Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 O in Fe
O in Fe O
O film on Fe formed in an NaOH solution containing oxidants
film on Fe formed in an NaOH solution containing oxidantsHaruna, Takumi*; Miyataki, Yuki*; Hirohata, Yohei*; Shibata, Toshio*; Taniguchi, Naoki; Tachikawa, Hirokazu*
Zairyo To Kankyo, 67(9), p.375 - 380, 2018/09
This research aimed to confirm the formation of Fe O
O film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D
film on Fe immersed in aqueous 45 mass% NaOH solution containing some oxidants at the boiling temperature, to recognize the optimum immersion time for the formation of thick and protective film, and to reveal the absorption behavior of D O in the Fe
O in the Fe O
O film at room temperature. The results were obtained as follows. It was confirmed that Fe
film at room temperature. The results were obtained as follows. It was confirmed that Fe O
O film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D
film was formed on Fe immersed in the NaOH solution for a time more than 0.6 ks, and the film thickness increased parabolically with an increase in the immersion time. D O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D
O absorption test was carried out to the films formed in the NaOH solution for immersion times of 1.2 and 3.6 ks. An amount of D O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D
O absorbed into the film increased with an increase in an absorption time up to 1000 ks, and an absorption time more than 1000 ks made an amount of D O constant. The constant amount of D
O constant. The constant amount of D O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D
O was larger for the film formed on Fe immersed in the NaOH solution for 3.6 ks than that for 1.2 ks. The transient of the amount of D O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D
O absorbed into the film was analyzed on the basis of Fick's law for diffusion, and diffusion coefficients of D O were obtained to be 5.1
O were obtained to be 5.1 10
10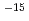 cm
cm s
s and 9.9
and 9.9 10
10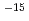 cm
cm s
s for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe
for the films formed for 1.2 and 3.6 ks, respectively. Therefore it was estimated that the diffusion coefficient of the Fe O
O film was in the region from 5.1
film was in the region from 5.1 10
10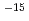 cm
cm s
s to 9.9
to 9.9 10
10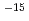 cm
cm s
s .
.
 O in the film on Fe oxidized at high temperature in air
O in the film on Fe oxidized at high temperature in airHaruna, Takumi*; Yamamoto, Tatsuya*; Miyairi, Yoji*; Shibata, Toshio*; Taniguchi, Naoki; Sakamaki, Keiko; Tachikawa, Hirokazu*
Zairyo To Kankyo, 64(5), p.201 - 206, 2015/05
Diffusion coefficients of D O in the films was determined in order to estimate corrosion rate of carbon steel for the overpack in ground water. Fe plates were heated to form oxide films. The films were characterized with XRD and SEM. After that, the specimen was contacted with D
O in the films was determined in order to estimate corrosion rate of carbon steel for the overpack in ground water. Fe plates were heated to form oxide films. The films were characterized with XRD and SEM. After that, the specimen was contacted with D O for 5184 ks, followed by subjected to TDS to obtain an amount of D
O for 5184 ks, followed by subjected to TDS to obtain an amount of D O absorbing into the film. As a result, single-layered film of Fe
O absorbing into the film. As a result, single-layered film of Fe O
O was formed at 573 and 723 K, and double-layered film of Fe
was formed at 573 and 723 K, and double-layered film of Fe O
O and Fe
and Fe O
O was formed at 873 K. It was found that an amount of D
was formed at 873 K. It was found that an amount of D O in the film correlated linearly with a square root of the absorption period, and that the amount was steady for a long period. From the results and Fick's second law, diffusion coefficients of D
O in the film correlated linearly with a square root of the absorption period, and that the amount was steady for a long period. From the results and Fick's second law, diffusion coefficients of D O was determined as 9.7
O was determined as 9.7 10
10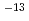 cm
cm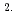 s
s for the Fe
for the Fe O
O film, and 5.5
film, and 5.5 10
10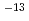 cm
cm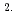 s
s to 2.2
to 2.2 10
10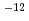 cm
cm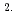 s
s for Fe
for Fe O
O film.
film.
Shibata, Toshio*; Haruna, Takumi*; Fujimoto, Shinji*; Zhang, S.*
JAERI-Tech 2000-061, 38 Pages, 2000/09
no abstracts in English
Haruna, Takumi*; Miyataki, Yuki*; Hirohata, Yohei*; Taniguchi, Naoki; Tachikawa, Hirokazu*
no journal, ,
no abstracts in English
Kai, Itsuki*; Nakanishi, Yuki*; Taniguchi, Naoki; Dobashi, Ryuta*; Hirohata, Yohei*; Haruna, Takumi*
no journal, ,
no abstracts in English