.1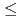
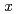
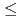 0.9) catalysts were synthesized by an impregnation/freeze-drying procedure. The catalytic activity for electro-oxidation of hydrazine hydrate was evaluated using a 16-channel electrochemical electrode array. The Ni
0.9) catalysts were synthesized by an impregnation/freeze-drying procedure. The catalytic activity for electro-oxidation of hydrazine hydrate was evaluated using a 16-channel electrochemical electrode array. The Ni La
La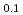 /C catalyst oxidized hydrazine hydrate at a lower potential and exhibited higher mass activity in comparison with a similarly made Ni/C catalyst. Chemical insight suggests that the cause of improved performance for the Ni
/C catalyst oxidized hydrazine hydrate at a lower potential and exhibited higher mass activity in comparison with a similarly made Ni/C catalyst. Chemical insight suggests that the cause of improved performance for the Ni La
La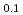 /C catalyst is likely multifunctional synergism of the components. However, X-ray absorption fine structure (XAFS) and high voltage electron microscopy (HVEM) unexpectedly show some hcp LaNi
/C catalyst is likely multifunctional synergism of the components. However, X-ray absorption fine structure (XAFS) and high voltage electron microscopy (HVEM) unexpectedly show some hcp LaNi shells coating the fcc Ni catalyst particles. As a result of the screening tests, an unsupported Ni
shells coating the fcc Ni catalyst particles. As a result of the screening tests, an unsupported Ni La
La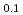 catalyst was synthesized by spray pyrolysis and tested in a direct hydrazine hydrate fuel cell MEA (DHFC) producing 453 mW cm
catalyst was synthesized by spray pyrolysis and tested in a direct hydrazine hydrate fuel cell MEA (DHFC) producing 453 mW cm .
.
朝澤 浩一郎*; 山本 和矢*; 山田 浩次*; 田中 裕久*; 松村 大樹; 田村 和久; 西畑 保雄; Atanassov, P.*
ECS Transactions, 33(1), p.1751 - 1755, 2010/10
Coポリピロールカーボン(PPyC)をX線吸収分光法により構造解析を行った。この試料は酸処理により酸素還元活性が高くなる。酸処理前の近接構造は2つのピークからなり、1つはCo-O/Nからなる軽元素の配位であり、もう1つは水酸化コバルトに起因するピークであった。後者のピークのみが酸処理により消滅した。触媒活性の結果と合わせて、熱処理を施さずとも、活性に寄与すると推測されるCo-Nの配位が存在することがわかった。