Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Khuyagbaatar, J.*; Yakushev, A.*; D llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
Physical Review C, 102(6), p.064602_1 - 064602_9, 2020/12
Times Cited Count:78 Percentile:99.01(Physics, Nuclear)A search for production of the superheavy elements with atomic numbers 119 and 120 was performed in the 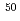 Ti+
Ti+ Bk and
Bk and 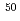 Ti+
Ti+ Cf fusion-evaporation reactions, respectively, at the gas-filled recoil separator TASCA. Over four months of irradiation, neither was detected at cross-section sensitivity levels of 65 and 200 fb, respectively. The non-observation of elements 119 and 120 is discussed within the concept of fusion-evaporation reactions including various theoretical predictions on the fission-barrier heights of superheavy nuclei in the region of the island of stability.
Cf fusion-evaporation reactions, respectively, at the gas-filled recoil separator TASCA. Over four months of irradiation, neither was detected at cross-section sensitivity levels of 65 and 200 fb, respectively. The non-observation of elements 119 and 120 is discussed within the concept of fusion-evaporation reactions including various theoretical predictions on the fission-barrier heights of superheavy nuclei in the region of the island of stability.
 Ca+
Ca+ Bk leading to formation of the element Ts (Z=117)
Bk leading to formation of the element Ts (Z=117)Khuyagbaatar, J.*; Yakushev, A.*; D llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
Physical Review C, 99(5), p.054306_1 - 054306_16, 2019/05
Times Cited Count:33 Percentile:92.64(Physics, Nuclear)We have performed an experiment to synthesize the element 117 (Ts) with the  Ca+
Ca+ Bk fusion reaction. Four
Bk fusion reaction. Four  -decay chains attributed to the element 117 were observed. Two of them were long decay chains which can be assigned to the one originating from the
-decay chains attributed to the element 117 were observed. Two of them were long decay chains which can be assigned to the one originating from the  decay of
decay of 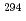 Ts. The other two were short decay chains which are consistent with the one originating from the
Ts. The other two were short decay chains which are consistent with the one originating from the  decay of
decay of 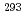 Ts. We have compared the present results with the literature data, and found that our present results mostly confirmed the literature data, leading to the firm confirmation of the synthesis of the element 117.
Ts. We have compared the present results with the literature data, and found that our present results mostly confirmed the literature data, leading to the firm confirmation of the synthesis of the element 117.
Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:71.15(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 and W(CO)
and W(CO)
Usoltsev, I.*; Eichler, R.*; Wang, Y.*; Even, J.*; Yakushev, A.*; Haba, Hiromitsu*; Asai, Masato; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; et al.
llmann, Ch. E.*; et al.
Radiochimica Acta, 104(3), p.141 - 151, 2016/03
Times Cited Count:34 Percentile:94.30(Chemistry, Inorganic & Nuclear)Conditions of the production and decomposition of hexacarbonyl complexes of short-lived Mo and W isotopes were investigated to study thermal stability of the heaviest group 6 hexacarbonyl complex Sg(CO) . A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO)
. A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO) and W(CO)
and W(CO) , indicating that the first bond dissociation energy of Sg(CO)
, indicating that the first bond dissociation energy of Sg(CO) could be determined with this technique.
could be determined with this technique.
Even, J.*; Ackermann, D.*; Asai, Masato; Block, M.*; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(3), p.2457 - 2466, 2015/03
Times Cited Count:15 Percentile:73.95(Chemistry, Analytical)Rapid In situ synthesis of metal carbonyl complexes has been demonstrated using short-lived isotopes produced in nuclear fission and fusion reactions. The short-lived isotopes with high recoil energy directly react with carbon-monoxides and form carbonyl complexes. Only highly volatile complexes were fast transported in a gas stream to counting and chemistry devices. Short-lived Mo, Tc, Ru, Rh, W, Re, Os, and Ir were found to form volatile carbonyl complexes, while no volataile complex of Hf and Ta were detected. This technique has been applied to a chemical investigation of the superheavy element Sg (atomic number 106), and will be applicable to various fields of nuclear science with short-lived transition metal isotopes.
Even, J.*; Yakushev, A.*; D llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
Science, 345(6203), p.1491 - 1493, 2014/09
Times Cited Count:67 Percentile:82.96(Multidisciplinary Sciences)A new superheavy element complex, a seaborgium carbonyl, has been successfully synthesized, and its adsorption property has been studied using a cryo-thermochromatography and  -detection apparatus COMPACT. Nuclear reaction products of short-lived
-detection apparatus COMPACT. Nuclear reaction products of short-lived  Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of
Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of 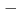 50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO)
50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO) . This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
. This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
 Ca +
Ca +  Bk fusion reaction leading to element Z = 117; Long-lived
Bk fusion reaction leading to element Z = 117; Long-lived  -decaying
-decaying 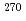 Db and discovery of
Db and discovery of  Lr
LrKhuyagbaatar, J.*; Yakushev, A.*; D llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Asai, Masato; Block, M.*; Boll, R. A.*; Brand, H.*; Cox, D. M.*; et al.
Physical Review Letters, 112(17), p.172501_1 - 172501_5, 2014/05
Times Cited Count:232 Percentile:98.35(Physics, Multidisciplinary)The superheavy element with atomic number 117 was produced in the  Ca +
Ca +  Bk fusion reaction using the gas-filled recoil separator TASCA at GSI in Germany. This result verified the previous result of the discovery of new element 117 reported by Flerov Laboratory of Nuclear Reactions in Russia, which makes certain the synthesis and discovery of element 117 in human history. On the other hand, the last nucleus in the
Bk fusion reaction using the gas-filled recoil separator TASCA at GSI in Germany. This result verified the previous result of the discovery of new element 117 reported by Flerov Laboratory of Nuclear Reactions in Russia, which makes certain the synthesis and discovery of element 117 in human history. On the other hand, the last nucleus in the  decay chain from the element 117 was assigned to be the unknown nucleus
decay chain from the element 117 was assigned to be the unknown nucleus  Lr instead of the previously reported
Lr instead of the previously reported 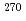 Db, and
Db, and 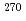 Db was found to be the
Db was found to be the  -decaying nucleus with very long half-life.
-decaying nucleus with very long half-life.
Yakushev, A.*; Gates, J. M.*; T rler, A.*; Sch
rler, A.*; Sch del, M.; D
del, M.; D llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Block, M.*; Br
llmann, Ch. E.*; Ackermann, D.*; Andersson, L.-L.*; Block, M.*; Br chle, W.*; Dvorak, J.*; et al.
chle, W.*; Dvorak, J.*; et al.
Inorganic Chemistry, 53(3), p.1624 - 1629, 2014/02
Times Cited Count:113 Percentile:98.88(Chemistry, Inorganic & Nuclear)We report on a gas-solid chromatography study of the adsorption of element 114 (flerovium, Fl) on a Au surface. Fl was produced in the nuclear fusion reaction  Pu(
Pu( Ca, 3-4n)
Ca, 3-4n)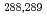 Fl and was isolated in-flight from the primary
Fl and was isolated in-flight from the primary  beam in a physical recoil separator. The adsorption behavior of Fl, its nuclear
beam in a physical recoil separator. The adsorption behavior of Fl, its nuclear  -decay product Cn, their lighter homologues in groups 14 and 12, i.e., Pb and Hg, and the noble gas Rn were studied simultaneously by isothermal gas chromatography and thermochromatography. Two Fl atoms were detected. They adsorbed on a Au surface at room temperature, but not as readily as Pb and Hg. The observed adsorption behavior of Fl points to a higher inertness compared to its nearest homologue in the group, Pb. However, the measured lower limit for the adsorption enthalpy of Fl on a Au surface points to the formation of a metal-metal bond of Fl with Au. Fl is the least reactive element in the group, but still a metal.
-decay product Cn, their lighter homologues in groups 14 and 12, i.e., Pb and Hg, and the noble gas Rn were studied simultaneously by isothermal gas chromatography and thermochromatography. Two Fl atoms were detected. They adsorbed on a Au surface at room temperature, but not as readily as Pb and Hg. The observed adsorption behavior of Fl points to a higher inertness compared to its nearest homologue in the group, Pb. However, the measured lower limit for the adsorption enthalpy of Fl on a Au surface points to the formation of a metal-metal bond of Fl with Au. Fl is the least reactive element in the group, but still a metal.
Br chle, W.*; Andrassy, M.*; Angert, R.*; Eberhardt, K.*; Fricke, B.*; Gregorich, K. E.*; G
chle, W.*; Andrassy, M.*; Angert, R.*; Eberhardt, K.*; Fricke, B.*; Gregorich, K. E.*; G nther, R.*; Hartmann, W.*; Heimann, R.*; Hoffman, D. C.*; et al.
nther, R.*; Hartmann, W.*; Heimann, R.*; Hoffman, D. C.*; et al.
1st International Conference on the Chemistry and Physics of the Transactinide Elements; Extended Abstracts, 4 Pages, 1999/00
no abstracts in English