Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yamashita, Shinichi; Baldacchino, G.*; Maeyama, Takuya*; Taguchi, Mitsumasa; Muroya, Yusa*; Lin, M.*; Kimura, Atsushi; Murakami, Takeshi*; Katsumura, Yosuke
Free Radical Research, 46(7), p.861 - 871, 2012/07
Times Cited Count:26 Percentile:54.24(Biochemistry & Molecular Biology)Radiation-induced reactions in aqueous solutions of a water-soluble coumarin derivative, coumarin-3-carboxyl acid (C3CA), have been investigated by pulse radiolysis with 35-MeV electron beam, final product analysis after  Co
Co 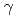 -irradiations, and deterministic model simulations. It was found that C3CA reacts with the hydroxyl radical (
-irradiations, and deterministic model simulations. It was found that C3CA reacts with the hydroxyl radical ( OH) as well as the hydrated electron at nearly diffusion-controlled rate constants: 6.8
OH) as well as the hydrated electron at nearly diffusion-controlled rate constants: 6.8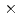 10
10 and 2.1
and 2.1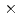 10
10 M
M s
s , respectively. Reactivity of C3CA toward O
, respectively. Reactivity of C3CA toward O

 was not confirmed. Production of a fluorescent molecule 7-hydroxy-coumarin-3-carboxylic acid (7OH-C3CA) was detected by a fluorescence spectrometer coupled with high performance liquid chromatography. Production yields of 7OH-C3CA were in a range from 0.025 to 0.18 (100 eV)
was not confirmed. Production of a fluorescent molecule 7-hydroxy-coumarin-3-carboxylic acid (7OH-C3CA) was detected by a fluorescence spectrometer coupled with high performance liquid chromatography. Production yields of 7OH-C3CA were in a range from 0.025 to 0.18 (100 eV) , depending on irradiation conditions. A variety of the yield with saturating gas, additive, and C3CA concentration implied that there are at least two pathways from scavenging reaction of C3CA toward
, depending on irradiation conditions. A variety of the yield with saturating gas, additive, and C3CA concentration implied that there are at least two pathways from scavenging reaction of C3CA toward  OH to 7OH-C3CA: peroxidation reaction followed by elimination of perhydroxyl radical and disproportionation reaction. A reaction mechanism involving the two pathways was proposed and incorporated into the simulations, showing good explanation of experimentally measured 7OH-C3CA yields with a constant conversion factor from
OH to 7OH-C3CA: peroxidation reaction followed by elimination of perhydroxyl radical and disproportionation reaction. A reaction mechanism involving the two pathways was proposed and incorporated into the simulations, showing good explanation of experimentally measured 7OH-C3CA yields with a constant conversion factor from  OH scavenging to 7OH-C3CA production, 4.7%, unless
OH scavenging to 7OH-C3CA production, 4.7%, unless 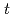 -BuOH is not added.
-BuOH is not added.
Maeyama, Takuya*; Yamashita, Shinichi; Taguchi, Mitsumasa; Baldacchino, G.*; Sihver, L.*; Murakami, Takeshi*; Katsumura, Yosuke
Radiation Physics and Chemistry, 80(12), p.1352 - 1357, 2011/12
Times Cited Count:15 Percentile:69.66(Chemistry, Physical)Coumari-3-carboxylic acid scavenges OH radical produced in water radiolysis, leading to production of a fluorescence probe at almost constant ratio relative to the amount of the scavenged OH radicals. This was applied in estimation of OH radical yield in water radiolysis especially with therapeutic heavy ions of GeV-class energies, i.e.  C
C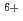 beams of 135, 290 and 400 MeV/u. OH yields upstream of the Bragg peaks decreased with increasing penetration depth of the projectile ions while that downstream suddenly jumped up to near the value for low-LET radiations such as
beams of 135, 290 and 400 MeV/u. OH yields upstream of the Bragg peaks decreased with increasing penetration depth of the projectile ions while that downstream suddenly jumped up to near the value for low-LET radiations such as  -rays. This is due to low-LET secondary fragmentation ions produced during long trajectory of the primary projectile C ion. Quantitative explanation by nuclear fragmentation simulations with PHITS code was attempted and resulted in 15-45% underestimation in the region behind the Bragg peaks, which would be due to the difference in geometries between irradiations of the sample solutions and dosimetry with a small ionization chamber.
-rays. This is due to low-LET secondary fragmentation ions produced during long trajectory of the primary projectile C ion. Quantitative explanation by nuclear fragmentation simulations with PHITS code was attempted and resulted in 15-45% underestimation in the region behind the Bragg peaks, which would be due to the difference in geometries between irradiations of the sample solutions and dosimetry with a small ionization chamber.
Maeyama, Takuya*; Yamashita, Shinichi; Baldacchino, G.*; Taguchi, Mitsumasa; Kimura, Atsushi; Murakami, Takeshi*; Katsumura, Yosuke
Radiation Physics and Chemistry, 80(4), p.535 - 539, 2011/04
Times Cited Count:41 Percentile:93.20(Chemistry, Physical)Aqueous coumarin-3-carboxylic Acid (3CCA) solutions were irradiated with eight different ion beams covering LET range from 0.5 to above 2000 eV/nm. 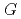 -values of 7OH-3CCA, one of hydroxylated products in radiolysis of the solutions, have been determined by fluorescence-HPLC technique in 3CCA concentration range from 0.1 to 26 mM. The formation yield of 7OH-3CCA increased with increasing concentration of 3CCA while it decreased with increasing LET value of ion beam. Compared with our previous reports on
-values of 7OH-3CCA, one of hydroxylated products in radiolysis of the solutions, have been determined by fluorescence-HPLC technique in 3CCA concentration range from 0.1 to 26 mM. The formation yield of 7OH-3CCA increased with increasing concentration of 3CCA while it decreased with increasing LET value of ion beam. Compared with our previous reports on 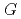 (
( OH) at a scavenging capacity of 10
OH) at a scavenging capacity of 10 s with absorption spectroscopy, it was found that
s with absorption spectroscopy, it was found that 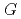 (7OH-3CCA) is about (4.7
(7OH-3CCA) is about (4.7 0.6)% of
0.6)% of 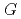 (
( OH), which is consistent for all of the ion beams used in the present study. However, 7OH-3CCA yields in high CCA concentration region, especially by using extremely high LET ions, were much higher than expected values based on the above conversion factor and
OH), which is consistent for all of the ion beams used in the present study. However, 7OH-3CCA yields in high CCA concentration region, especially by using extremely high LET ions, were much higher than expected values based on the above conversion factor and 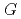 (
( OH) value predicted in theoretical work.
OH) value predicted in theoretical work.
Yamashita, Shinichi; Taguchi, Mitsumasa; Baldacchino, G.*; Katsumura, Yosuke
Charged Particle and Photon Interactions with Matter; Recent Advances, Applications, and Interfaces, p.325 - 354, 2010/12
Irradiation effects induced by heavy ions are much different from those by low-LET radiations. Such distinctiveness has been applied to cancer therapy, surface processing, analytical techniques, and so on. However, detailed mechanism through which such distinctiveness appears has not been clarified yet. In this chapter, what have experimentally been found recently, especially within these five years, in water radiolysis with positively-charged atomic heavy ions are shown. Steady-state radiolysis studies and pulse radiolysis studies are separately summarized because these two approaches inherently involve own advantages and limitations. Gathering reported products yields and kinetics, track structures and intra-track dynamics are discussed. One of the most characteristic features of recent studies is that track-segment or -differential yields were intensively measured compared to earlier studies.
Lin, M.; Muroya, Yusa*; Baldacchino, G.*; Katsumura, Yosuke*
Recent Trends in Radiation Chemistry, p.255 - 277, 2010/05
In this review paper, we have summarized the important results obtained in the last decade concerning the radiation chemistry studies on high temperature and supercritical water, especially the radiolytic yields of water decomposition products, the reaction rate constants, and the spectral properties of transient species, etc. The importance of density effects under supercritical conditions is pointed out.
Taguchi, Mitsumasa; Baldacchino, G.*; Kurashima, Satoshi; Kimura, Atsushi; Sugo, Yumi; Katsumura, Yosuke*; Hirota, Koichi
Radiation Physics and Chemistry, 78(12), p.1169 - 1174, 2009/12
Times Cited Count:4 Percentile:29.39(Chemistry, Physical)no abstracts in English
Baldacchino, G.*; Maeyama, Takuya*; Yamashita, Shinichi; Taguchi, Mitsumasa; Kimura, Atsushi; Katsumura, Yosuke; Murakami, Takeshi*
Chemical Physics Letters, 468(4-6), p.275 - 279, 2009/01
Times Cited Count:44 Percentile:81.92(Chemistry, Physical)This paper reports a sensitive method using HPLC-fluorescence detection of  OH in liquid water under high-energy heavy-ion irradiation. The coumarin-3-carboxylic-acid (3CCA) molecule was selected for probing
OH in liquid water under high-energy heavy-ion irradiation. The coumarin-3-carboxylic-acid (3CCA) molecule was selected for probing  OH and providing the fluorescent 7-hydroxy-coumarin-3-carboxylic-acid (7OH-3CCA). Since the concentration limit achievable is better than 1 nM, the radiolytic yields were determined with a sensitivity of 2
OH and providing the fluorescent 7-hydroxy-coumarin-3-carboxylic-acid (7OH-3CCA). Since the concentration limit achievable is better than 1 nM, the radiolytic yields were determined with a sensitivity of 2 10
10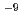 mol/J for 4.8-GeV-
mol/J for 4.8-GeV- C
C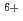 of and 20-GeV-
of and 20-GeV-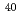 Ar
Ar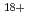 in the ns time-range. They decrease with the linear energy transfer from 2.8
in the ns time-range. They decrease with the linear energy transfer from 2.8 10
10 to 1.3
to 1.3 10
10 mol/J (
mol/J ( C
C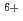 of 11 eV/nm) and 1.5
of 11 eV/nm) and 1.5 10
10 to 0.9
to 0.9 10
10 mol/J (
mol/J (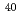 Ar
Ar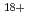 of 90 eV/nm) which is in agreement with the literature data.
of 90 eV/nm) which is in agreement with the literature data.
Taguchi, Mitsumasa; Kimura, Atsushi; Hirota, Koichi; Kurashima, Satoshi; Baldacchino, G.*; Katsumura, Yosuke*
JAEA-Review 2007-060, JAEA Takasaki Annual Report 2006, P. 158, 2008/03
no abstracts in English
Taguchi, Mitsumasa; Zennyoji, Yoshihiro*; Takigami, Machiko*; Baldacchino, G.*; Kimura, Atsushi; Hiratsuka, Hiroshi*; Namba, Hideki; Kojima, Takuji
Radiation Physics and Chemistry, 75(5), p.564 - 571, 2006/03
Times Cited Count:6 Percentile:40.23(Chemistry, Physical)The suspended fine particles formed in aqueous phenol solution under high dose irradiation were studied using the MeV electron beam and  Co
Co  -ray irradiations. The fine particle's size was 100-800 nm in diameter and became bigger at higher dose and lower dose rate. The distributions of the particle size became narrower for irradiation at higher dose rate. The fine particles were regarded to consist of molecules of about 1.9
-ray irradiations. The fine particle's size was 100-800 nm in diameter and became bigger at higher dose and lower dose rate. The distributions of the particle size became narrower for irradiation at higher dose rate. The fine particles were regarded to consist of molecules of about 1.9 10
10 molecular weight.
molecular weight.
Taguchi, Mitsumasa; Kimura, Atsushi; Hirota, Koichi; Kurashima, Satoshi; Baldacchino, G.*; Katsumura, Yosuke*
no journal, ,
no abstracts in English
Taguchi, Mitsumasa; Kimura, Atsushi; Baldacchino, G.*; Katsumura, Yosuke*; Hirota, Koichi
no journal, ,
Heavy ions give unique irradiation effects on target materials. Reactions in water under heavy ion irradiation are mainly induced by OH (hydroxyl) radicals. OH radicals yields have been investigated by the (1) product analysis and (2) pulse radiolysis methods. (1) Aqueous phenol solutions were irradiated with He, C and Ne ions. The yields of the OH radicals were estimated by analyzing the yields of the irradiation products of phenol. The yields of the OH radicals increased with the specific energy, but decreased with both the mass of each ion and elapsed time. (2) Transient absorption measurement system was developed for the direct observation of radical behaviors in water under heavy ion irradiation. A small absorbance change less than 10 was recorded. The transient absorbance of (SCN)
was recorded. The transient absorbance of (SCN)
 formed by the reaction of OH radicals with SCN
formed by the reaction of OH radicals with SCN in aqueous KSCN solution was observed under pulsed C ion irradiation.
in aqueous KSCN solution was observed under pulsed C ion irradiation.
Baldacchino, G.*; Taguchi, Mitsumasa; Hichel, B.*
no journal, ,
no abstracts in English
 OH yields with fluorescent probe, 1; Investigation into reaction mechanism and reactivity toward water decomposition radicals of coumarin-3-carboxylic acid
OH yields with fluorescent probe, 1; Investigation into reaction mechanism and reactivity toward water decomposition radicals of coumarin-3-carboxylic acidMaeyama, Takuya*; Yamashita, Shinichi; Baldacchino, G.*; Muroya, Yusa*; Taguchi, Mitsumasa; Kimura, Atsushi; Katsumura, Yosuke
no journal, ,
no abstracts in English
Maeyama, Takuya*; Yamashita, Shinichi; Baldacchino, G.*; Taguchi, Mitsumasa; Kimura, Atsushi; Katsumura, Yosuke; Murakami, Takeshi*
no journal, ,
no abstracts in English
Taguchi, Mitsumasa; Kurashima, Satoshi; Baldacchino, G.*; Sugo, Yumi; Kimura, Atsushi; Hirota, Koichi; Katsumura, Yosuke*
no journal, ,
no abstracts in English
 OH yield near the Bragg peak
OH yield near the Bragg peakMaeyama, Takuya*; Yamashita, Shinichi; Katsumura, Yosuke; Baldacchino, G.*; Taguchi, Mitsumasa; Kimura, Atsushi; Murakami, Takeshi*
no journal, ,
Aqueous solution of Coumarin-3-carboxylic acid (CCA) has been applied to yield measurement of  OH in water. Production yield of a fluorescent probe, 7OH-CCA, which is a stable product produced after scavenging reaction for
OH in water. Production yield of a fluorescent probe, 7OH-CCA, which is a stable product produced after scavenging reaction for  OH by CCA, was determined by using HPLC connected to a fluorometer. Taking sensitiveness of fluorometry as an advantage,
OH by CCA, was determined by using HPLC connected to a fluorometer. Taking sensitiveness of fluorometry as an advantage,  OH yields near the Bragg peaks, which are known very small compared to those in plateau LET region, have been measured. Contribution of fragmentations, which are known to be significant near the Bragg peaks of high-energy heavy ions, is also being discussed.
OH yields near the Bragg peaks, which are known very small compared to those in plateau LET region, have been measured. Contribution of fragmentations, which are known to be significant near the Bragg peaks of high-energy heavy ions, is also being discussed.
Taguchi, Mitsumasa; Baldacchino, G.*; Kimura, Atsushi; Kurashima, Satoshi; Hirota, Koichi
no journal, ,
no abstracts in English
Taguchi, Mitsumasa; Kurashima, Satoshi; Baldacchino, G.*; Kimura, Atsushi; Sugo, Yumi; Katsumura, Yosuke*; Hirota, Koichi
no journal, ,
no abstracts in English
Taguchi, Mitsumasa; Kurashima, Satoshi; Sugo, Yumi; Kimura, Atsushi; Hirota, Koichi; Baldacchino, G.*; Katsumura, Yosuke*
no journal, ,
no abstracts in English
Taguchi, Mitsumasa; Baldacchino, G.*; Kurashima, Satoshi; Kimura, Atsushi; Katsumura, Yosuke*; Hirota, Koichi
no journal, ,
no abstracts in English