Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 condensed state in
condensed state in 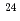 Mg using the
Mg using the  C+
C+ C scattering
C scatteringFujikawa, Y.*; Kawabata, T.*; Adachi, S.*; Hirose, Kentaro; Makii, Hiroyuki; Nishio, Katsuhisa; Orlandi, R.; Suzaki, Fumi; 13 of others*
Physics Letters B, 848, p.133834_1 - 133834_6, 2024/01
Times Cited Count:0 Percentile:0.02(Astronomy & Astrophysics)
Tamii, Atsushi*; Pellegri, L.*; S derstr
derstr m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
m, P.-A.*; Allard, D.*; Goriely, S.*; Inakura, Tsunenori*; Khan, E.*; Kido, Eiji*; Kimura, Masaaki*; Litvinova, E.*; et al.
European Physical Journal A, 59(9), p.208_1 - 208_21, 2023/09
Times Cited Count:1 Percentile:0.02(Physics, Nuclear)no abstracts in English
 clusters in dilute neutron-rich matter
clusters in dilute neutron-rich matterTanaka, Junki*; Yang, Z.*; Typel, S.*; Adachi, Satoshi*; Bai, S.*; van Beek, P.*; Beaumel, D.*; Fujikawa, Yuki*; Han, J.*; Heil, S.*; et al.
Science, 371(6526), p.260 - 264, 2021/01
Times Cited Count:48 Percentile:99.12(Multidisciplinary Sciences)By employing quasi-free  -cluster-knockout reactions, we obtained direct experimental evidence for the formation of
-cluster-knockout reactions, we obtained direct experimental evidence for the formation of  clusters at the surface of neutron-rich tin isotopes. The observed monotonous decrease of the reaction cross sections with increasing mass number, in excellent agreement with the theoretical prediction, implies a tight interplay between
clusters at the surface of neutron-rich tin isotopes. The observed monotonous decrease of the reaction cross sections with increasing mass number, in excellent agreement with the theoretical prediction, implies a tight interplay between  -cluster formation and the neutron skin.
-cluster formation and the neutron skin.
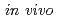 dosimetry system based on an optical fiber-coupled microsized photostimulable phosphor for stereotactic body radiation therapy
dosimetry system based on an optical fiber-coupled microsized photostimulable phosphor for stereotactic body radiation therapyYada, Ryuichi*; Maenaka, Kazusuke*; Miyamoto, Shuji*; Okada, Go*; Sasakura, Aki*; Ashida, Motoi*; Adachi, Masashi*; Sato, Tatsuhiko; Wang, T.*; Akasaka, Hiroaki*; et al.
Medical Physics, 47(10), p.5235 - 5249, 2020/10
Times Cited Count:7 Percentile:52.3(Radiology, Nuclear Medicine & Medical Imaging)The 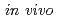 dosimeter system is capable of real-time, accurate, and precise measurement under stereotactic body radiation therapy (SBRT) conditions. The probe is smaller than a conventional dosimeter, has excellent spatial resolution, and can be valuable in SBRT with a steep dose distribution over a small field. The developed PSP dosimeter system appears to be suitable for in vivo SBRT dosimetry.
dosimeter system is capable of real-time, accurate, and precise measurement under stereotactic body radiation therapy (SBRT) conditions. The probe is smaller than a conventional dosimeter, has excellent spatial resolution, and can be valuable in SBRT with a steep dose distribution over a small field. The developed PSP dosimeter system appears to be suitable for in vivo SBRT dosimetry.
 O(
O( He,
He,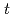 )
) F reaction; Identification of the low-energy "super" -Gamow-Teller state
F reaction; Identification of the low-energy "super" -Gamow-Teller stateFujita, Hirohiko*; Fujita, Yoshitaka*; Utsuno, Yutaka; Yoshida, Kenichi*; Adachi, Tatsuya*; Algora, A.*; Csatl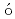 s, M.*; Deaven, J. M.*; Estevez-Aguado, E.*; Guess, C. J.*; et al.
s, M.*; Deaven, J. M.*; Estevez-Aguado, E.*; Guess, C. J.*; et al.
Physical Review C, 100(3), p.034618_1 - 034618_13, 2019/09
Times Cited Count:12 Percentile:77.09(Physics, Nuclear)no abstracts in English
Gepr gs, S.*; Kehlberger, A.*; Coletta, F.*; Qiu, Z.*; Guo, E.-J.*; Schulz, T.*; Mix, C.*; Meyer, S.*; Kamra, A.*; Althammer, M.*; et al.
gs, S.*; Kehlberger, A.*; Coletta, F.*; Qiu, Z.*; Guo, E.-J.*; Schulz, T.*; Mix, C.*; Meyer, S.*; Kamra, A.*; Althammer, M.*; et al.
Nature Communications (Internet), 7, p.10452_1 - 10452_6, 2016/02
Times Cited Count:152 Percentile:97.33(Multidisciplinary Sciences)Hiromoto, Takeshi; Adachi, Motoyasu; Shibazaki, Chie; Schrader, T. E.*; Ostermann, A.*; Kuroki, Ryota
JPS Conference Proceedings (Internet), 8, p.033003_1 - 033003_6, 2015/09
T4 phage lysozyme (T4L) is an endoacetylmuramidase that degrades the murein of the bacterial cell wall by cleaving the  -1,4-glycosidic bond between
-1,4-glycosidic bond between  -acetylmuramic acid and
-acetylmuramic acid and  -acetylglucosamine. We previously reported that the substitution of the catalytic Thr26 with the nucleophilic His converts the wild-type (WT) T4L from an inverting to a retaining glycosidase, in which the
-acetylglucosamine. We previously reported that the substitution of the catalytic Thr26 with the nucleophilic His converts the wild-type (WT) T4L from an inverting to a retaining glycosidase, in which the  -configuration of the substrate is retained in the product. We also found that the Thr26His (T26H) mutant can catalyze a transglycosylation reaction more effectively than hydrolysis, although the WT-T4L has no transglycosidase activity. To clarify the role of the substituted His26 in transglycosylation and to investigate its relationship to the neighboring acidic residue Asp20 using neutron crystallography, a perdeuterated recombinant protein of the T26H mutant (d-T26H) was prepared for crystallization. The perdeuterated form was produced in
-configuration of the substrate is retained in the product. We also found that the Thr26His (T26H) mutant can catalyze a transglycosylation reaction more effectively than hydrolysis, although the WT-T4L has no transglycosidase activity. To clarify the role of the substituted His26 in transglycosylation and to investigate its relationship to the neighboring acidic residue Asp20 using neutron crystallography, a perdeuterated recombinant protein of the T26H mutant (d-T26H) was prepared for crystallization. The perdeuterated form was produced in 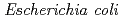 cells cultured in deuterated-rich media. After purification, the d-T26H mutant was crystallized under deuterated conditions and grown to a volume of 0.12 mm
cells cultured in deuterated-rich media. After purification, the d-T26H mutant was crystallized under deuterated conditions and grown to a volume of 0.12 mm using a macroseeding technique. A preliminary neutron-diffraction experiment at 100 K at the FRM II research reactor (Munich, Germany) gave diffraction spots of up to 2.8
using a macroseeding technique. A preliminary neutron-diffraction experiment at 100 K at the FRM II research reactor (Munich, Germany) gave diffraction spots of up to 2.8  resolution after a 1.5 h exposure.
resolution after a 1.5 h exposure.
Ohgama, Kazuya; Ando, Yoko; Yamaguchi, Mika; Ikuta, Yuko; Shinohara, Nobuo; Murakami, Hiroyuki; Yamashita, Kiyonobu; Uesaka, Mitsuru*; Demachi, Kazuyuki*; Komiyama, Ryoichi*; et al.
JAEA-Review 2013-004, 76 Pages, 2013/05
JAEA together with the Japan Nuclear Human Resource Development Network (JN-HRD Net), the University of Tokyo (UT) and the Japan Atomic Industrial Forum (JAIF) cohosted the IAEA-Nuclear Energy Management School in Tokai Village, aiming that Japan will be the center of nuclear HRD in the Asian region. In the school, not only lectures by IAEA experts, but also lectures by Japanese experts and technical visits were included for foreign participants. The school contributed to the internationalization of Japanese young professionals, development of nuclear human resource of other countries, and enhancement of cooperation between IAEA and Japan. Additionally, collaborative relationship within JN-HRD Net was strengthened by the school. In this report, findings obtained during the preparatory work and the school period are reported for future international nuclear HRD activities in Japan.
 cuprates
cupratesWakimoto, Shuichi; Ishii, Kenji; Kimura, Hiroyuki*; Ikeuchi, Kazuhiko*; Yoshida, Masahiro*; Adachi, Tadashi*; Casa, D.*; Fujita, Masaki*; Fukunaga, Yasushi*; Gog, T.*; et al.
Physical Review B, 87(10), p.104511_1 - 104511_7, 2013/03
Times Cited Count:10 Percentile:43.41(Materials Science, Multidisciplinary)Hidaka, Koshi*; Kimura, Toru*; Abdel-Rahman, H. M.*; Nguyen, J.-T.*; McDaniel, K. F.*; Kohlbrenner, W. E.*; Molla, A.*; Adachi, Motoyasu; Tamada, Taro; Kuroki, Ryota; et al.
Journal of Medicinal Chemistry, 52(23), p.7604 - 7617, 2009/07
Times Cited Count:19 Percentile:44.7(Chemistry, Medicinal)A series of HIV protease inhibitor based on the allophenylnorstatine structure with various P2' moieties were synthesized. Among these analogues, we discovered that a small allyl group would maintain potent enzyme inhibitory activity compared to that of the 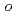 -methylbenzyl moiety in clinical candidate 1 (KNI-764, also known as JE-2147, AG-1776 or SM-319777). Introduction of an anilinic amino group to 2 (KNI-727) improved water-solubility and anti-HIV-1 activity. X-ray crystallographic analysis of 13k (KNI-1689) with a
-methylbenzyl moiety in clinical candidate 1 (KNI-764, also known as JE-2147, AG-1776 or SM-319777). Introduction of an anilinic amino group to 2 (KNI-727) improved water-solubility and anti-HIV-1 activity. X-ray crystallographic analysis of 13k (KNI-1689) with a  -methallyl group at P2' position revealed hydrophobic interactions with Ala28, Ile84, and Ile50' similar to that of 1. The presence of an additional methyl group on the allyl group in compound 13k significantly increased anti-HIV activity over 1, while providing a rational drug design for structural minimization and improving membrane permeability.
-methallyl group at P2' position revealed hydrophobic interactions with Ala28, Ile84, and Ile50' similar to that of 1. The presence of an additional methyl group on the allyl group in compound 13k significantly increased anti-HIV activity over 1, while providing a rational drug design for structural minimization and improving membrane permeability.
Takamura, Shuichi*; Kado, Shinichiro*; Fujii, Takashi*; Fujiyama, Hiroshi*; Takabe, Hideaki*; Adachi, Kazuo*; Morimiya, Osamu*; Fujimori, Naoji*; Watanabe, Takayuki*; Hayashi, Yasuaki*; et al.
Kara Zukai, Purazuma Enerugi No Subete, P. 164, 2007/03
no abstracts in English
 Cu
CuHara, Keigo*; Adachi, Takeshi*; Akimune, Hidetoshi*; Daito, Izuru*; Fujimura, Hisako*; Fujita, Yoshitaka*; Fujiwara, Mamoru; Fushimi, Kenichi*; Hara, Kaoru*; Harakeh, M. N.*; et al.
Physical Review C, 68(6), p.064612_1 - 064612_9, 2003/12
Times Cited Count:11 Percentile:58.08(Physics, Nuclear)no abstracts in English
Oku, Takayuki*; *; *; Yamagata, Yutaka*; Omori, Hitoshi*; Adachi, Tomohiro*; Shimizu, Hirohiko*; *; Hirota, T.*; Iwasa, Hirokatsu*; et al.
JAERI-Conf 2001-002, p.537 - 541, 2001/03
no abstracts in English
Nishio, Satoshi; Ueda, Shuzo; Kurihara, Ryoichi; Kuroda, Toshimasa*; Miura, H.*; Sako, Kiyoshi*; Takase, Kazuyuki; Seki, Yasushi; Adachi, Junichi*; Yamazaki, Seiichiro*; et al.
Fusion Engineering and Design, 48(3-4), p.271 - 279, 2000/09
Times Cited Count:17 Percentile:72(Nuclear Science & Technology)no abstracts in English
Aoyama, S.*; Ishii, T.*; Horita, M.*; Adachi, T.*; Horie, Y.*; Hokari, T.*
PNC TJ1060 92-001, 240 Pages, 1992/03
None
*; Iida, Hiromasa; *; Adachi, Junichi*; ; Ebisawa, Katsuyuki*; *; Fukaya, Kiyoshi; *; *; et al.
JAERI-M 87-219, 336 Pages, 1988/01
no abstracts in English