Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Tanaka, Taiki*; Narikiyo, Yoshihiro*; Morita, Kosuke*; Fujita, Kunihiro*; Kaji, Daiya*; Morimoto, Koji*; Yamaki, Sayaka*; Wakabayashi, Yasuo*; Tanaka, Kengo*; Takeyama, Mirei*; et al.
Journal of the Physical Society of Japan, 87(1), p.014201_1 - 014201_9, 2018/01
Times Cited Count:18 Percentile:74.47(Physics, Multidisciplinary)Excitation functions of quasielastic scattering cross sections for the  Ca +
Ca + 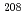 Pb,
Pb, 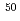 Ti +
Ti + 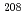 Pb, and
Pb, and  Ca +
Ca +  Cm reactions were successfully measured by using the gas-filled recoil-ion separator GARIS. Fusion barrier distributions were extracted from these data, and compared with the coupled-channels calculations. It was found that the peak energies of the barrier distributions for the
Cm reactions were successfully measured by using the gas-filled recoil-ion separator GARIS. Fusion barrier distributions were extracted from these data, and compared with the coupled-channels calculations. It was found that the peak energies of the barrier distributions for the  Ca +
Ca + 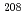 Pb and
Pb and 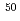 Ti +
Ti + 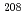 Pb systems coincide with those of the 2n evaporation channel cross sections for the systems, while that of the
Pb systems coincide with those of the 2n evaporation channel cross sections for the systems, while that of the  Ca +
Ca +  Cm is located slightly below the 4n evaporation ones. This results provide us helpful information to predict the optimum beam energy to synthesize superheavy nuclei.
Cm is located slightly below the 4n evaporation ones. This results provide us helpful information to predict the optimum beam energy to synthesize superheavy nuclei.
Yamazaki, Yuji*; Tamada, Taro; Kasai, Noriyuki*; Urakawa, Itaru*; Aono, Yukiko*; Hasegewa, Hisashi*; Fujita, Toshiro*; Kuroki, Ryota; Yamashita, Takeyoshi*; Fukumoto, Seiji*; et al.
Journal of Bone and Mineral Research, 23(9), p.1509 - 1518, 2008/09
Times Cited Count:150 Percentile:94.53Fibroblast growth factor (FGF)23 is proposed to play a physiological role in the regulation of phosphate and vitamin D metabolism; deranged circulatory levels of FGF23 cause several diseases with abnormal mineral metabolism. We developed two antibodies (FN1 and FC1) that recognize the N- and C-terminal regions of FGF23, respectively. Both FN1 and FC1 inhibited FGF23 activity in a cell-based Klotho-dependent reporter assay. The present study using neutralizing antibodies confirms that FGF23 is a physiological regulator of phosphate and vitamin D metabolism. Then, we addressed the mechanism of action for these neutralizing antibodies. Structural analysis of the FGF23/FN1-Fab complex revealed that FN1 masked putative FGF receptor-binding sites in the N-terminal domain of FGF23, while biochemical analyses showed that FC1 interfered with the association between FGF23 and Klotho by binding to the C-terminal domain of FGF23. Taken together, our results suggest that the N- and C-terminal domains of FGF23 are responsible for association with cognate FGF receptors and Klotho, respectively, and that these interactions are indispensable for FGF23 activity.
 -ray emitters including
-ray emitters including 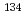 Cs, 1; Collaborative experiment by the committee on preparation of reference materials for radioactivity analysis of JSAC
Cs, 1; Collaborative experiment by the committee on preparation of reference materials for radioactivity analysis of JSACYonezawa, Chushiro*; Kakita, Kazutoshi*; Takahashi, Takanori*; Aono, Tatsuo*; Maeda, Satoshi; Abe, Takaaki*; Arakawa, Fumihiro*; Kiho, Nobuharu*; Akiyama, Masakazu*; Muramatsu, Isamu*; et al.
no journal, ,
no abstracts in English