Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
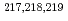 At revealed by in-source laser spectroscopy
At revealed by in-source laser spectroscopyBarzakh, A. E.*; Cubiss, J. G.*; Andreyev, A. N.; Seliverstov, M. D.*; Andel, B.*; Antalic, S.*; Ascher, P.*; Atanasov, D.*; Beck, D.*; Biero , J.*; et al.
, J.*; et al.
Physical Review C, 99(5), p.054317_1 - 054317_9, 2019/05
Times Cited Count:12 Percentile:77.09(Physics, Nuclear)Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:27 Percentile:69.46(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
Rothe, S.*; Andreyev, A. N.*; Antalic, S.*; Borschevsky, A.*; Capponi, L.*; Cocolios, T. E.*; De Witte, H.*; Eliav, E.*; Fedorov, D. V.*; Fedosseev, V. N.*; et al.
Nature Communications (Internet), 4, p.1835_1 - 1835_6, 2013/05
Times Cited Count:83 Percentile:94.59(Multidisciplinary Sciences)