Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Massey, D.*; Williams, C. D.*; Mu, J.*; Masters, A. J.*; Motokawa, Ryuhei; Aoyagi, Noboru; Ueda, Yuki; Antonio, M. R.*
Journal of Physical Chemistry B, 127(9), p.2052 - 2065, 2023/03
Times Cited Count:0 Percentile:0(Chemistry, Physical)Narita, Hirokazu*; Nicolson, R. M.*; Motokawa, Ryuhei; Ito, Fumiyuki*; Morisaku, Kazuko*; Goto, Midori*; Tanaka, Mikiya*; Heller, W. T.*; Shiwaku, Hideaki; Yaita, Tsuyoshi; et al.
Inorganic Chemistry, 58(13), p.8720 - 8734, 2019/07
Times Cited Count:14 Percentile:69.69(Chemistry, Inorganic & Nuclear)Motokawa, Ryuhei; Kobayashi, Toru; Endo, Hitoshi; Mu, J.*; Williams, C. D.*; Masters, A. J.*; Antonio, M. R.*; Heller, W. T.*; Nagao, Michihiro*
ACS Central Science, 5(1), p.85 - 96, 2019/01
Times Cited Count:46 Percentile:86.05(Chemistry, Multidisciplinary) -butyl phosphate/
-butyl phosphate/ -octane mixtures by X-ray and neutron scattering in a wide
-octane mixtures by X-ray and neutron scattering in a wide 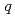 range
rangeMotokawa, Ryuhei; Suzuki, Shinichi; Ogawa, Hiroki*; Antonio, M. R.*; Yaita, Tsuyoshi
Journal of Physical Chemistry B, 116(4), p.1319 - 1327, 2012/02
Times Cited Count:36 Percentile:65.56(Chemistry, Physical)Okamura, Hiroyuki; Ueda, Yuki; Motokawa, Ryuhei; Mu, J.*; Masters, A. J.*; Antonio, M. R.*
no journal, ,
Recently, cluster formation of metal-extractant complexes in organic solvents has been shown to play important roles in liquid-liquid extraction. Although a number of reaction mechanisms have been proposed for liquid-liquid extraction, application of slope analysis method for evaluating metal-extractant stoichiometry fails when the solute concentrations are high. In this study, we performed the analysis of solvent extraction equilibria for practical liquid-liquid systems with consideration of the formation of cluster aggregates in the organic phase during the extraction process. Molecular dynamics (MD) simulation indicates the formation of aggregated clusters of 1 to 9 Zr(NO )
) (TBP)
(TBP) complexes in
complexes in  -octane, leading to the determination of composition and molar fraction of each cluster. Furthermore, the extraction equilibrium reactions were studied, and the extraction equilibrium constant K
-octane, leading to the determination of composition and molar fraction of each cluster. Furthermore, the extraction equilibrium reactions were studied, and the extraction equilibrium constant K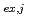 (
(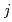 = 1-9) for each cluster could be determined from the parameters obtained by the MD analysis. The distribution curve calculated from K
= 1-9) for each cluster could be determined from the parameters obtained by the MD analysis. The distribution curve calculated from K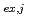 agrees well with the experimental values. Therefore, one MD simulation can accurately reproduce the distribution curve in the clustering/aggregation liquid-liquid extraction system, which enabled us to overcome the limitations of slope analysis method.
agrees well with the experimental values. Therefore, one MD simulation can accurately reproduce the distribution curve in the clustering/aggregation liquid-liquid extraction system, which enabled us to overcome the limitations of slope analysis method.
Okamura, Hiroyuki; Ueda, Yuki; Motokawa, Ryuhei; Mu, J.*; Masters, A. J.*; Antonio, M. R.*
no journal, ,
Under practical liquid-liquid extraction conditions with high solute concentrations, clusters of metal-extractant complexes can form in the organic phase. In these conditions, application of the slope analysis method for determining metal-extractant stoichiometry becomes difficult, and the deviations from ideal appear as non-linear responses and non-integer slopes. In this study, we developed novel extraction equilibrium analysis for practical liquid-liquid systems with consideration of the cluster formation and investigated the extension of the classical equilibrium analysis. Molecular dynamics (MD) simulation snapshots indicate the formation of aggregated clusters of 1 to 9 Zr(NO )
) (TBP)
(TBP) complexes in
complexes in  -octane, leading to the determination of composition and molar fraction of each cluster. Considering the extraction equilibrium of clusters composed of
-octane, leading to the determination of composition and molar fraction of each cluster. Considering the extraction equilibrium of clusters composed of 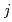 Zr(NO
Zr(NO )
) (TBP)
(TBP) complexes, the extraction equilibrium constants K
complexes, the extraction equilibrium constants K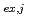 (
(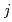 = 1-9) were calculated from the distribution ratio and the molar fraction for each cluster obtained by the MD analysis. The distribution curve calculated from the obtained K
= 1-9) were calculated from the distribution ratio and the molar fraction for each cluster obtained by the MD analysis. The distribution curve calculated from the obtained K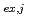 values agrees well with the experimental values. Therefore, MD simulation can accurately reproduce the experimental values in the clustering/aggregation liquid-liquid extraction system, which enabled us to extend the classical equilibrium analysis.
values agrees well with the experimental values. Therefore, MD simulation can accurately reproduce the experimental values in the clustering/aggregation liquid-liquid extraction system, which enabled us to extend the classical equilibrium analysis.