Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

McGrady, J.; 熊谷 友多; 渡邉 雅之; 桐島 陽*; 秋山 大輔*; 紀室 辰伍; 石寺 孝充
Journal of Nuclear Science and Technology, 60(12), p.1586 - 1594, 2023/12
被引用回数:0 パーセンタイル:0.01(Nuclear Science & Technology)Raman spectroscopy was used to investigate the effect of bicarbonate solution on the hyperstoichiometric UO surface oxide of UO
surface oxide of UO pellets. The hyperstoichiometry causes distortion of the UO
pellets. The hyperstoichiometry causes distortion of the UO lattice, leading to the formation of defect peaks which arise in the Raman spectrum due to a loss of symmetry. By deconvolution of the defect peaks, the extent of oxygen inclusion and defect formation in the surface oxide as a function of bicarbonate concentration was investigated. Immersion in solution caused a reduction in the amount of interstitial oxygen due to dissolution U(V) and U(VI) in the UO
lattice, leading to the formation of defect peaks which arise in the Raman spectrum due to a loss of symmetry. By deconvolution of the defect peaks, the extent of oxygen inclusion and defect formation in the surface oxide as a function of bicarbonate concentration was investigated. Immersion in solution caused a reduction in the amount of interstitial oxygen due to dissolution U(V) and U(VI) in the UO oxide surface. The defect concentration increased upon immersion due to the formation of an equilibrium between dissolution and reprecipitation. The bicarbonate concentration affected the defect content which was attributed to different forms of U in solution (hydrolysed UO
oxide surface. The defect concentration increased upon immersion due to the formation of an equilibrium between dissolution and reprecipitation. The bicarbonate concentration affected the defect content which was attributed to different forms of U in solution (hydrolysed UO
 and UO
and UO (CO
(CO )
)
 ) leading to different rates of reduction and precipitation.
) leading to different rates of reduction and precipitation.
 O
O decomposition at the U
decomposition at the U O
O surface in bicarbonate solution
surface in bicarbonate solutionMcGrady, J.; 熊谷 友多; 渡邉 雅之; 桐島 陽*; 秋山 大輔*; 北村 暁; 紀室 辰伍
RSC Advances (Internet), 11(46), p.28940 - 28948, 2021/08
被引用回数:5 パーセンタイル:38.19(Chemistry, Multidisciplinary)The rate of U release is affected by bicarbonate (HCO
 ) concentrations in the groundwater, as well as H
) concentrations in the groundwater, as well as H O
O produced by water radiolysis. To understand the dissolution of U
produced by water radiolysis. To understand the dissolution of U O
O by H
by H O
O in bicarbonate solution (0.1 - 50 mM), dissolved U concentrations were measured upon H
in bicarbonate solution (0.1 - 50 mM), dissolved U concentrations were measured upon H O
O addition (300
addition (300  M) to U
M) to U O
O /bicarbonate mixtures. As the H
/bicarbonate mixtures. As the H O
O decomposition mechanism is integral to U dissolution, the kinetics and mechanism of H
decomposition mechanism is integral to U dissolution, the kinetics and mechanism of H O
O decomposition at the U
decomposition at the U O
O surface was investigated. The dissolution of U increased with bicarbonate concentration which was attributed to a change in the H
surface was investigated. The dissolution of U increased with bicarbonate concentration which was attributed to a change in the H O
O decomposition mechanism from catalytic at low bicarbonate (
decomposition mechanism from catalytic at low bicarbonate (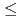 5 mM HCO
5 mM HCO
 ) to oxidative at high bicarbonate (
) to oxidative at high bicarbonate ( 10 mM HCO
10 mM HCO
 ). Catalytic H
). Catalytic H O
O at low bicarbonate was attributed to the formation of an oxidised surface layer.
at low bicarbonate was attributed to the formation of an oxidised surface layer.
McGrady, J.; 熊谷 友多; 日下 良二
no journal, ,
Two new pathways to the formation of amorphous uranyl peroxide under ambient conditions have been identified in this study. UO powder was immersed in H
powder was immersed in H O
O solution, and in-situ Raman spectroscopy was used to analyse the surface oxide. At the interface uranyl peroxide was identified, yet underneath this surface amorphous uranyl peroxide was generated. The amorphous phase was found to be transient over time. Drying of the sample after immersion showed an enhanced Raman peak signal for the amorphous phase indicating significant generation, suggesting the formation mechanism involves dehydration. Addition of the amorphous uranyl peroxide into water showed decomposition of the amorphous phase by disappearance of the Raman peak, and the release of gas was observed via the Raman optical microscope which was thought to be O
solution, and in-situ Raman spectroscopy was used to analyse the surface oxide. At the interface uranyl peroxide was identified, yet underneath this surface amorphous uranyl peroxide was generated. The amorphous phase was found to be transient over time. Drying of the sample after immersion showed an enhanced Raman peak signal for the amorphous phase indicating significant generation, suggesting the formation mechanism involves dehydration. Addition of the amorphous uranyl peroxide into water showed decomposition of the amorphous phase by disappearance of the Raman peak, and the release of gas was observed via the Raman optical microscope which was thought to be O .
.