Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -octylnitrilo-triacetamide (HONTA) complexes of americium and europium
-octylnitrilo-triacetamide (HONTA) complexes of americium and europiumToigawa, Tomohiro; Peterman, D. R.*; Meeker, D. S.*; Grimes, T. S.*; Zalupski, P. R.*; Mezyk, S. P.*; Cook, A. R.*; Yamashita, Shinichi*; Kumagai, Yuta; Matsumura, Tatsuro; et al.
Physical Chemistry Chemical Physics, 23(2), p.1343 - 1351, 2021/01
Times Cited Count:14 Percentile:82.54(Chemistry, Physical)The candidate An(III)/Ln(III) separation ligand hexa- -octylnitrilo-triacetamide (HONTA) was irradiated under envisioned SELECT (Solvent Extraction from Liquid waste using Extractants of CHON-type for Transmutation) process conditions using a solvent test loop in conjunction with cobalt-60 gamma irradiation. We demonstrate that HONTA undergoes exponential decay with increasing gamma dose to produce a range of degradation products which have been identified and quantified by HPLC-ESI-MS/MS techniques. The combination of HONTA destruction and degradation product ingrowth, particularly dioctylamine, negatively impacts the extraction and back-extraction of both americium and europium ions. The loss of HONTA was attributed to its reaction with the solvent (
-octylnitrilo-triacetamide (HONTA) was irradiated under envisioned SELECT (Solvent Extraction from Liquid waste using Extractants of CHON-type for Transmutation) process conditions using a solvent test loop in conjunction with cobalt-60 gamma irradiation. We demonstrate that HONTA undergoes exponential decay with increasing gamma dose to produce a range of degradation products which have been identified and quantified by HPLC-ESI-MS/MS techniques. The combination of HONTA destruction and degradation product ingrowth, particularly dioctylamine, negatively impacts the extraction and back-extraction of both americium and europium ions. The loss of HONTA was attributed to its reaction with the solvent ( -dodecane) radical cation of
-dodecane) radical cation of 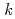 (HONTA + R
(HONTA + R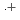 ) = (7.61
) = (7.61  0.82)
0.82)  10
10 M
M s
s obtained by pulse radiolysis techniques. However, when this ligand is bound to either americium or europium ions, the observed
obtained by pulse radiolysis techniques. However, when this ligand is bound to either americium or europium ions, the observed  -dodecane radical cation kinetics increase by over an order of magnitude. This large reactivity increase to additional reaction pathways occurring upon metal-ion binding. Lastly nanosecond time-resolved measurements showed that both direct and indirect HONTA radiolysis yielded the short-lived (
-dodecane radical cation kinetics increase by over an order of magnitude. This large reactivity increase to additional reaction pathways occurring upon metal-ion binding. Lastly nanosecond time-resolved measurements showed that both direct and indirect HONTA radiolysis yielded the short-lived ( 100 ns) HONTA radical cation as well as a longer-lived (
100 ns) HONTA radical cation as well as a longer-lived ( s) HONTA triplet excited state. These HONTA species are important precursors to the suite of HONTA degradation products observed.
s) HONTA triplet excited state. These HONTA species are important precursors to the suite of HONTA degradation products observed.
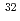 Mg; Intruder amplitudes in
Mg; Intruder amplitudes in 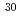 Ne and implications for the binding of
Ne and implications for the binding of 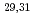 F
FFallon, P.*; Rodriguez-Vieitez, E.*; Macchiavelli, A. O.*; Gade, A.*; Tostevin, J. A.*; Adrich, P.*; Bazin, D.*; Bowen, M.*; Campbell, C. M.*; Clark, R. M.*; et al.
Physical Review C, 81(4), p.041302_1 - 041302_5, 2010/04
Times Cited Count:40 Percentile:88.58(Physics, Nuclear)no abstracts in English
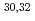 Mg; Mapping the transition into the "island of inversion"
Mg; Mapping the transition into the "island of inversion"Terry, J. R.*; Brown, B. A.*; Campbell, C. M.*; Cook, J. M.*; Davies, A. D.*; Dinca, D.-C.*; Gade, A.*; Glasmacher, T.*; Hansen, P. G.*; Sherrill, B. M.*; et al.
Physical Review C, 77(1), p.014316_1 - 014316_12, 2008/01
Times Cited Count:77 Percentile:95.29(Physics, Nuclear)no abstracts in English
Sato, Tomonori; Shao, Y.*; Cook, W. G.*; Lister, D. H.*; Uchida, Shunsuke
Corrosion, 63(8), p.770 - 780, 2007/08
Times Cited Count:23 Percentile:73.01(Materials Science, Multidisciplinary)Flow-assisted corrosion (FAC) has often caused serious damage to carbon steel piping in nuclear power plants. As a first stage of experiments to determine the effects of water chemistry parameters on FAC, corrosion rates of carbon steel were measured in 140 degrees centigrade pure water with an on-line corrosion rate monitor based on electrical resistance measurement as [O ] and flow velocity were changed. The data were compiled as a function of electrochemical corrosion potential (ECP) and flow velocity. The conclusions are summarized as follows; (1) The FAC rate was below the detectable limit in highly oxygenated conditions, where [O
] and flow velocity were changed. The data were compiled as a function of electrochemical corrosion potential (ECP) and flow velocity. The conclusions are summarized as follows; (1) The FAC rate was below the detectable limit in highly oxygenated conditions, where [O ] was greater than 50 ppb and ECP was above than -0.2 V-SHE. (2) The effects of pre-oxidation treatment disappeared rapidly under deaerated conditions. (3) It is strongly recommended that the effects of flow on FAC rate should be evaluated from the viewpoint of Reynolds number as well as flow velocity.
] was greater than 50 ppb and ECP was above than -0.2 V-SHE. (2) The effects of pre-oxidation treatment disappeared rapidly under deaerated conditions. (3) It is strongly recommended that the effects of flow on FAC rate should be evaluated from the viewpoint of Reynolds number as well as flow velocity.
 I/
I/ I ratios in surface waters of the English Lake District
I ratios in surface waters of the English Lake DistrictAtarashi-Andoh, Mariko; Schnabel, C.*; Cook, G.*; MacKenzie, A. B.*; Dougans, A.*; Ellam, R. M.*; Freeman, S.*; Maden, C.*; Olive, V.*; Synal, H.-A.*; et al.
Applied Geochemistry, 22(3), p.628 - 636, 2007/03
Times Cited Count:16 Percentile:39.17(Geochemistry & Geophysics)Accelerator Mass Spectrometry (AMS) was used to measure  I/
I/ I ratios in surface sea, lake and river water samples collected in 2004 and 2005 from the English Lake District and from South west Scotland, areas which are in relatively close proximity to the Sellafield nuclear fuel reprocessing plant in north west England. The
I ratios in surface sea, lake and river water samples collected in 2004 and 2005 from the English Lake District and from South west Scotland, areas which are in relatively close proximity to the Sellafield nuclear fuel reprocessing plant in north west England. The  I/
I/ I ratios in surface water collected from the shore of the Irish Sea were in the range 2.8
I ratios in surface water collected from the shore of the Irish Sea were in the range 2.8 10
10 to 8.2
to 8.2 10
10 . These ratios are one order of magnitude higher than that of seawater collected from the Irish Sea in 1992, correlating with the increase in
. These ratios are one order of magnitude higher than that of seawater collected from the Irish Sea in 1992, correlating with the increase in  I content of the Sellafield liquid effluent discharge over the last decade. The
I content of the Sellafield liquid effluent discharge over the last decade. The  I/
I/ I ratios in lakes in the Lake District were in the range 0.7
I ratios in lakes in the Lake District were in the range 0.7 10
10 to 6.4
to 6.4 10
10 and decreased exponentially as a function of distance from Sellafield.
and decreased exponentially as a function of distance from Sellafield.
Terry, J. R.*; Basin, D.*; Brown, B. A.*; Campbell, C. M.*; Church, J. A.*; Cook, J. M.*; Davies, A. D.*; Dinca, D.-C.*; Enders, J.*; Gade, A.*; et al.
Physics Letters B, 640(3), p.86 - 90, 2006/09
Times Cited Count:59 Percentile:93.13(Astronomy & Astrophysics)no abstracts in English
Gulden, W.*; Cook, I.*; Marbach, G.*; Raeder, J.*; Petti, D.*; Seki, Yasushi
Fusion Engineering and Design, 51-52(Part.B), p.419 - 427, 2000/11
Times Cited Count:5 Percentile:37.61(Nuclear Science & Technology)no abstracts in English