Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
根本 文也*; 古府 麻衣子; 長尾 道弘*; 大石 一城*; 高田 慎一; 鈴木 淳市*; 山田 武*; 柴田 薫; 上木 岳士*; 北沢 侑造*; et al.
Journal of Chemical Physics, 149(5), p.054502_1 - 054502_11, 2018/08
被引用回数:19 パーセンタイル:70.02(Chemistry, Physical)Alkyl-methyl-imidazolium ionic liquids C mimX (
mimX ( : alkyl-carbon number, X: anion) have short-range layer structures consisting of ionic and neutral (alkylchain) domains. To investigate the temperature dependences of the interlayer, interionic group, and inter-alkylchain correlations, we have measured the neutron diffraction (ND) of C
: alkyl-carbon number, X: anion) have short-range layer structures consisting of ionic and neutral (alkylchain) domains. To investigate the temperature dependences of the interlayer, interionic group, and inter-alkylchain correlations, we have measured the neutron diffraction (ND) of C mimPF
mimPF (
( = 16, 9.5, 8). The quasielastic neutron scattering (QENS) of C16mimPF
= 16, 9.5, 8). The quasielastic neutron scattering (QENS) of C16mimPF was also measured to study the dynamics of each correlation. C16mimPF
was also measured to study the dynamics of each correlation. C16mimPF shows a first-order transition between the liquid (L) and liquid crystalline (LC) phases at
shows a first-order transition between the liquid (L) and liquid crystalline (LC) phases at 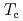 = 394 K. C8mimPF
= 394 K. C8mimPF exhibits a glass transition at
exhibits a glass transition at 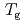 = 200 K. C9.5mimPF
= 200 K. C9.5mimPF has both transitions at
has both transitions at 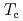 = 225 K and
= 225 K and 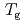 = 203 K. In the ND experiments, all samples exhibit three peaks corresponding to the correlations mentioned above. The widths of the interlayer peak at ca. 0.2
= 203 K. In the ND experiments, all samples exhibit three peaks corresponding to the correlations mentioned above. The widths of the interlayer peak at ca. 0.2 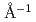 changed drastically at the L-LC transitions, while the interionic peaks at ca. 1
changed drastically at the L-LC transitions, while the interionic peaks at ca. 1 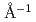 exhibited a small jump at
exhibited a small jump at 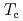 . The peak position and area of the three peaks did not change much at the transition. The structural changes were minimal at
. The peak position and area of the three peaks did not change much at the transition. The structural changes were minimal at 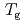 . The QENS experiments demonstrated that the relaxation time of the interlayer motion increased tenfold at
. The QENS experiments demonstrated that the relaxation time of the interlayer motion increased tenfold at 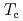 , while those of other motions were monotonous in the whole temperature region. The structural and dynamical changes are characteristic of the L-LC transition in imidazolium-based ionic liquids.
, while those of other motions were monotonous in the whole temperature region. The structural and dynamical changes are characteristic of the L-LC transition in imidazolium-based ionic liquids.
高橋 憲司*; 酒井 慎吾*; 手塚 博昭*; 比江嶋 祐介*; 勝村 庸介; 渡邉 正義*
Journal of Physical Chemistry B, 111(18), p.4807 - 4811, 2007/05
被引用回数:41 パーセンタイル:68.99(Chemistry, Physical)Photodetachment of electrons from iodide ions produced diiodide anion radicals in ionic liquids containing ammonium, pyrrolidinium, and piperidinium cations. The rates of reaction between diiodide anion radicals in molecular solvents such as H O, methanol, and ethanol could be estimated by the Debye-Smoluchowski equation, which accounts for electrostatic interactions using dielectric constants for the molecular solvents. In contrast, the rates of reaction between diiodide anion radicals in the ionic liquids were close to the diffusion limited rates for the neutral molecules, suggesting that electrostatic repulsion between the diiodide anion radicals is weakened by Coulombic shielding in the ionic liquids.
O, methanol, and ethanol could be estimated by the Debye-Smoluchowski equation, which accounts for electrostatic interactions using dielectric constants for the molecular solvents. In contrast, the rates of reaction between diiodide anion radicals in the ionic liquids were close to the diffusion limited rates for the neutral molecules, suggesting that electrostatic repulsion between the diiodide anion radicals is weakened by Coulombic shielding in the ionic liquids.