Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Honda, Mitsunori; Goto, Takuya*; Sakanaka, Yoshihide*; Yaita, Tsuyoshi; Suzuki, Shinichi*
AIMS Electronics and Electrical Engineering (Internet), 3(2), p.102 - 110, 2019/03
The possibility of removal and controlling crystal formation from weathered biotite (WB) in clay minerals were investigated using molten salt electrochemistry (EC) in molten NaCl-CaCl under an electrochemical reductive reaction. Cyclic Voltammogram (CV) measurements were performed in the range of +0.5 V to -2.2 V. Several peaks were confirmed in the CV spectra. The peak at -1.4 V represents a reduction reaction of Fe in WB, so we conducted an experiment at -1.4 V for 2 h to reduce Iron (Fe). The Cs removal rate after EC treatment was determined by X-ray fluorescence analysis, and almost 100% Cs removal was confirmed. To understand the effect of the reductive reaction, we performed X-ray Adsorption Fine Structure (XAFS) analysis. Before EC treatment, the Fe in WB was present as a mixture of Fe
under an electrochemical reductive reaction. Cyclic Voltammogram (CV) measurements were performed in the range of +0.5 V to -2.2 V. Several peaks were confirmed in the CV spectra. The peak at -1.4 V represents a reduction reaction of Fe in WB, so we conducted an experiment at -1.4 V for 2 h to reduce Iron (Fe). The Cs removal rate after EC treatment was determined by X-ray fluorescence analysis, and almost 100% Cs removal was confirmed. To understand the effect of the reductive reaction, we performed X-ray Adsorption Fine Structure (XAFS) analysis. Before EC treatment, the Fe in WB was present as a mixture of Fe and Fe
and Fe . After EC treatment, the presence of Fe
. After EC treatment, the presence of Fe was confirmed by XAFS analysis. Based on this finding, EC treatment is effective for reducing Fe in WB. This result indicated that Fe
was confirmed by XAFS analysis. Based on this finding, EC treatment is effective for reducing Fe in WB. This result indicated that Fe O
O formation was suppressed, and the reduction reaction was effective for controlling crystal formation.
formation was suppressed, and the reduction reaction was effective for controlling crystal formation.
 -rays at the ANNRI in the MLF of the J-PARC
-rays at the ANNRI in the MLF of the J-PARCTakada, Shusuke*; Okudaira, Takuya*; Goto, Fumiya*; Hirota, Katsuya*; Kimura, Atsushi; Kitaguchi, Masaaki*; Koga, Jun*; Nakao, Taro*; Sakai, Kenji; Shimizu, Hirohiko*; et al.
Journal of Instrumentation (Internet), 13(2), p.P02018_1 - P02018_21, 2018/02
Times Cited Count:6 Percentile:31.02(Instruments & Instrumentation)Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:64.63(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 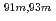 Mo and
Mo and 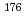 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
Kasada, Ryuta*; Goto, Takuya*; Fujioka, Shinsuke*; Hiwatari, Ryoji*; Oyama, Naoyuki; Tanigawa, Hiroyasu; Miyazawa, Junichi*; Young Scientists Special Interest Group on Fusion Reactor Realization*
Purazuma, Kaku Yugo Gakkai-Shi, 89(4), p.193 - 198, 2013/04
Japanese young researchers who have interest in realizing fusion reactor have analyzed Technology Readiness Levels (TRL) in Young Scientists Special Interest Group on Fusion Reactor Realization. In this report, brief introduction to TRL assessment and a view of TRL assessment against fusion reactor projects conducting in Japan.
 2 surface
2 surfaceYamamoto, Yoshihisa*; Togashi, Hideaki*; Kato, Atsushi*; Hasegawa, Satoshi*; Goto, Seiichi*; Nakano, Takuya*; Suemitsu, Maki*; Narita, Yuzuru*; Yoshigoe, Akitaka; Teraoka, Yuden
no journal, ,
We have investigated the initial oxidation process on an Si(110)-16 2 surface by real time synchrotron radiation photoemission spectroscopy (SR-XPS). As a result, a rapid initial oxidation regime was found to exist on this surface, which is not present on other crystal orientations. Analyses of the O1s spectra suggest insertion of oxygen atoms at the Si-Si bonds as the predominant process during initial oxidation. The rapid initial oxidation is accompanied by decrease of the Si2p subpeak that is related to Si(110)-16
2 surface by real time synchrotron radiation photoemission spectroscopy (SR-XPS). As a result, a rapid initial oxidation regime was found to exist on this surface, which is not present on other crystal orientations. Analyses of the O1s spectra suggest insertion of oxygen atoms at the Si-Si bonds as the predominant process during initial oxidation. The rapid initial oxidation is accompanied by decrease of the Si2p subpeak that is related to Si(110)-16 2 reconstruction, indicating the preferential oxidation at the pentagon pair.
2 reconstruction, indicating the preferential oxidation at the pentagon pair.
 2 surface
2 surfaceTogashi, Hideaki*; Yamamoto, Yoshihisa*; Goto, Seiichi*; Takahashi, Yuya*; Nakano, Takuya*; Kato, Atsushi*; Hasegawa, Satoshi*; Konno, Atsushi*; Suemitsu, Maki*; Asaoka, Hidehito; et al.
no journal, ,
no abstracts in English
 2
2Nakano, Takuya*; Hasegawa, Satoshi*; Togashi, Hideaki*; Kato, Atsushi*; Goto, Seiichi*; Teraoka, Yuden; Yoshigoe, Akitaka; Suemitsu, Maki*
no journal, ,
no abstracts in English
 2 surface and surface reconstruction
2 surface and surface reconstructionTogashi, Hideaki*; Yamamoto, Yoshihisa*; Goto, Seiichi*; Takahashi, Yuya*; Nakano, Takuya*; Konno, Atsushi*; Suemitsu, Maki*; Asaoka, Hidehito; Yoshigoe, Akitaka; Teraoka, Yuden
no journal, ,
Initial oxidaion processes at Si(110)-16 2 clean surface with O
2 clean surface with O mokecules have been observed by real-time SR-XPS and STM methods. SR-XPS experiments were performed at BL23SU in SPring-8 and STM experiments were performed in JAEA Tokai research center. From comparison of oxygen uptake curves of Si(110)-16
mokecules have been observed by real-time SR-XPS and STM methods. SR-XPS experiments were performed at BL23SU in SPring-8 and STM experiments were performed in JAEA Tokai research center. From comparison of oxygen uptake curves of Si(110)-16 2 with Si(001)-2
2 with Si(001)-2 1, following conclusions were obtained. Rapid initial oxidation occured at the Si(110) surface and layer-by-layer oxidation was also took place. In-situ observation with STM revealed that the rapid initial oxidation was due to oxygen adsorption at pentagon-pair Si atoms on the topmost surface, at least 4 kinds of oxygen-adsorbed states, and the DD site was a condensed oxidation state.
1, following conclusions were obtained. Rapid initial oxidation occured at the Si(110) surface and layer-by-layer oxidation was also took place. In-situ observation with STM revealed that the rapid initial oxidation was due to oxygen adsorption at pentagon-pair Si atoms on the topmost surface, at least 4 kinds of oxygen-adsorbed states, and the DD site was a condensed oxidation state.
 2 surface observed by SR-PES
2 surface observed by SR-PESYamamoto, Yoshihisa*; Togashi, Hideaki*; Kato, Atsushi*; Hasegawa, Satoshi*; Nakano, Takuya*; Goto, Seiichi*; Teraoka, Yuden; Yoshigoe, Akitaka; Suemitsu, Maki*
no journal, ,
In this study, we have investigated adsorption of oxygen molecules on Si(110)-16 2 clean surface at room temperature by synchrotron radiation photoemission spectros (SR-PES). The SR-PES experiments were conducted at the surface chemistry end-station of BL23SU in the SPring-8. The photon energy was 334 eV. Samples were B-doped p-type Si(110) wafers, and were flash-annealed in an UHV ambient prior to the exposure to oxygen gas at room temperature at the gas pressure of 6.7
2 clean surface at room temperature by synchrotron radiation photoemission spectros (SR-PES). The SR-PES experiments were conducted at the surface chemistry end-station of BL23SU in the SPring-8. The photon energy was 334 eV. Samples were B-doped p-type Si(110) wafers, and were flash-annealed in an UHV ambient prior to the exposure to oxygen gas at room temperature at the gas pressure of 6.7 10
10 Pa. By observing a structural change after a mild annealing, we have confirmed presence of a metastable state of oxidation on this RT-oxidized Si(110) surface.
Pa. By observing a structural change after a mild annealing, we have confirmed presence of a metastable state of oxidation on this RT-oxidized Si(110) surface.
 2 surface using SR-PES and STM
2 surface using SR-PES and STMYamamoto, Yoshihisa*; Togashi, Hideaki*; Goto, Seiichi*; Takahashi, Yuya*; Nakano, Takuya*; Kato, Atsushi*; Hasegawa, Satoshi*; Konno, Atsushi*; Suemitsu, Maki*; Yoshigoe, Akitaka; et al.
no journal, ,
We have investigated that initial oxidation processes of Si(110) surfaces at room temperature and thermal stability of the oxide layer via synchrotron radiation photoemission spectroscopy (SR-PES) and scanning tunneling microscopy (STM). We found in the SR-PES experiments that metastable oxygen-condensed local structures were formed in the early stage of Si(110) oxidation at room temperature. After thermal annealing of 15 min at 573 K, highly-oxidized components of Si2p spectrum, Si and Si
and Si , were enhanced whereas Si
, were enhanced whereas Si was decreased. This is due to structural relaxation of the strained metastable oxidation structures on the surface and following change to stable highly-oxidized structures. Furthermore, the structural change from the metastable structure in the surface oxidized at room temperature to the stable cluster structure was directly observed via STM.
was decreased. This is due to structural relaxation of the strained metastable oxidation structures on the surface and following change to stable highly-oxidized structures. Furthermore, the structural change from the metastable structure in the surface oxidized at room temperature to the stable cluster structure was directly observed via STM.
 2 surface by Si 2p photoemission spectroscopy
2 surface by Si 2p photoemission spectroscopyYamamoto, Yoshihisa*; Togashi, Hideaki*; Kato, Atsushi*; Hasegawa, Satoshi*; Goto, Seiichi*; Nakano, Takuya*; Suemitsu, Maki*; Narita, Yuzuru*; Yoshigoe, Akitaka; Teraoka, Yuden
no journal, ,
Initial oxidation process of Si(110)-16 2 clean surface has been investigated with synchrotron radiation photoemission spectroscopy. We found that the intensity of the alpha peak, one of the surface core level shifts in the Si 2p photoemission spectra, decreased in the just early stage of oxidation. And it was possible to relate this component with the rapid initial oxidation, which was unique to Si(110) surface. According to recent studies, this component was attributed to the 1st and the 2nd Si layer. We also found that there were significant amount of Si
2 clean surface has been investigated with synchrotron radiation photoemission spectroscopy. We found that the intensity of the alpha peak, one of the surface core level shifts in the Si 2p photoemission spectra, decreased in the just early stage of oxidation. And it was possible to relate this component with the rapid initial oxidation, which was unique to Si(110) surface. According to recent studies, this component was attributed to the 1st and the 2nd Si layer. We also found that there were significant amount of Si component just after introducing the oxygen gas, which was contrast to the oxidation of Si(001) surface.
component just after introducing the oxygen gas, which was contrast to the oxidation of Si(001) surface.
Togashi, Hideaki*; Yamamoto, Yoshihisa*; Goto, Seiichi*; Takahashi, Yuya*; Nakano, Takuya*; Konno, Atsushi*; Suemitsu, Maki*; Asaoka, Hidehito; Yoshigoe, Akitaka; Teraoka, Yuden
no journal, ,
Oxygen-adsorbed Si(110)-16 2 surfaces at room temperature and their thermally-treated surfaces were analyzed by real-time photoemission spectroscopy with synchrotron radiation (SR-PES) and scanning tunneling microscopy (STM). Thermal stability of the oxygen-adsorbed Si(110) surfaces was clearified. By thermal annealing of Si(110) surface exposed to 10L oxygen gas at room temperature, photoemission peak positions of Si
2 surfaces at room temperature and their thermally-treated surfaces were analyzed by real-time photoemission spectroscopy with synchrotron radiation (SR-PES) and scanning tunneling microscopy (STM). Thermal stability of the oxygen-adsorbed Si(110) surfaces was clearified. By thermal annealing of Si(110) surface exposed to 10L oxygen gas at room temperature, photoemission peak positions of Si and Si
and Si components were shifted to higher energy side by 0.20eV and 0.12eV,respectively, and reached nearly to those of thermal oxide films. This behaviour is due to change of Si-O-Si bond angle and Si-O bond length. The STM observation for Si(110) surfaces revealed that the oxygen-adsorbed surface structure changed to the condensed oxide structure by thermal annealing. These SR-PES and STM results showed that the oxygen-adsorbed Si(110)-16
components were shifted to higher energy side by 0.20eV and 0.12eV,respectively, and reached nearly to those of thermal oxide films. This behaviour is due to change of Si-O-Si bond angle and Si-O bond length. The STM observation for Si(110) surfaces revealed that the oxygen-adsorbed surface structure changed to the condensed oxide structure by thermal annealing. These SR-PES and STM results showed that the oxygen-adsorbed Si(110)-16 2 structure was metastable and changed to more stable condensed oxide structure by thermal annealing.
2 structure was metastable and changed to more stable condensed oxide structure by thermal annealing.
Goto, Yukari*; Nakagawa, Takuya*; Makino, Yuko*; Yamada, Satoshi*; Yamada, Mina*; Sakoda, Akihiro; Ishimori, Yuu
no journal, ,
Tottori University and Japan Atomic Energy Agency started a joint study to develop an environmental remediation technique for agricultural soil. Nine plants were water-cultured and examined for screening. A few were selected as candidates for demonstrations in fields. Preselected plants were mainly halophytes. Easily cultivated and harvested plants without harmful effect on new agriculture were also considered. Seedings prepared were first grown for a certain term. Additive-free,  Cs and
Cs and  Sr groups, which are both stable isotopes, were then made. Stems, leaves and roots were harvested, in principle, two weeks after the addition, to measure K, Ca, Mg, Sr and Cs concentrations in them. It was concluded that New Zealand spinach and ice plant were most adequate for removing contaminants from surface soil. The two accumulate Cs and Sr mostly in the shoots, are prostrate, and spread the roots shallowly. For valid application, growth-phase dependences of absorption and distribution, growth property and root distribution should be elucidated. Finally, the application study plan was developed based on screening test results.
Sr groups, which are both stable isotopes, were then made. Stems, leaves and roots were harvested, in principle, two weeks after the addition, to measure K, Ca, Mg, Sr and Cs concentrations in them. It was concluded that New Zealand spinach and ice plant were most adequate for removing contaminants from surface soil. The two accumulate Cs and Sr mostly in the shoots, are prostrate, and spread the roots shallowly. For valid application, growth-phase dependences of absorption and distribution, growth property and root distribution should be elucidated. Finally, the application study plan was developed based on screening test results.
Toyoshima, Atsushi; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Towards electrolytic reduction of Sg, batch-wise electrolytic reduction of carrier-free  Mo and
Mo and 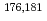 W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.
W radiotracers was studied using a flow electrolytic column (FEC). The electrolyzed samples from a FEC were chemically analyzed by solvent extraction with TOA and HDEHP to separate and identify reduced species from the stable Mo(VI) and W(VI) ones based on their different extraction behavior.  Mo and
Mo and 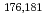 W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
W were applied as radiotracers. We also performed cyclic voltammetry and UV/Vis absorption spectrometry of macro amounts of Mo and W in acidic solutions to obtain information on redox reactions of these elements under given conditions. In the conference, the present status of the preparatory reduction experiments with Mo and W will be presented.
Toyoshima, Atsushi; Miyashita, Sunao*; Oe, Kazuhiro*; Kitayama, Yuta*; Lerum, H. V.*; Goto, Naoya*; Kaneya, Yusuke; Komori, Yukiko*; Mitsukai, Akina*; Vascon, A.; et al.
no journal, ,
no abstracts in English
Toyoshima, Atsushi; Oe, Kazuhiro*; Asai, Masato; Attallah, M. F.*; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Kaneko, Masashi*; Kaneya, Yusuke; Kasamatsu, Yoshitaka*; et al.
no journal, ,
Due to short half-lives less than 10 s and extremely low production rates, transactinide elements heavier than seaborgium (Sg) are produced on an atom per hour scale. Therefore, a continuous rapid chemistry assembly is required to study aqueous-phase chemistry of these heaviest elements. In the present study, we started developments of a continuous chemistry assembly. Our first attempt was made in on-line experiments with Mo and W, lighter homologs of Sg, to optimize a chemistry assembly consisting of a newly developed membrane degasser as an interface between gas-jet and aqueous phase, a flow electrolytic column apparatus utilized to control oxidation states of Mo and W ions, and the continuous liquid-liquid extraction apparatus of SISAK for separation. In the conference, present status of the developments will be presented.
Honda, Mitsunori; Goto, Takuya*; Sakanaka, Yoshihide*; Shimoyama, Iwao; Okamoto, Yoshihiro; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
Honda, Mitsunori; Goto, Takuya*; Sakanaka, Yoshihide*; Shimoyama, Iwao; Okamoto, Yoshihiro; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
Honda, Mitsunori; Goto, Takuya*; Sakanaka, Yoshihide*; Shimoyama, Iwao; Okamoto, Yoshihiro; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English
Honda, Mitsunori; Goto, Takuya*; Sakanaka, Yoshihide*; Shimoyama, Iwao; Okamoto, Yoshihiro; Suzuki, Shinichi; Yaita, Tsuyoshi
no journal, ,
no abstracts in English