Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kawasaki, Takuro; Fukuda, Tatsuo; Yamanaka, Satoru*; Murayama, Ichiro*; Kato, Takanori*; Baba, Masaaki*; Hashimoto, Hideki*; Harjo, S.; Aizawa, Kazuya; Tanaka, Hirohisa*; et al.
Journal of Applied Physics, 137(9), p.094101_1 - 094101_7, 2025/03
Times Cited Count:0 Percentile:0.00(Physics, Applied)Murase, Kiyoka*; Kataoka, Ryuho*; Nishiyama, Takanori*; Nishimura, Koji*; Hashimoto, Taishi*; Tanaka, Yoshimasa*; Kadokura, Akira*; Tomikawa, Yoshihiro*; Tsutsumi, Masaki*; Ogawa, Yasunobu*; et al.
Journal of Space Weather and Space Climate (Internet), 12, p.18_1 - 18_16, 2022/06
Times Cited Count:3 Percentile:28.94(Astronomy & Astrophysics)We identified two energetic electron precipitation (EEP) events during the growth phase of moderate substorms and estimated the mesospheric ionization rate for an EEP event for which the most comprehensive dataset from ground-based and space-born instruments was available. The mesospheric ionization signature reached below 70 km altitude and continued for ~15 min until the substorm onset, as observed by the PANSY radar and imaging riometer at Syowa Station in the Antarctic region. We also used energetic electron flux observed by the Arase and POES 15 satellites as the input for the air-shower simulation code PHITS to quantitatively estimate the mesospheric ionization rate. Combining the cutting-edge observations and simulations, we shed new light on the space weather impact of the EEP events during geomagnetically quiet times, which is important to understand the possible link between the space environment and climate.
 structure observation of pyroelectric ceramics during power generation cycle
structure observation of pyroelectric ceramics during power generation cycleKawasaki, Takuro; Fukuda, Tatsuo; Yamanaka, Satoru*; Sakamoto, Tomokazu*; Murayama, Ichiro*; Kato, Takanori*; Baba, Masaaki*; Hashimoto, Hideki*; Harjo, S.; Aizawa, Kazuya; et al.
Journal of Applied Physics, 131(13), p.134103_1 - 134103_7, 2022/04
Times Cited Count:2 Percentile:15.59(Physics, Applied)Kim, J.*; Yamanaka, Satoru*; Murayama, Ichiro*; Kato, Takanori*; Sakamoto, Tomokazu*; Kawasaki, Takuro; Fukuda, Tatsuo; Sekino, Toru*; Nakayama, Tadachika*; Takeda, Masatoshi*; et al.
Sustainable Energy & Fuels (Internet), 4(3), p.1143 - 1149, 2020/03
Times Cited Count:22 Percentile:63.45(Chemistry, Physical) Cs + ICs' (Contract research)
Cs + ICs' (Contract research)Kobayashi, Takanori; Hashimoto, Masashi; Yokoyama, Keiichi
JAEA-Research 2015-014, 7 Pages, 2015/12
To discuss the exchange reaction of Cs isotope by CsI + Cs' Cs + ICs', the structure and chemical properties of Cs
Cs + ICs', the structure and chemical properties of Cs I intermediate and potential energy surface are calculated using M06/def2-TZVPPD density functional calculation. The calculation shows that the reaction to the intermediate has no barrier and the two Cs-I bonds of Cs
I intermediate and potential energy surface are calculated using M06/def2-TZVPPD density functional calculation. The calculation shows that the reaction to the intermediate has no barrier and the two Cs-I bonds of Cs I are chemically equivalent. Thus, the collision of CsI + Cs' results in Cs exchange with the high probability.
I are chemically equivalent. Thus, the collision of CsI + Cs' results in Cs exchange with the high probability.
 Li
LiJeong, S.-C.*; Katayama, Ichiro*; Kawakami, Hirokane*; Watanabe, Yutaka*; Ishiyama, Hironobu*; Imai, Nobuaki*; Hirayama, Yoshikazu*; Miyatake, Hiroari; Sataka, Masao; Sugai, Hiroyuki; et al.
Japanese Journal of Applied Physics, 47(8), p.6413 - 6415, 2008/08
Times Cited Count:5 Percentile:22.08(Physics, Applied)A non-destructive and on-line diffusion tracing in Li ionic conductors has been demonstrated. As a tracer, pulsed beam of  Li was implanted into LiGa. By analyzing the time dependent yield of the
Li was implanted into LiGa. By analyzing the time dependent yield of the  -particles decaying from
-particles decaying from  Li, diffusion coefficients were extracted with a high accuracy. The ordering of Li vacancies in the Li-deficient
Li, diffusion coefficients were extracted with a high accuracy. The ordering of Li vacancies in the Li-deficient  phase of LiGa was observed for the first time in terms of the Li diffusion.
phase of LiGa was observed for the first time in terms of the Li diffusion.
 Li short-lived radiotracer in Li ionic conductors of NaTl-type intermetallic compounds
Li short-lived radiotracer in Li ionic conductors of NaTl-type intermetallic compoundsSugai, Hiroyuki; Sataka, Masao; Okayasu, Satoru; Ichikawa, Shinichi; Nishio, Katsuhisa; Mitsuoka, Shinichi; Nakanoya, Takamitsu; Osa, Akihiko; Sato, Tetsuya; Hashimoto, Takashi; et al.
Defect and Diffusion Forum, 273-276, p.667 - 672, 2008/00
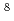 Li
LiJeong, S.-C.*; Katayama, Ichiro*; Kawakami, Hirokane*; Watanabe, Yutaka*; Ishiyama, Hironobu*; Imai, Nobuaki*; Hirayama, Yoshikazu*; Miyatake, Hiroari; Sataka, Masao; Okayasu, Satoru; et al.
Journal of Phase Equilibria and Diffusion, 26(5), p.472 - 476, 2005/09
Authors developed a radiotracer method for diffusion studies in solids. The experimental test was performed by the measurement of the diffusion coefficients of Li in a sample of the compound  LiAl using an
LiAl using an  -emitting radiotracer of
-emitting radiotracer of 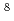 Li(T
Li(T = 0.84 s). It was found that the time-dependent yields of the alpha particles from the diffusing 8Li that was initially implanted in the sample could be used as a measure of the diffusivity of the tracer in a nondestructive way. The method was applied to measure the self-diffusion coefficients of Li in
= 0.84 s). It was found that the time-dependent yields of the alpha particles from the diffusing 8Li that was initially implanted in the sample could be used as a measure of the diffusivity of the tracer in a nondestructive way. The method was applied to measure the self-diffusion coefficients of Li in  LiGa, and for investigating how the Li diffusion in the Li ionic conductors is affected by the concentration of atomic defects.
LiGa, and for investigating how the Li diffusion in the Li ionic conductors is affected by the concentration of atomic defects.
 Li
LiJeong, S.-C.*; Katayama, Ichiro*; Kawakami, Hirokane*; Watanabe, Yutaka*; Ishiyama, Hironobu*; Miyatake, Hiroari*; Sataka, Masao; Okayasu, Satoru; Sugai, Hiroyuki; Ichikawa, Shinichi; et al.
Nuclear Instruments and Methods in Physics Research B, 230(1-4), p.596 - 600, 2005/04
Times Cited Count:6 Percentile:44.85(Instruments & Instrumentation)no abstracts in English
Yamashita, Shinichiro; Otsuka, Satoshi; Watanabe, Masashi*; Uchida, Yosuke*; Suda, Takanori*; Hashimoto, Naoyuki*; Onuki, Somei*; Shibayama, Tamaki*
no journal, ,
no abstracts in English
Uchida, Yosuke*; Nagai, Yoshiyasu*; Suda, Takanori*; Hashimoto, Naoyuki*; Onuki, Somei*; Shibayama, Tamaki*; Yamashita, Shinichiro; Akasaka, Naoaki
no journal, ,
no abstracts in English
 CsI + Cs
CsI + CsKobayashi, Takanori; Hashimoto, Masashi; Yokoyama, Keiichi
no journal, ,
To investigate the possibility of the Cs exchange reaction, we have analyzed the Cs-I-Cs triatomic molecule by using quantum chemistry calculation. Furthermore, by using the newly developed capture model, the Cs exchange reaction rate and its temperature dependence were estimated. 2-dimensional potential energy surface around CsI against Cs atom shows that the reaction has no entrance barrier. The reaction rate was found to be close to the collision rate and to have a little temperature dependence would be expected.
 Li short-lived radiotracer
Li short-lived radiotracerSugai, Hiroyuki; Sataka, Masao; Okayasu, Satoru; Ichikawa, Shinichi; Nishio, Katsuhisa; Mitsuoka, Shinichi; Nakanoya, Takamitsu; Osa, Akihiko; Sato, Tetsuya; Hashimoto, Takashi; et al.
no journal, ,
Non-destructive and on-line Li diffusion experiments in Li ionic conductors are conducted using the short-lived  -emitting radiotracer of
-emitting radiotracer of  Li. The radiotracers produced as an energetic and pulsed ion beam from TRIAC (Tokai Radioactive Ion Accelerator Complex) are implanted into a structural defect mediated Li ionic conductor of NaTl-type intermetallic compounds
Li. The radiotracers produced as an energetic and pulsed ion beam from TRIAC (Tokai Radioactive Ion Accelerator Complex) are implanted into a structural defect mediated Li ionic conductor of NaTl-type intermetallic compounds  -LiGa. The diffusion coefficients obtained for
-LiGa. The diffusion coefficients obtained for  -LiGa with Li content of 43-54 at.% at room temperature are discussed in terms of the interaction between Li-ion and the structural defects in the specimen, compared with the case of
-LiGa with Li content of 43-54 at.% at room temperature are discussed in terms of the interaction between Li-ion and the structural defects in the specimen, compared with the case of  -LiAl and
-LiAl and  -LiIn. The nonlinear Li-content dependency of Li diffusion coefficients for
-LiIn. The nonlinear Li-content dependency of Li diffusion coefficients for  -LiGa suggests that the Li diffusion with Li-deficient region is obstructed by the defect complex composed of vacancies at the Li sites.
-LiGa suggests that the Li diffusion with Li-deficient region is obstructed by the defect complex composed of vacancies at the Li sites.
Kobayashi, Takanori; Hashimoto, Masashi; Yokoyama, Keiichi
no journal, ,
To investigate the possibility of the Cs exchange reaction between CsI molecule and Cs atom, we have analyzed the Cs-I-Cs triatomic molecule by using quantum chemistry calculation. And, by using the newly developed capture model, the Cs exchange reaction rate and its temperature dependence were also estimated. The stabilization energy for CsICs formation is calculated as 12 kcal mol , which is much smaller than the calculated Cs-I bond energy of 82 kcal mol-1(spin-orbit correlation is not included). However, the bond lengths of both Cs-I are optimized as the same value (3.57 A). That is, Cs of CsI and colliding Cs atom cannot be distinguished if the triatomic structure is formed. And 2-dimensional potential energy surface around CsI against Cs atom shows that the reaction has no entrance barrier. The reaction rate was estimated to be the same value as the collision rate and little temperature dependence would be expected.
, which is much smaller than the calculated Cs-I bond energy of 82 kcal mol-1(spin-orbit correlation is not included). However, the bond lengths of both Cs-I are optimized as the same value (3.57 A). That is, Cs of CsI and colliding Cs atom cannot be distinguished if the triatomic structure is formed. And 2-dimensional potential energy surface around CsI against Cs atom shows that the reaction has no entrance barrier. The reaction rate was estimated to be the same value as the collision rate and little temperature dependence would be expected.
Ito, Yoshiyuki; Matsushima, Ryotatsu; Sato, Fuminori; Hashimoto, Takanori*; Suzuki, Tatsuya*; Miyabe, Shinsuke*; Sakuma, Takashi*; Kikuchi, Kaoru*; Izumi, Takeshi*
no journal, ,
no abstracts in English