Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Ge
Ge
藤原 秀紀*; 中谷 泰博*; 荒谷 秀和*; 金井 惟奈*; 山神 光平*; 濱本 諭*; 木須 孝幸*; 関山 明*; 田中 新*; 海老原 孝雄*; et al.
New Physics; Sae Mulli, 73(12), p.1062 - 1066, 2023/12
We have performed soft X-ray absorption spectroscopy (XAS) on the heavy fermion superconductor CeNi Ge
Ge to examine the local 4
to examine the local 4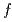 electronic structure. The XAS spectra are qualitatively explained by ionic calculations, indicating a degree of localized character of the 4
electronic structure. The XAS spectra are qualitatively explained by ionic calculations, indicating a degree of localized character of the 4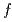 states, while sizable
states, while sizable 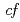 -hybridization effects are also observed in the XAS spectra and those magnetic circular dichroism. In addition, the temperature dependence of linear dichroism in the XAS spectra indicates that the ground state symmetry is
-hybridization effects are also observed in the XAS spectra and those magnetic circular dichroism. In addition, the temperature dependence of linear dichroism in the XAS spectra indicates that the ground state symmetry is 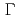
 , and the excited states with
, and the excited states with 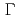
 symmetry is above 390 K.
symmetry is above 390 K.
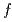 symmetry for anisotropic
symmetry for anisotropic 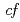 hybridization in the heavy-fermion superconductor CeNi
hybridization in the heavy-fermion superconductor CeNi Ge
Ge
藤原 秀紀*; 中谷 泰博*; 荒谷 秀和*; 金井 惟奈*; 山神 光平*; 濱本 諭*; 木須 孝幸*; 山崎 篤志*; 東谷 篤志*; 今田 真*; et al.
Physical Review B, 108(16), p.165121_1 - 165121_10, 2023/10
被引用回数:0 パーセンタイル:0(Materials Science, Multidisciplinary)We report the ground-state symmetry of the Ce 4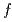 states in the heavy-fermion superconductor CeNi
states in the heavy-fermion superconductor CeNi Ge
Ge , yielding anisotropic
, yielding anisotropic 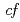 hybridization between the Ce 4
hybridization between the Ce 4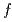 states and conducting electrons. By analyzing linear dichroism in soft X-ray absorption and core-level hard X-ray photoemission spectra, the 4
states and conducting electrons. By analyzing linear dichroism in soft X-ray absorption and core-level hard X-ray photoemission spectra, the 4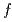 symmetry is determined as
symmetry is determined as  -type
-type 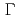
 , promoting predominant hybridization with the conducting electrons originating from the Ge site. The band structures probed by soft X-ray angle-resolved photoemission indicates that the Ge 4
, promoting predominant hybridization with the conducting electrons originating from the Ge site. The band structures probed by soft X-ray angle-resolved photoemission indicates that the Ge 4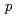 components contribute to the band renormalization through the anisotropic hybridization effects, suggesting that the control of the electronic structures of Ge orbital gives an impact to achieve the exotic phenomena in CeNi
components contribute to the band renormalization through the anisotropic hybridization effects, suggesting that the control of the electronic structures of Ge orbital gives an impact to achieve the exotic phenomena in CeNi Ge
Ge .
.
古池 美彦*; Ouyang, D.*; 富永 大輝*; 松尾 龍人*; 向山 厚*; 川北 至信; 藤原 悟*; 秋山 修志*
Communications Physics (Internet), 5(1), p.75_1 - 75_12, 2022/04
被引用回数:4 パーセンタイル:67.44(Physics, Multidisciplinary)Circadian clock proteins often reveal temperature-compensatory responses that counteract temperature influences to keep their enzymatic activities constant over a physiological range of temperature. This temperature-compensating ability at the reaction level is likely crucial for circadian clock systems, to which the clock proteins are incorporated, to achieve the system-level temperature compensation of the oscillation frequency. Nevertheless, temperature compensation is yet a puzzling phenomenon, since side chains that make up the clock proteins fluctuate more frequently due to greater thermal energy at higher temperature. Here, we investigated temperature influences on the dynamics of KaiC, a temperature-compensated enzyme (ATPase) that hydrolyzes ATP into ADP in the cyanobacterial circadian clock system, using quasielastic neutron scattering. The frequency of picosecond to subnanosecond incoherent local motions in KaiC was accelerated by a factor of only 1.2 by increasing the temperature by 10 C. This temperature insensitivity of the local motions was not necessarily unique to KaiC, but confirmed also for a series of temperature-sensitive mutants of KaiC and proteins other than clock-related proteins. Rather, the dynamics associated with the temperature-compensatory nature of the reaction- and system-level was found in global diffusional motions, which was suggested to regulate the temperature dependence of ATPase activity and dephosphorylation process presumably through changes in the hexamer conformation of KaiC. The spatiotemporal scale at which cross-scale causality of the temperature sensitivity is established is finite, and extends down to picosecond to subnanosecond dynamics only in a very limited part of KaiC, not in its entire part.
C. This temperature insensitivity of the local motions was not necessarily unique to KaiC, but confirmed also for a series of temperature-sensitive mutants of KaiC and proteins other than clock-related proteins. Rather, the dynamics associated with the temperature-compensatory nature of the reaction- and system-level was found in global diffusional motions, which was suggested to regulate the temperature dependence of ATPase activity and dephosphorylation process presumably through changes in the hexamer conformation of KaiC. The spatiotemporal scale at which cross-scale causality of the temperature sensitivity is established is finite, and extends down to picosecond to subnanosecond dynamics only in a very limited part of KaiC, not in its entire part.
草野 完也*; 一本 潔*; 石井 守*; 三好 由純*; 余田 成男*; 秋吉 英治*; 浅井 歩*; 海老原 祐輔*; 藤原 均*; 後藤 忠徳*; et al.
Earth, Planets and Space (Internet), 73(1), p.159_1 - 159_29, 2021/12
被引用回数:6 パーセンタイル:51.19(Geosciences, Multidisciplinary)PSTEPとは、2015年4月から2020年3月まで日本国内の太陽・地球惑星圏に携わる研究者が協力して実施した科研費新学術領域研究である。この研究枠組みから500以上の査読付き論文が発表され、様々なセミナーやサマースクールが実施された。本論文では、その成果をまとめて報告する。
武田 哲明*; 稲垣 嘉之; 相原 純; 青木 健; 藤原 佑輔; 深谷 裕司; 後藤 実; Ho, H. Q.; 飯垣 和彦; 今井 良行; et al.
High Temperature Gas-Cooled Reactors; JSME Series in Thermal and Nuclear Power Generation, Vol.5, 464 Pages, 2021/02
本書は、原子力機構における今までの高温ガス炉の研究開発の総括として、HTTRの設計、燃料、炉内構造物や中間熱交換器などの要素技術の開発、出力上昇試験、950 Cの高温運転、安全性実証試験などの運転経験及び成果についてまとめたものである。また、HTTRでの知見をもとに、商用炉の設計、高性能燃料、ヘリウムガスタービン、ISプロセスによる水素製造などの要素技術開発の現状について記述しており、今後の高温ガス炉の開発に非常に有用である。本書は、日本機械学会の動力エネルギーシステム部門による化石燃料及び原子力によるエネルギーシステムの技術書のシリーズの一冊として刊行されるものである。
Cの高温運転、安全性実証試験などの運転経験及び成果についてまとめたものである。また、HTTRでの知見をもとに、商用炉の設計、高性能燃料、ヘリウムガスタービン、ISプロセスによる水素製造などの要素技術開発の現状について記述しており、今後の高温ガス炉の開発に非常に有用である。本書は、日本機械学会の動力エネルギーシステム部門による化石燃料及び原子力によるエネルギーシステムの技術書のシリーズの一冊として刊行されるものである。
藤原 悟*; 松尾 龍人*; 杉本 泰伸*; 柴田 薫
Journal of Physical Chemistry Letters (Internet), 10(23), p.7505 - 7509, 2019/12
被引用回数:4 パーセンタイル:21.85(Chemistry, Physical)無秩序なポリペプチド鎖のダイナミクスの特性評価は、本質的に無秩序状態なタンパク質およびフォールディングプロセスに関連する非ネイティブ状態下のタンパク質の挙動を解明するために必要である。本研究では、小角X線散乱測定データと動的光散乱測定データと組み合わせて準弾性中性子散乱測定データから、タンパク質のセグメント運動と分子全体の拡散および局所側鎖運動を評価する方法を独自に開発した。そしてこの方法を、非フォールディング状態およびメルトグロビュール(MG)状態のタンパク質RNase Aに適用し、セグメント運動から生じる拡散係数を評価し、非フォールディング状態とMG状態で異なる値をとることを明らかにした。またこの方法で得られた値は、蛍光現象を用いた別の測定技術を使用して得られた値と一致していることも確認できた。これらの研究成果は、この方法の、さまざまな無秩序状態でのタンパク質の挙動を特徴付ける実行可能性だけでなく、有用性も示している。
 -synuclein related to propensity to amyloid fibril formation
-synuclein related to propensity to amyloid fibril formation藤原 悟*; 河野 史明*; 松尾 龍人*; 杉本 泰伸*; 松本 友治*; 成田 哲博*; 柴田 薫
Journal of Molecular Biology, 431(17), p.3229 - 3245, 2019/08
被引用回数:12 パーセンタイル:49.22(Biochemistry & Molecular Biology)パーキンソン病発症には、脳細胞中の「 -シヌクレイン」というタンパク質が線維状に集合した状態(「アミロイド線維」と呼ばれる)となることが関係すると考えられており、どのようなメカニズムでこのアミロイド線維が形成されるのかに強い関心が寄せられている。そこで研究チームは、タンパク質分子の「動き」に着目し、アミロイド線維のできやすさが様々に異なった条件でのタンパク質の動きを、J-PARCの中性子準弾性散乱装置を用いて調べた。その結果、タンパク質分子の動きの違いによりアミロイド線維のできやすさが変わること、特にアミロイド線維ができるためには、タンパク質同士が集合しやすくなるような特定の動きが必要なことを明らかにした。
-シヌクレイン」というタンパク質が線維状に集合した状態(「アミロイド線維」と呼ばれる)となることが関係すると考えられており、どのようなメカニズムでこのアミロイド線維が形成されるのかに強い関心が寄せられている。そこで研究チームは、タンパク質分子の「動き」に着目し、アミロイド線維のできやすさが様々に異なった条件でのタンパク質の動きを、J-PARCの中性子準弾性散乱装置を用いて調べた。その結果、タンパク質分子の動きの違いによりアミロイド線維のできやすさが変わること、特にアミロイド線維ができるためには、タンパク質同士が集合しやすくなるような特定の動きが必要なことを明らかにした。
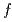 symmetry in CeCu
symmetry in CeCu Ge
Ge ; Soft X-ray absorption and hard X-ray photoemission spectroscopy
; Soft X-ray absorption and hard X-ray photoemission spectroscopy荒谷 秀和*; 中谷 泰博*; 藤原 秀紀*; 川田 萌樹*; 金井 惟奈*; 山神 光平*; 藤岡 修平*; 濱本 諭*; 久我 健太郎*; 木須 孝幸*; et al.
Physical Review B, 98(12), p.121113_1 - 121113_6, 2018/09
被引用回数:5 パーセンタイル:25.82(Materials Science, Multidisciplinary)We present a detailed study on the 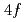 ground state symmetry of the pressure-induced superconductor CeCu
ground state symmetry of the pressure-induced superconductor CeCu Ge
Ge probed by soft X-ray absorption and hard X-ray photoemission spectroscopy. The revised Ce
probed by soft X-ray absorption and hard X-ray photoemission spectroscopy. The revised Ce 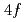 ground states are determined as
ground states are determined as 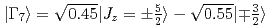 with
with 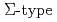 in-plane rotational symmetry. This gives an in-plane magnetic moment consistent with the antiferromagnetic moment as reported in neutron measurements. Since the in-plane symmetry is the same as that for the superconductor CeCu
in-plane rotational symmetry. This gives an in-plane magnetic moment consistent with the antiferromagnetic moment as reported in neutron measurements. Since the in-plane symmetry is the same as that for the superconductor CeCu Si
Si , we propose that the charge distribution along the
, we propose that the charge distribution along the 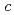 -axis plays an essential role in driving the system into a superconducting phase.
-axis plays an essential role in driving the system into a superconducting phase.
松尾 龍人; 荒田 敏昭*; 小田 俊郎*; 中島 健次; 河村 聖子; 菊地 龍弥; 藤原 悟
Biochemistry and Biophysics Reports (Internet), 6, p.220 - 225, 2016/07
Hydration water is essential for a protein to perform its biological function properly. In this study, the dynamics of hydration water around F-actin and myosin subfragment-1 (S1), which are the partner proteins playing a major role in various cellular functions related to cell motility, was characterized by incoherent quasielastic neutron scattering (QENS). The QENS spectra of hydration water around F-actin and S1 provided the translational diffusion coefficient, the residence time, and the rotational correlation time. The differences in these parameters indicate a significant difference in mobility of the hydration water between S1 and F-actin: S1 has the typical hydration water, the mobility of which is reduced compared with that of bulk water, while F-actin has the unique hydration water, the mobility of which is close to that of bulk water rather than the typical hydration water around proteins.
 -synuclein studied by quasielastic neutron scattering
-synuclein studied by quasielastic neutron scattering藤原 悟; 荒木 克哉*; 松尾 龍人; 八木 寿梓*; 山田 武*; 柴田 薫; 望月 秀樹*
PLOS ONE (Internet), 11(4), p.e0151447_1 - e0151447_17, 2016/04
被引用回数:24 パーセンタイル:64.99(Multidisciplinary Sciences)Filamentous aggregates (amyloid fibrils) of the protein  -synuclein (
-synuclein ( -Syn) are related to the pathogenesis of Parkinson's disease. To understand the pathogenesis mechanism of this disease, the mechanism of the amyloid fibril formation of
-Syn) are related to the pathogenesis of Parkinson's disease. To understand the pathogenesis mechanism of this disease, the mechanism of the amyloid fibril formation of  -Syn must be elucidated. As a first step toward this ultimate goal, dynamical behavior of
-Syn must be elucidated. As a first step toward this ultimate goal, dynamical behavior of  -Syn in the monomeric and the fibril states was investigated using quasielastic neutron scattering (QENS). Analysis of the QENS spectra of solution samples of
-Syn in the monomeric and the fibril states was investigated using quasielastic neutron scattering (QENS). Analysis of the QENS spectra of solution samples of  -Syn shows that diffusive global motions are observed in the monomeric state but largely suppressed in the fibril state. However, the amplitude of the side chain motion is shown to be larger in the fibril state than in the monomeric state. This implies that significant solvent space exists within the fibrils, which is attributed to the
-Syn shows that diffusive global motions are observed in the monomeric state but largely suppressed in the fibril state. However, the amplitude of the side chain motion is shown to be larger in the fibril state than in the monomeric state. This implies that significant solvent space exists within the fibrils, which is attributed to the  -Syn molecules within the fibrils having a distribution of conformations. The larger amplitude of the side chain motion in the fibril state than in the monomeric state implies that the fibril state is entropically favorable.
-Syn molecules within the fibrils having a distribution of conformations. The larger amplitude of the side chain motion in the fibril state than in the monomeric state implies that the fibril state is entropically favorable.
茶竹 俊行*; 藤原 悟
Acta Crystallographica Section D; Structural Biology (Internet), 72(1), p.71 - 82, 2016/01
被引用回数:5 パーセンタイル:37.86(Biochemical Research Methods)A technique for the determination of the deuterium/hydrogen (D/H) contrast map in neutron macromolecular crystallography has been developed and evaluated using ribonuclease A. In this technique, the contrast map between the D O-solvent and H
O-solvent and H O-solvent crystals is calculated using subtraction in real space. The present technique can thus utilize all the amplitudes of the neutron structure factors for both of the D
O-solvent crystals is calculated using subtraction in real space. The present technique can thus utilize all the amplitudes of the neutron structure factors for both of the D O-solvent and H
O-solvent and H O solvent crystals. The neutron D/H contrast maps clearly demonstrate powerful detectability of the H/D exchange in proteins. In fact, alternative protonation states and alternative conformations of hydroxyl groups are observed at a medium resolution (1.8
O solvent crystals. The neutron D/H contrast maps clearly demonstrate powerful detectability of the H/D exchange in proteins. In fact, alternative protonation states and alternative conformations of hydroxyl groups are observed at a medium resolution (1.8  ). Moreover, water molecules can be categorized into three types according to their tendency for rotational disorder. These results directly indicate improvement in the neutron crystal structure analysis. Combination of this technique with the conventional neutron structure determination protocols thus makes more precise and efficient determination of the D atom positions in proteins possible.
). Moreover, water molecules can be categorized into three types according to their tendency for rotational disorder. These results directly indicate improvement in the neutron crystal structure analysis. Combination of this technique with the conventional neutron structure determination protocols thus makes more precise and efficient determination of the D atom positions in proteins possible.
松尾 龍人; 武田 壮一*; 小田 俊郎*; 藤原 悟
Biophysics and Physicobiology (Internet), 12, p.145 - 158, 2015/12
Troponin (Tn), consisting of three subunits, TnC, TnI, and TnT, is a protein that plays a major role in regulation of muscle contraction. Various mutations of Tn cause familial hypertrophic cardiomyopathy. Here we focus on the mutations E244D and K247R of TnT, which induce an increase in the maximum tension of cardiac muscle without changes in Ca -sensitivity, and carried out small-angle X-ray scattering experiments on the Tn core domain containing the wild type subunits and those containing the mutant TnT in the absence and presence of Ca
-sensitivity, and carried out small-angle X-ray scattering experiments on the Tn core domain containing the wild type subunits and those containing the mutant TnT in the absence and presence of Ca . Changes in the overall shape induced by the mutations were detected for the first time by the changes in the radius of gyration and the maximum dimension between the wild type and the mutants. Analysis by model calculations shows that TnC adopts a dumbbell structure regardless of the mutations, and that the mutations change the distributions of the conformational ensembles so that the flexible N- and C-terminal regions of TnT become close to the center of the whole molecule.
. Changes in the overall shape induced by the mutations were detected for the first time by the changes in the radius of gyration and the maximum dimension between the wild type and the mutants. Analysis by model calculations shows that TnC adopts a dumbbell structure regardless of the mutations, and that the mutations change the distributions of the conformational ensembles so that the flexible N- and C-terminal regions of TnT become close to the center of the whole molecule.
松尾 龍人; 荒田 敏昭*; 小田 俊郎*; 中島 健次; 河村 聖子; 菊地 龍弥; 藤原 悟
Biochemical and Biophysical Research Communications, 459(3), p.493 - 497, 2015/04
被引用回数:4 パーセンタイル:12.91(Biochemistry & Molecular Biology)Various biological functions related to cell motility are driven by the interaction between the partner proteins, actin and myosin. To obtain insights into how this interaction occurs, the internal dynamics of F-actin and myosin subfragment-1 (S1) were characterized by quasielastic neutron scattering measurements on the solution samples of F-actin and S1. Contributions of the internal motions of the proteins to the scattering spectra were separated from those of the global macromolecular diffusion. Analysis of the spectra arising from the internal dynamics showed that the correlation times of the atomic motions were about two times shorter for F-actin than for S1, suggesting that F-actin fluctuates more rapidly than S1. It was also shown that the fraction of the immobile atoms is larger for S1 than for F-actin. These results suggest that F-actin actively facilitates the binding of myosin by utilizing the more frequent conformational fluctuations than those of S1.
田代 孝二*; 塙坂 真*; 山元 博子*; Wasanasuk, K.*; Jayaratri, P.*; 吉澤 功徳*; 田中 伊知朗*; 新村 信雄*; 日下 勝弘*; 細谷 孝明*; et al.
高分子論文集, 71(11), p.508 - 526, 2014/11
被引用回数:6 パーセンタイル:22.37(Polymer Science)高分子結晶構造の詳細を、水素原子位置まで含めて明らかにすることを目的とし、高エネルギーX線および中性子回折データの収集ならびにそれらの解析結果を、さまざまの結晶性高分子を例として総合的に記述した。まず、最近にまで至る高分子構造解析手法の発展について概要を述べるとともに、それらの各段階における問題点について考察した。斜方晶型ポリエチレン、アタクティックポリビニルアルコール、ポリ乳酸およびそのステレオコンプレックスなど、いろいろの意味で重要な高分子について、これまでに提案されてきた構造を再吟味するとともに、新たに提案した構造について記述した。水素原子位置についても精確に決定された場合は、それらの構造情報に基づく極限力学物性の定量的予測を行った。さらにはポリジアセチレンの場合について、X線および中性子構造解析によって得られた精密な電子密度分布および原子位置座標の情報にいわゆるX-N法を適用し、主鎖骨格に沿った結合電子密度分布についての導出についても言及した。構造物性相関解明における高分子結晶構造解析の今後の展開についても言及した。
松尾 龍人; 荒田 敏昭*; 小田 俊郎*; 藤原 悟
Biophysics, 9, p.99 - 106, 2013/07
Hydration structures around F-actin and myosin subfragment-1 (S1), which play central roles as counterparts in muscle contraction, were investigated by small-angle X-ray scattering (SAXS) and small-angle neutron scattering (SANS). The radius of gyration of S1 was evaluated to be 41.3  1.1
1.1  for SAXS, 40.1
for SAXS, 40.1  3.0
3.0  for SANS in H
for SANS in H O, and 37.8
O, and 37.8  0.8
0.8  for SANS in D
for SANS in D O, respectively. The values of the cross-sectional radius of gyration of F-actin were 25.4
O, respectively. The values of the cross-sectional radius of gyration of F-actin were 25.4  0.03
0.03  for SAXS, 23.4
for SAXS, 23.4  2.4
2.4  for SANS in H
for SANS in H O, and 22.6
O, and 22.6  0.6
0.6  for SANS in D
for SANS in D O, respectively. Analysis showed that the hydration shell of S1 has the average density 10-15% higher than bulk water, being the typical hydration shell, while the hydration shell of F-actin has the average density more than 19% higher than bulk water, indicating that F-actin has a denser, unusual hydration structure. The results indicate a difference in the hydration structures around F-actin and S1.
O, respectively. Analysis showed that the hydration shell of S1 has the average density 10-15% higher than bulk water, being the typical hydration shell, while the hydration shell of F-actin has the average density more than 19% higher than bulk water, indicating that F-actin has a denser, unusual hydration structure. The results indicate a difference in the hydration structures around F-actin and S1.
藤原 悟; Plazanet, M.*; 小田 俊郎*
Biochemical and Biophysical Research Communications, 431(3), p.542 - 546, 2013/02
被引用回数:4 パーセンタイル:11.19(Biochemistry & Molecular Biology)Quasi-elastic neutron scattering experiments of powder samples of F-actin and G-actin, hydrated either with D O or H
O or H O, at hydration ratios of 0.4 and 1.0, were performed. Combined analysis of the quasi-elastic neutron scattering spectra provided the parameters characterizing the water dynamics in the first hydration layer and those outside of the first layer. The translational diffusion coefficients (D
O, at hydration ratios of 0.4 and 1.0, were performed. Combined analysis of the quasi-elastic neutron scattering spectra provided the parameters characterizing the water dynamics in the first hydration layer and those outside of the first layer. The translational diffusion coefficients (D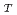 ) of the hydration water in the first layer were found to be 1.2
) of the hydration water in the first layer were found to be 1.2  10
10 cm
cm /s and 1.7
/s and 1.7  10
10 cm
cm /s for F-actin and G-actin, respectively, while that for bulk water was 2.8
/s for F-actin and G-actin, respectively, while that for bulk water was 2.8  10
10 cm
cm /s. The residence times were 6.6 ps and 5.0 ps for F-actin and G-actin, respectively, while that for bulk water was 0.63 ps. These differences between F-actin and G-actin, indicating that the hydration water around G-actin is more mobile than that around F-actin, are in concert with the results of the internal dynamics of F-actin and G-actin, showing that G-actin fluctuates more rapidly than F-actin. This implies that the dynamics of the hydration water is coupled to the internal dynamics of the actin molecules. The D
/s. The residence times were 6.6 ps and 5.0 ps for F-actin and G-actin, respectively, while that for bulk water was 0.63 ps. These differences between F-actin and G-actin, indicating that the hydration water around G-actin is more mobile than that around F-actin, are in concert with the results of the internal dynamics of F-actin and G-actin, showing that G-actin fluctuates more rapidly than F-actin. This implies that the dynamics of the hydration water is coupled to the internal dynamics of the actin molecules. The D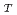 values of the water molecules outside of the first hydration layer were found to be similar to that of bulk water though the residence times are strongly affected by the first hydration layer. This supports the recent observation on intracellular water that shows bulk-like behavior.
values of the water molecules outside of the first hydration layer were found to be similar to that of bulk water though the residence times are strongly affected by the first hydration layer. This supports the recent observation on intracellular water that shows bulk-like behavior.
藤原 悟; 中川 洋
波紋, 23(1), p.72 - 80, 2013/02
中性子非弾性散乱(準弾性散乱及び非干渉性弾性散乱を含む)は、ピコ秒時間領域の蛋白質ダイナミクスを直接測定できるという特徴を持つ。この時間領域の蛋白質ダイナミクスは蛋白質の機能発現に不可欠であるため、蛋白質機能の真の理解のためには、蛋白質ダイナミクスの解明は不可欠である。中性子非弾性散乱による蛋白質ダイナミクス研究には、「モデル」蛋白質を用いた蛋白質ダイナミクスの物理的基盤を探る研究と、より生物学的な興味に基づいた研究との2つの方向性がある。本解説では、この2つの方向性の研究のそれぞれについて、具体的例を挙げながら概観し、その有用性を示すとともに、中性子非弾性散乱を用いた蛋白質ダイナミクス研究の将来について簡単に議論する。
松尾 龍人; Natali, F.*; Plazanet, M.*; Zaccai, G.*; 藤原 悟
Journal of the Physical Society of Japan, 82(Suppl.A), p.SA020_1 - SA020_5, 2013/01
被引用回数:3 パーセンタイル:27.27(Physics, Multidisciplinary)トロポニンと呼ばれる蛋白質は、心筋の収縮を細胞内カルシウム濃度依存的に調節する。また、トロポニン分子内に起こるさまざまな変異が、肥大型心筋症等の疾患に繋がることが知られている。変異によってトロポニン分子の機能に異常が起こり、疾患へと繋がる。蛋白質が機能するためには、それを構成する原子の熱揺らぎ(ダイナミクス)が必須であると考えられているため、変異によるトロポニンの機能変化がダイナミクスの違いに起因する可能性が考えられる。そこで、変異によるトロポニンのダイナミクス変化を検出するために、野生型及び変異型(K247R)ヒト心筋トロポニンの溶液試料を用いて中性子散乱実験を行った。実験はフランスILLのIN13分光器を用いて行い、280Kから292Kの温度範囲を3Kごとに変化させて弾性散乱強度を測定した。得られたデータから見かけのバネ定数(蛋白質の柔らかさの指標)を計算すると、0.077N/m(野生型), 0.046N/m(変異型)となった。この結果は、変異によりトロポニン分子全体の柔軟性が増大することを示している。分子全体が過度に柔軟になることで、トロポニンのカルシウムシグナル伝達機構が正常に機能しなくなると考えられる。
中川 洋; 藤原 悟
波紋, 22(3), p.268 - 273, 2012/08
Proteins are constantly fluctuating under the influence of environment. These thermal fluctuations, or dynamics, of the proteins at a picosecond time scale are known to be indispensable for functions of the proteins. Neutron inelastic scattering provides a unique tool to directly measure dynamics of the proteins. In particular, since the incoherent neutron scattering cross section of hydrogen is significantly larger than any other atoms commonly found in biological materials, the techniques of incoherent neutron scattering is important for the measurements of the dynamics of the protein, a half of the atoms in which are hydrogen atoms. Furthermore, since a distribution of hydrogen atoms is fairly homogeneous in the protein, the neutron scattering spectra arising from hydrogen atoms in the proteins provides a global view of the protein dynamics. Here, various aspects of three techniques of incoherent neutron scattering: elastic incoherent neutron scattering, quasi-elastic neutron scattering, and inelastic neutron scattering, are briefly described as a basis for application to characterization of the protein dynamics.
 at the MLF, J-PARC
at the MLF, J-PARC高橋 伸明; 柴田 薫; 川北 至信; 中島 健次; 稲村 泰弘; 中谷 健; 中川 洋; 藤原 悟; 佐藤 卓*; 筑紫 格*; et al.
Journal of the Physical Society of Japan, 80(Suppl.B), p.SB007_1 - SB007_4, 2011/12
被引用回数:6 パーセンタイル:43.42(Physics, Multidisciplinary)A TOF-BSS named  has been under construction on BL02 in the MLF of J-PARC. We have estimated expected performances of several candidates under realistic neutron source parameters at MLF. The expected neutron intensity under comparable energy resolutions of the
has been under construction on BL02 in the MLF of J-PARC. We have estimated expected performances of several candidates under realistic neutron source parameters at MLF. The expected neutron intensity under comparable energy resolutions of the  -type is 2.6 times higher than that of the BASIS-type. Consequently, we have chosen the CM with pulse-shaping device for
-type is 2.6 times higher than that of the BASIS-type. Consequently, we have chosen the CM with pulse-shaping device for 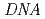 . Pulse-shaping is a good technique from a view point of a variability of resolution. On the other hand, a neutron energy band passing through the pulse-shaping chopper is limited and thus scanning range with one phase of the chopper is narrow. Of course,
. Pulse-shaping is a good technique from a view point of a variability of resolution. On the other hand, a neutron energy band passing through the pulse-shaping chopper is limited and thus scanning range with one phase of the chopper is narrow. Of course, 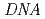 also can access larger energy transfers by appropriate phasing of the pulse-shaping chopper. In addition,
also can access larger energy transfers by appropriate phasing of the pulse-shaping chopper. In addition, 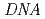 will be able to utilize Repetition Rate Multiplication (RRM). RRM is essentially a way to employ multiple pulse-shaped incident neutron beams to effectively increase neutron counting time to more efficiently measure the inelastic region. In this presentation we will show the chopper sequence and introduce the RRM mode of the forthcoming backscattering spectrometer
will be able to utilize Repetition Rate Multiplication (RRM). RRM is essentially a way to employ multiple pulse-shaped incident neutron beams to effectively increase neutron counting time to more efficiently measure the inelastic region. In this presentation we will show the chopper sequence and introduce the RRM mode of the forthcoming backscattering spectrometer 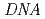 in detail.
in detail.