Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Chiera, N. M.*; Sato, Tetsuya; Eichler, R.*; Tomitsuka, Tomohiro; Asai, Masato; Adachi, Sadia*; Dressler, R.*; Hirose, Kentaro; Inoue, Hiroki*; Ito, Yuta; et al.
Angewandte Chemie; International Edition, 60(33), p.17871 - 17874, 2021/08
Times Cited Count:5 Percentile:25.61(Chemistry, Multidisciplinary)The formation and the chemical characterization of single atoms of dubnium (Db, element 105), in the form of its volatile oxychloride, was investigated using the on-line gas phase chromatography technique, in the temperature range 350 - 600  C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl
C. Under the exact same chemical conditions, comparative studies with the lighter homologs of group-5 in the Periodic Table clearly indicate the volatility sequence being NbOCl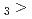 TaOCl
TaOCl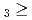 DbOCl
DbOCl . From the obtained experimental results, thermochemical data for DbOCl
. From the obtained experimental results, thermochemical data for DbOCl were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
were derived. The present study delivers reliable experimental information for theoretical calculations on the chemical properties of transactinides.
Ozama, Eiki*; Adachi, Sadia*; Takayanagi, Toshiyuki*; Shiga, Motoyuki
Chemistry; A European Journal, 24(48), p.12716 - 12721, 2018/08
Times Cited Count:5 Percentile:17.57(Chemistry, Multidisciplinary)The structures of trivalent actinium cation in helium clusters (Ac He
He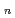 ) have been studied by quantum path integral molecular dynamics simulations with different cluster sizes,
) have been studied by quantum path integral molecular dynamics simulations with different cluster sizes,  = 18-200. The nuclear quantum effect of helium atoms plays an important role in the vibrational amplitude of the Ac
= 18-200. The nuclear quantum effect of helium atoms plays an important role in the vibrational amplitude of the Ac -He complex at low temperatures (1-3 K) where the complex is stable. We found that the coordination number of helium atoms comprising the first solvation shell can be as high as eighteen. In this case, the helium atoms are arranged in D
-He complex at low temperatures (1-3 K) where the complex is stable. We found that the coordination number of helium atoms comprising the first solvation shell can be as high as eighteen. In this case, the helium atoms are arranged in D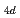 symmetry. The Ac
symmetry. The Ac -He
-He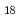 complex becomes more rigid as the cluster increases in sizes, implying that it becomes more stable. The simulation results are based on an accurate description of the Ac
complex becomes more rigid as the cluster increases in sizes, implying that it becomes more stable. The simulation results are based on an accurate description of the Ac -He interaction using relativistic ab initio calculations.
-He interaction using relativistic ab initio calculations.
 solution
solutionKato, Mizuho*; Adachi, Sadia*; Toyoshima, Atsushi*; Tsukada, Kazuaki; Asai, Masato; Haba, Hiromitsu*; Yokokita, Takuya*; Komori, Yukiko*; Shigekawa, Yudai*; Sueki, Keisuke*
no journal, ,
A few studies have been so far performed on aqueous chemistry of Db, the group-5 superheavy element, by anion-exchange experiments in HF and HF/HNO mixture solutions. Although the distribution coefficient (Kd) of Db was shown to trend to Ta
mixture solutions. Although the distribution coefficient (Kd) of Db was shown to trend to Ta  Nb
Nb  Db
Db  Pa in the group-5 homologs and pseudo-homolog elements, its chemical species has not been clarified. In this work, for identification of fluoride species of Db, we performed anion-exchange experiments of Nb, Ta, and Db in HF/1.0 M HNO
Pa in the group-5 homologs and pseudo-homolog elements, its chemical species has not been clarified. In this work, for identification of fluoride species of Db, we performed anion-exchange experiments of Nb, Ta, and Db in HF/1.0 M HNO mixture solutions by using an automated rapid chemistry apparatus, ARCA.
mixture solutions by using an automated rapid chemistry apparatus, ARCA.
 solution for understanding chemical species of Db
solution for understanding chemical species of DbAdachi, Sadia*; Sueki, Keisuke*; Toyoshima, Atsushi*; Tsukada, Kazuaki; Haba, Hiromitsu*; Komori, Yukiko*; Yokokita, Takuya*; Mori, Taiki*
no journal, ,
Anion exchange behavior of group-5 elements, Nb, Ta, and their pseudo homologue Pa in HF and HF/HNO solution was investigated to understand chemical properties of Db. Adsorption behavior of Db is clearly different from that of Ta and is similar to those of Nb and Pa. In the present study, anion exchange experiments of Nb, Ta and Pa were carried out with HF/HNO
solution was investigated to understand chemical properties of Db. Adsorption behavior of Db is clearly different from that of Ta and is similar to those of Nb and Pa. In the present study, anion exchange experiments of Nb, Ta and Pa were carried out with HF/HNO aqueous solution as basic experiments for estimating the species of Db fluoride complex. Adsorption behavior was investigated by changing the fluoride ion concentration for each HNO
aqueous solution as basic experiments for estimating the species of Db fluoride complex. Adsorption behavior was investigated by changing the fluoride ion concentration for each HNO concentration. Based on the observed distribution coefficients of these elements, we discuss the formation reaction of the anionic fluoride complex and chemical species of Db.
concentration. Based on the observed distribution coefficients of these elements, we discuss the formation reaction of the anionic fluoride complex and chemical species of Db.
Sato, Tetsuya; Chiera, N. M.*; Tomitsuka, Tomohiro; Tokoi, Katsuyuki*; Suzuki, Hayato*; Ito, Yuta; Asai, Masato; Shirai, Kaori*; Inoue, Hiroki*; Adachi, Sadia*; et al.
no journal, ,
The influence of strong relativistic effects on chemical properties has been interesting in the superheavy element region. Their chemical properties, however, have not been investigated sufficiently because of experimental difficulties owing to their low production rates and short half-lives. In order to elucidate the chemical properties of dubnium (Db, Z = 105), we have conducted on-line isothermal gas chromatographic experiments of oxychloride of group-5 elements. We confirmed the formation of volatile oxychlorides of Db and its lighter homologs Nb and Ta by using  Db (half-life,
Db (half-life, 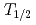 = 33.8 s),
= 33.8 s),  Nb (
Nb (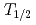 = 14.5 min.), and
= 14.5 min.), and 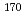 Ta (
Ta (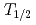 = 6.76 min.), respectively. We successfully determined the adsorption enthalpies of the oxychlorides of each element on the quartz surface from their isothermal gas chromatographic behavior. The obtained volatility sequence of the group-5 elements is found to be Nb
= 6.76 min.), respectively. We successfully determined the adsorption enthalpies of the oxychlorides of each element on the quartz surface from their isothermal gas chromatographic behavior. The obtained volatility sequence of the group-5 elements is found to be Nb  Ta
Ta  Db.
Db.