Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Fukazawa, Hiroshi; Arakawa, Masashi*; Kagi, Hiroyuki; Yamauchi, Hiroki; Chakoumakos, B. C.*; Fernandez-Baca, J. A.*
Physics and Chemistry of Ice 2010, p.421 - 428, 2011/03
Whether or not ice in the universe exists as ferroelectric is a question that has attracted interest in astrophysics and physical chemistry, because long-range electrostatic forces, caused by the ferroelectricity, might be an important factor for materials evolution and hydrogen bonding. From neutron diffraction and scattering measurements, we have studied ice with impurities, such as potassium, sodium and lithium, that acted as a catalyst. Time-resolved diffraction experiments show that ferroelectric ice XI with hydrogen-ordered arrangement nucleates and grows with time for about 5 days. We found that the doped ice that has once been converted to ice XI is a stronger ferroelectric ice than that has never been converted. We also show the existence of the ferroelectric ice under high-pressure and its formation from compressed amorphous ice.
Arakawa, Masashi*; Kagi, Hiroyuki; Fernandez-Baca, J. A.*; Chakoumakos, B. C.*; Fukazawa, Hiroshi
Physics and Chemistry of Ice 2010, p.329 - 338, 2011/03
We measured neutron diffraction profiles of KOD, NaOD, LiOD, Ca(OD) , and ND
, and ND -doped ices. Ice XI, which is a hydrogen-ordered phase of normal ice (ice Ih), was observed in the KOD and NaOD-doped ices although Ca(OD)
-doped ices. Ice XI, which is a hydrogen-ordered phase of normal ice (ice Ih), was observed in the KOD and NaOD-doped ices although Ca(OD) and ND
and ND -doped ice did not transformed to ice XI. The mass fraction of ice XI to that of the doped ice (
-doped ice did not transformed to ice XI. The mass fraction of ice XI to that of the doped ice (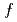 ) was estimated using Rietveld analysis for each doped ice. The
) was estimated using Rietveld analysis for each doped ice. The 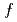 value of the doped ice, which had once experienced being ice XI, was larger than that of the doped ice, which had never experienced being ice XI. The large
value of the doped ice, which had once experienced being ice XI, was larger than that of the doped ice, which had never experienced being ice XI. The large 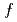 value of the doped ice indicates that small hydrogen-ordered domains remained above the transition temperature between ice XI and Ih. Our results suggest that large amounts of ice on icy bodies in our solar system can transform to ice XI. In this paper, we discussed the existence of the small hydrogen-ordered domains in space and the evolution of icy grain.
value of the doped ice indicates that small hydrogen-ordered domains remained above the transition temperature between ice XI and Ih. Our results suggest that large amounts of ice on icy bodies in our solar system can transform to ice XI. In this paper, we discussed the existence of the small hydrogen-ordered domains in space and the evolution of icy grain.
Fukazawa, Hiroshi; Hoshikawa, Akinori*; Chakoumakos, B. C.*; Fernandez-Baca, J. A.*
Nuclear Instruments and Methods in Physics Research A, 600(1), p.279 - 281, 2009/02
Times Cited Count:12 Percentile:60.72(Instruments & Instrumentation)We measured time-resolved neutron powder diffraction of ices doped with very small amounts of some catalysts using the wide-angle neutron diffractometer (WAND) at the High Flax Isotope Reactor (HFIR) in Oak Ridge, Tennessee. We succeeded in making ferroelectric ice, which prepared at high pressures (0.2 and 0.4 GPa), in a 60 to 75 K temperature range over tens hours. Neutron diffraction experiments confirmed the existence of a large ferroelectric structure in the sample. Therefore, thick surface (about 500 Km) of ferroelectric ice may exist on cold icy bodies, such as, Pluto and Kuiper Belt Objects (KBO). It suggests that about 70% of Pluto body is ferroelectric ice. Future telescope or planetary probe will be able to detect the huge ferroelectric-ice mass. In this symposium we report recent results at the HFIR and our future plan using higher intensity neutron beam.
Fukazawa, Hiroshi; Hoshikawa, Akinori; Ishii, Yoshinobu; Chakoumakos, B. C.*; Fernandez-Baca, J. A.*
Astrophysical Journal, 652(1, Part2), p.L57 - L60, 2006/11
Times Cited Count:47 Percentile:71.00(Astronomy & Astrophysics)Whether ferroelectric ice, named ice XI, exists in a stable low-temperature phase attracts much interest. This question arose as a condensed-matter issue, and became of interest in astronomy (e.g., does ice XI exist on Pluto?) because astronomical observations identified the existence of crystalline ice in our solar system. From neutron diffraction experiments, we found the temperature conditions for the transformation of the largest fraction of ice into ice XI using the lowest level of impurity dopant. The finding of bulk crystal of ordered structure firmly supports that ice XI is stable. This suggests the existence of naturally occurring ice XI at a narrow temperature range (57-66 K) in our solar system.
Rondinone, A. J.*; Chakoumakos, B. C.*; Rawn, C. J.*; Ishii, Yoshinobu
Journal of Physical Chemistry B, 107(25), p.6046 - 6050, 2003/06
Times Cited Count:22 Percentile:48.51(Chemistry, Physical)no abstracts in English
 hydrate; Synthesis, composition, structure, dissociation behavior, and a comparison to structure I CH
hydrate; Synthesis, composition, structure, dissociation behavior, and a comparison to structure I CH hydrate
hydrateCircone, S.*; Stern, L. A.*; Kirby, S. H.*; Durham, W. B.*; Chakoumakos, B. C.*; Rawn, C. J.*; Rondinone, A. J.*; Ishii, Yoshinobu
Journal of Physical Chemistry B, 107(23), p.5529 - 5539, 2003/06
Times Cited Count:197 Percentile:96.08(Chemistry, Physical)no abstracts in English
Jones, C. Y.*; Marshall, S. L.*; Chakoumakos, B. C.*; Rawn, C. J.*; Ishii, Yoshinobu
Journal of Physical Chemistry B, 107(25), p.6026 - 6031, 2003/03
Times Cited Count:40 Percentile:68.43(Chemistry, Physical)no abstracts in English
Rondinone, A. J.*; Chakoumakos, B. C.*; Rawn, C. J.*; Ishii, Yoshinobu
Proceedings of 4th International Conference on Gas Hydrates (ICGH-4), p.625 - 629, 2002/05
no abstracts in English
Rawn, C. J.*; Rondinone, A. J.*; Chakoumakos, B. C.*; Marshall, S. L.*; Stern, L. A.*; Circone, S.*; Kirby, S. H.*; Jones, C. Y.*; Toby, B. H.*; Ishii, Yoshinobu
Proceedings of 4th International Conference on Gas Hydrates (ICGH-4), p.595 - 598, 2002/05
no abstracts in English
Chakoumakos, B. C.*; Rawn, C. J.*; Rondinone, A. J.*; Marshall, S. L.*; Stern, L. A.*; Circone, S.*; Kirby, S. H.*; Jones, C. Y.*; Toby, B. H.*; Ishii, Yoshinobu
Proceedings of 4th International Conference on Gas Hydrates (ICGH-4), p.655 - 658, 2002/05
no abstracts in English
Sekine, Yurina; Fukazawa, Hiroshi; Songxue, C.*; Chakoumakos, B.*; Fernandez-Baca, J. A.*
no journal, ,
no abstracts in English
Sekine, Yurina; Fukazawa, Tomoko*; Yamauchi, Hiroki; Songxue, C.*; Chakoumakos, B.*; Fernandez-Baca, J. A.*; Fukazawa, Hiroshi
no journal, ,
Neutron diffraction data of deuterated poly-N,N,-dimethylacrylamide (PDMAA) hydrogel were measured at temperatures of 10 - 290 K in order to investigate the structure and properties of water in the hydrogel. Diffraction peaks observed below 270 K indicate the existence of a solid ice in the hydrogel. The positions of some peaks from the ice are lower than those in ordinary hexagonal ice (Ih). The lower shifts of the peaks indicates that the lattice constants of the c-axis in the ice are about 0.3% higher than those in ice Ih, whereas those of the a-axis are almost consist. The results show that bulk of a low-density ice crystal exists in the PDMAA hydrogel. The water molecules, which have a significant interaction with the polymer chains, play an important role in the chemical and mechanical properties of the hydrogel.