Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:60.46(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 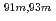 Mo and
Mo and 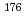 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Akiyama, Kazuhiko; Nishinaka, Ichiro; Hirata, Masaru; Yaita, Tsuyoshi; Ichikawa, Shinichi; Nagame, Yuichiro; et al.
Journal of the American Chemical Society, 126(16), p.5219 - 5224, 2004/04
Times Cited Count:44 Percentile:72.41(Chemistry, Multidisciplinary)Fluoride complexation of element 104, rutherfordium (Rf), produced in the  Cm(
Cm( O,5n)
O,5n) Rf reaction has been studied by anion-exchange chromatography on an atom-at-a-time scale. The anion-exchangechromatographic behavior of Rf was investigated in 1.9-13.9 M hydrofluoric acid together with those of the group-4 elements Zr and Hf produced in the
Rf reaction has been studied by anion-exchange chromatography on an atom-at-a-time scale. The anion-exchangechromatographic behavior of Rf was investigated in 1.9-13.9 M hydrofluoric acid together with those of the group-4 elements Zr and Hf produced in the  O-induced reactions on Ge and Gd targets, respectively. It was found that the adsorption behavior of Rf on anion-exchange resin is quite different from those of Zr and Hf, suggesting the influence of relativistic effect on the fluoride complexation of Rf.
O-induced reactions on Ge and Gd targets, respectively. It was found that the adsorption behavior of Rf on anion-exchange resin is quite different from those of Zr and Hf, suggesting the influence of relativistic effect on the fluoride complexation of Rf.
Toyoshima, Atsushi; Miyashita, Sunao*; Oe, Kazuhiro*; Kitayama, Yuta*; Lerum, H. V.*; Goto, Naoya*; Kaneya, Yusuke; Komori, Yukiko*; Mitsukai, Akina*; Vascon, A.; et al.
no journal, ,
no abstracts in English
 SO
SO into toluene with Aliquat336 as model experiments for seaborgium (Sg)
into toluene with Aliquat336 as model experiments for seaborgium (Sg)Mitsukai, Akina; Toyoshima, Atsushi; Kaneya, Yusuke; Oe, Kazuhiro*; Sato, Daisuke*; Goto, Naoya*; Tsuto, Shohei*; Komori, Yukiko*; Murakami, Masashi*; Haba, Hiromitsu*; et al.
no journal, ,
We report on the extraction behavior of carrier-free radioisotopes  Mo and
Mo and  W, which are lighter homologs of Sg, produced at the RIKEN K70 AVF cyclotron, in the H
W, which are lighter homologs of Sg, produced at the RIKEN K70 AVF cyclotron, in the H SO
SO -Aliquat336 system. Results of the extraction experiments showed that the distribution ratios,
-Aliquat336 system. Results of the extraction experiments showed that the distribution ratios,  , of Mo and W decrease up to
, of Mo and W decrease up to  3 M as increasing [H
3 M as increasing [H SO
SO ]. This is probably due to protonation reactions of hydrolyzed Mo and W species. In
]. This is probably due to protonation reactions of hydrolyzed Mo and W species. In 
 3 M H
3 M H SO
SO , the
, the  values of W and Mo showed sharp increases. Based on the slope analysis separately carried out, it was suggested that hydrolyzed species of [HMO
values of W and Mo showed sharp increases. Based on the slope analysis separately carried out, it was suggested that hydrolyzed species of [HMO ]
] (M = Mo and W) are extracted in less than
(M = Mo and W) are extracted in less than  3 M [H
3 M [H SO
SO ], while anionic sulphate complexes of [MO
], while anionic sulphate complexes of [MO (SO
(SO )
) ]
] are formed in more than [H
are formed in more than [H SO
SO ]
]
 5 M.
5 M.
 SO
SO with Aliquat336 as homologues of seaborgium (Sg)
with Aliquat336 as homologues of seaborgium (Sg)Mitsukai, Akina; Toyoshima, Atsushi; Asai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Kaneya, Yusuke; Takeda, Shinsaku*; Nagame, Yuichiro; Komori, Yukiko*; Murakami, Masashi*; et al.
no journal, ,
We have started studying sulphate-complex formation of a transactinide element, seaborgium (Sg). In this study, we report on the extraction behavior of carrier-free radioisotopes  Mo and
Mo and  W which are lighter homologs of Sg, from aqueous H
W which are lighter homologs of Sg, from aqueous H SO
SO solution with amine extractant, Aliquat336, dissolved in toluene by a batch method. These radioisotopes were produced in the
solution with amine extractant, Aliquat336, dissolved in toluene by a batch method. These radioisotopes were produced in the 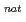 Zr(
Zr( ,
,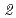 )
) Mo and
Mo and  Ta(
Ta(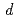 ,x
,x )
) W reactions at the RIKEN K70 AVF cyclotron. Results of the extraction experiments showed that the distribution ratios of Mo and W increase sharply above ~3.0 M H
W reactions at the RIKEN K70 AVF cyclotron. Results of the extraction experiments showed that the distribution ratios of Mo and W increase sharply above ~3.0 M H SO
SO . Based on the slope analysis, it was indicated that anionic sulphate-complex of [MO
. Based on the slope analysis, it was indicated that anionic sulphate-complex of [MO (SO
(SO )
) ]
] (M = Mo, W) are formed in
(M = Mo, W) are formed in  5 M H
5 M H SO
SO . These results suggest that the present system is applicable to the extraction of Sg.
. These results suggest that the present system is applicable to the extraction of Sg.
Toyoshima, Atsushi; Oe, Kazuhiro*; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Kaneko, Masashi*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Due to short half-lives less than 10 s and extremely low production rates, transactinide elements heavier than seaborgium (Sg) are produced on an atom per hour scale. Therefore, a continuous rapid chemistry assembly is required to study aqueous-phase chemistry of these heaviest elements. In the present study, we started developments of a continuous chemistry assembly. Our first attempt was made in on-line experiments with Mo and W, lighter homologs of Sg, to optimize a chemistry assembly consisting of a newly developed membrane degasser as an interface between gas-jet and aqueous phase, a flow electrolytic column apparatus utilized to control oxidation states of Mo and W ions, and the continuous liquid-liquid extraction apparatus of SISAK for separation. In the conference, present status of the developments will be presented.
Toyoshima, Atsushi; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Towards electrolytic reduction of Sg, batch-wise electrolytic reduction of carrier-free  Mo and
Mo and 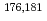 W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.
W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.  Mo and
Mo and 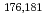 W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.