Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Shinohara, Masanori; Sumita, Junya; Shibata, Taiju; Hirata, Masaru
JAEA-Evaluation 2022-006, 198 Pages, 2022/11
Japan Atomic Energy Agency (JAEA) received a post-evaluation of the third medium-/long-term plan (from FY2015 to FY2021) and pre-evaluation of the fourth medium-/long-term plan (from FY2022 to FY2028) from the "Evaluation Committee of Research Activities for High Temperature Gas-cooled Reactor (hereinafter referred to as "HTGR") and Related Hydrogen Production Technology" (hereinafter referred to as "Evaluation Committee") which consists of specialists in the fields of evaluation subjects of HTGR and related heat application technologies. As a result, for the post-evaluation of the third medium-/ long-term plan, two of the ten technical committee members concluded a score of "S", seven members concluded "A" and one member concluded "B". The comprehensive evaluation concluded a score of "A". On the other hand, one of the two humanities and social sciences members concluded a score of "B", one members concluded "C". The comprehensive evaluation concluded a score of "B". For the pre-evaluation of the fourth medium-/long-term plan, although there were some items that several evaluation committee members rated as "needs improvement," the majority of the committee members judged the plan to be appropriate. This report describes the members of the Evaluation Committee, assessment items, assessment results and JAEA's measures following the assessment.
Shinohara, Masanori; Sumita, Junya; Shibata, Taiju; Hirata, Masaru
JAEA-Evaluation 2022-001, 104 Pages, 2022/06
Japan Atomic Energy Agency received the annual evaluation of FY2020, the research plan of FY2021 and the prospective evaluation of the third mid- to long-term plan (from FY2015 to FY2021) from the "Evaluation Committee of Research Activities for High Temperature Gas-cooled Reactor (hereinafter referred to as "HTGR") and Related Hydrogen Production Technology" (hereinafter referred to as "Evaluation Committee") which consists of specialists in the fields of evaluation subjects of HTGR and related heat application technology. As a result, for the annual evaluation of FY2020, one of the ten technical committee members concluded a score of "S", eight members concluded "A" and one member concluded "B". The comprehensive evaluation was concluded a score of "A". On the other hand, two humanities and social sciences committee members concluded "B". For the prospective evaluation of the third mid- to long-term plan, one of the ten technical committee members concluded a score of "S", eight members concluded "A" and one member concluded "B". The comprehensive evaluation was concluded a score of "A". On the other hand, two humanities and social sciences committee members concluded "B". This report describes the members of the Evaluation Committee, assessment items, assessment results and JAEA's measures following the assessment.
Suzuki, Chikashi; Nishi, Tsuyoshi; Nakada, Masami; Tsuru, Tomohito; Akabori, Mitsuo; Hirata, Masaru; Kaji, Yoshiyuki
Journal of Physics and Chemistry of Solids, 74(12), p.1769 - 1774, 2013/12
Times Cited Count:14 Percentile:51.30(Chemistry, Multidisciplinary)We investigated the electronic state of a CaF -type (Am,U) mixed oxide using the all-electron full potential linear augmented plane wave method and compared it with those of Am
-type (Am,U) mixed oxide using the all-electron full potential linear augmented plane wave method and compared it with those of Am O
O , AmO
, AmO , UO
, UO , and La
, and La U
U O
O . The valence of Am in the mixed oxide was close to that of Am
. The valence of Am in the mixed oxide was close to that of Am O
O and the valence of U in the mixed oxide was pentavalent. The electronic structure of AmO
and the valence of U in the mixed oxide was pentavalent. The electronic structure of AmO was different from that of Am
was different from that of Am O
O , particularly just above the Fermi level. In addition, the electronic states of Am and U in the mixed oxide were similar to those of trivalent Am and pentavalent U oxides. These electronic states reflected the high oxygen potential of AmO
, particularly just above the Fermi level. In addition, the electronic states of Am and U in the mixed oxide were similar to those of trivalent Am and pentavalent U oxides. These electronic states reflected the high oxygen potential of AmO and the heightened oxygen potential resulting from the addition of Am to UO
and the heightened oxygen potential resulting from the addition of Am to UO and also suggested the occurrence of charge transfer from Am to U in the solid solution process.
and also suggested the occurrence of charge transfer from Am to U in the solid solution process.
Komatsu, Mitsuru*; Nishigaki, Makoto*; Seno, Shoji*; Hirata, Yoichi*; Takenobu, Kazuyoshi*; Tagishi, Hirotaka*; Kunimaru, Takanori; Maekawa, Keisuke; Yamamoto, Yoichi; Toida, Masaru*; et al.
JAEA-Research 2012-001, 77 Pages, 2012/09
This research focused on methods of estimating the amounts of groundwater recharge, which are normally required as upper boundary conditions in groundwater flow analyses, based on measurements of infiltrating water in the ground, to systematize the methods and establish systems which are stable and measurable on site over a long time. Regarding developing measurement systems, fiber-optic strain measurement methods that enable multiple-point and long-distance measurement were used for measuring three quantities: suction pressure, soil moisture and volumetric water content rate obtained by applying water absorption swelling material, and each measurement was discussed. The results showed that the fiber-optic soil aquameter has two types of practical application: one for measuring suction pressure (Type I), and the other for measuring volumetric water content rate obtained by applying water absorption swelling material (Type III). Furthermore, by using measurement instruments in actual fields, the validity of the two methods for estimating the rainfall infiltration capacities of shallow-layer soils, that is, estimating the capacities either directly by measurements of soil water or from unsaturated hydraulic conductivities, was confirmed.
Shibata, Hiroki; Tsuru, Tomohito; Nishi, Tsuyoshi; Hirata, Masaru; Kaji, Yoshiyuki
Journal of Nuclear Science and Technology, 49(3), p.328 - 333, 2012/03
Times Cited Count:8 Percentile:50.20(Nuclear Science & Technology)The thermal and mechanical properties of neptunium nitride (NpN) were investigated by first principles calculations. From the Helmholtz free energy equilibrium lattice constants, thermal expansion coefficients, bulk moduli and specific heat capacities were calculated for temperatures up to 1500 K. The electronic specific heat capacity was also calculated from the electronic density of states (DOS) at the Fermi energy. The obtained specific heat capacity reproduced the experimental data well. It was thus clarified that the specific heat capacity of NpN consists of the lattice and electronic specific heat capacities and the contribution of the lattice dilatation to the specific heat capacity.
Suzuki, Chikashi; Nishi, Tsuyoshi; Nakada, Masami; Akabori, Mitsuo; Hirata, Masaru; Kaji, Yoshiyuki
Journal of Physics and Chemistry of Solids, 73(2), p.209 - 216, 2012/02
Times Cited Count:28 Percentile:68.56(Chemistry, Multidisciplinary)The authors investigated theoretically core-hole effects on X-ray absorption near-edge structures (XANES) of Np and Am L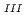 in neptunium dioxide (NpO
in neptunium dioxide (NpO ) and americium dioxide (AmO
) and americium dioxide (AmO ) with CaF
) with CaF -type crystal lattices using the all-electron full-potential linearized augmented plane-wave (FP-LAPW) method. The peak creation mechanism of XANES was shown by examining the electronic structures of these oxides, which indicated that core-hole screening was more marked for AmO
-type crystal lattices using the all-electron full-potential linearized augmented plane-wave (FP-LAPW) method. The peak creation mechanism of XANES was shown by examining the electronic structures of these oxides, which indicated that core-hole screening was more marked for AmO than for NpO
than for NpO because of the difference in the charge transfer between these oxides. Furthermore, the results of charge density analysis suggested that the white line was assigned to the quasi-bound state composed of the localized Np d or Am d components and O components, and that the tail structure was created as a result of delocalized standing waves between the Np or Am atoms.
because of the difference in the charge transfer between these oxides. Furthermore, the results of charge density analysis suggested that the white line was assigned to the quasi-bound state composed of the localized Np d or Am d components and O components, and that the tail structure was created as a result of delocalized standing waves between the Np or Am atoms.
 Am
Am )O
)O
Nishi, Tsuyoshi; Nakada, Masami; Suzuki, Chikashi; Shibata, Hiroki; Okamoto, Yoshihiro; Akabori, Mitsuo; Hirata, Masaru
Journal of Nuclear Materials, 418(1-3), p.311 - 312, 2011/11
Times Cited Count:20 Percentile:79.65(Materials Science, Multidisciplinary)The XAFS measurements at U-L and Am-L
and Am-L absorption edge of (U
absorption edge of (U Am
Am )O
)O were performed in transmission mode. Moreover, to clarify the valence state of Am in (U,Am)O
were performed in transmission mode. Moreover, to clarify the valence state of Am in (U,Am)O , the XANES spectrum of Am-L
, the XANES spectrum of Am-L absorption edge of (U
absorption edge of (U Am
Am )O
)O was verified using those of Am-L
was verified using those of Am-L absorption edge of AmO
absorption edge of AmO and Am
and Am O
O . It was found that the XANES spectrum of the Am-L
. It was found that the XANES spectrum of the Am-L edge of (U
edge of (U Am
Am )O
)O is in good accordance with that of Am
is in good accordance with that of Am O
O . Thus, Am in (U
. Thus, Am in (U Am
Am )O
)O is almost trivalent state.
is almost trivalent state.
Nagame, Yuichiro; Hirata, Masaru
Radiochimica Acta, 99(7-8), p.377 - 393, 2011/08
Times Cited Count:15 Percentile:71.85(Chemistry, Inorganic & Nuclear)Nagame, Yuichiro; Hirata, Masaru; Nakahara, Hiromichi*
Handbook of Nuclear Chemistry, p.817 - 875, 2011/02
 -uranium alloys; The Roll of d-d orbital interactions
-uranium alloys; The Roll of d-d orbital interactionsKurihara, Masayoshi*; Onoe, Jun*; Hirata, Masaru; Suzuki, Chikashi
Journal of Alloys and Compounds, 509(4), p.1152 - 1156, 2011/01
Times Cited Count:3 Percentile:23.28(Chemistry, Physical)The alloying behavior of transition metals (TMs) in solid  -phase uranium (
-phase uranium ( -U), which is expected to be used as fuel for next-generation nuclear reactors, is investigated using the discrete-variational Dirac-Fock-Slater molecular orbital method. Using a model cluster, U
-U), which is expected to be used as fuel for next-generation nuclear reactors, is investigated using the discrete-variational Dirac-Fock-Slater molecular orbital method. Using a model cluster, U /TM, as the minimum unit of
/TM, as the minimum unit of  -U/TM alloys, we evaluate the d-orbital energy of the TM (Md), the bond order between the TM and U atoms, and the orbital overlap population (OOP) between the atomic orbitals of the TM and U atoms. We subsequently examine the effect of these quantities on the maximum solid solubility (MSS) of the
-U/TM alloys, we evaluate the d-orbital energy of the TM (Md), the bond order between the TM and U atoms, and the orbital overlap population (OOP) between the atomic orbitals of the TM and U atoms. We subsequently examine the effect of these quantities on the maximum solid solubility (MSS) of the  -U/TM alloys. Md and the OOP (U 6d-TM d) exhibit good correlations with the MSS for
-U/TM alloys. Md and the OOP (U 6d-TM d) exhibit good correlations with the MSS for  -U/TM alloys, so that the interaction between the U-6d and TM-d atomic orbitals is found to play a key role in determining the MSS of the
-U/TM alloys, so that the interaction between the U-6d and TM-d atomic orbitals is found to play a key role in determining the MSS of the  -U/TM alloys. The magnitude of the MSS can be explained in terms of the stabilization energy caused by U 6d-TM d orbital interactions for
-U/TM alloys. The magnitude of the MSS can be explained in terms of the stabilization energy caused by U 6d-TM d orbital interactions for  -U/TM alloys, which is affected by the Md and the OOP. The exponential dependence of the MSS on Md and the OOP is explained in terms of the equilibrium constant obtained using a substitution cluster model for
-U/TM alloys, which is affected by the Md and the OOP. The exponential dependence of the MSS on Md and the OOP is explained in terms of the equilibrium constant obtained using a substitution cluster model for  -U/TM alloys. We also map the MSS of
-U/TM alloys. We also map the MSS of  -U/TM alloys using the Md and the OOP as the alloying parameters. These results will assist the quantum design of nuclear fuel materials for other alloy systems besides the present alloy system.
-U/TM alloys using the Md and the OOP as the alloying parameters. These results will assist the quantum design of nuclear fuel materials for other alloy systems besides the present alloy system.
 O
O and AmO
and AmO with XAFS spectroscopy
with XAFS spectroscopyNishi, Tsuyoshi; Nakada, Masami; Suzuki, Chikashi; Shibata, Hiroki; Ito, Akinori; Akabori, Mitsuo; Hirata, Masaru
Journal of Nuclear Materials, 401(1-3), p.138 - 142, 2010/06
Times Cited Count:34 Percentile:88.68(Materials Science, Multidisciplinary)XAFS studies were performed in a study of americium sesquioxide (Am O
O ) with A-type rare earth oxide structure and americium dioxide (AmO
) with A-type rare earth oxide structure and americium dioxide (AmO ) with fluorite structure. EXAFS results for Am-L
) with fluorite structure. EXAFS results for Am-L absorption edge of Am
absorption edge of Am O
O and AmO
and AmO were good agreement with the crystallographic data from X-ray diffraction analysis. In order to characterize XANES in aspect of the electronic states, the theoretical assignments for the Am
were good agreement with the crystallographic data from X-ray diffraction analysis. In order to characterize XANES in aspect of the electronic states, the theoretical assignments for the Am O
O and AmO
and AmO were performed with the all-electron full potential linearized augmented plane wave (FP-LAPW) method. The theoretical XANES spectra of Am
were performed with the all-electron full potential linearized augmented plane wave (FP-LAPW) method. The theoretical XANES spectra of Am O
O and AmO
and AmO well reproduced the experimental ones. In addition, it was found that the white line peak was created due to the interaction between Am-d and O-p components, and the broad peak and the tail peak were created due to the interaction between Am-d and O-d component.
well reproduced the experimental ones. In addition, it was found that the white line peak was created due to the interaction between Am-d and O-p components, and the broad peak and the tail peak were created due to the interaction between Am-d and O-d component.
Shibata, Hiroki; Tsuru, Tomohito; Hirata, Masaru; Kaji, Yoshiyuki
Journal of Nuclear Materials, 401(1-3), p.113 - 117, 2010/06
Times Cited Count:14 Percentile:65.92(Materials Science, Multidisciplinary)The mechanical properties of neptunium nitride (NpN) were investigated using first principles calculations based on density functional theory that take the Spin-Orbit Coupling (SOC) effect into consideration. With NaCl, CsCl and ZnS structures in nonmagnetic (NM), ferromagnetic (FM) and antiferromagnetic (AFM) states, the structure of NaCl in all magnetic states and that of ZnS, excluding the NM state without SOC, were found to be mechanically stable. The bulk moduli, elastic constants, Young's moduli, Poisson's ratios and anisotropic factors of NpN in stable states were also estimated. The enthalpies of NpN for NaCl and ZnS structures revealed that the phase transition of NpN from ZnS structure to NaCl structure took place at a pressure of -7.32 GPa, and hence the phase transition of NpN in a cubic system does not occur at positive pressure.
Hirata, Masaru
Nihon Genshiryoku Gakkai-Shi ATOMO , 51(10), P. 53, 2009/10
, 51(10), P. 53, 2009/10
The FISA2009 meeting was held in Prague from June 22 to June 25. The 450 conference participants, discussed the next generation nuclear energy systems, such as SFR, GFR etc. nuclear energy sources in Europe. In the review, I presented an outline of the important topics discussed at the conference.
Haba, Hiromitsu*; Akiyama, Kazuhiko*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Yaita, Tsuyoshi; Hirata, Masaru; Sueki, Keisuke*; Nagame, Yuichiro
Bulletin of the Chemical Society of Japan, 82(6), p.698 - 703, 2009/06
Times Cited Count:10 Percentile:39.05(Chemistry, Multidisciplinary)Chloride complexation of the group-4 elements Zr and Hf in 8.0-11.9 M HCl is investigated by Extended X-ray Absorption Fine Structure (EXAFS) spectroscopy to characterize chloro complexes of the transactinide element, rutherfordium (Rf). The complexes of Zr and Hf successively vary with the concentration of HCl from a hydrated complex [M(H O)
O)  ]
] at 8.0 M to a hexachloro complex [MCl
at 8.0 M to a hexachloro complex [MCl ]
] at 11.9 M (M = Zr and Hf). The present structural changes of the Zr and Hf complexes well reflect the previously studied anion-exchange behavior of Zr and Hf in HCl. From both the EXAFS and anion-exchange results, we suggest that Rf forms the same complexes as those of Zr and Hf in HCl, and that the complexation strength of the hexachloro complexes of the group-4 elements, [MCl
at 11.9 M (M = Zr and Hf). The present structural changes of the Zr and Hf complexes well reflect the previously studied anion-exchange behavior of Zr and Hf in HCl. From both the EXAFS and anion-exchange results, we suggest that Rf forms the same complexes as those of Zr and Hf in HCl, and that the complexation strength of the hexachloro complexes of the group-4 elements, [MCl ]
]  (M = Zr, Hf, and Rf), is in the sequence of Rf
(M = Zr, Hf, and Rf), is in the sequence of Rf  Zr
Zr  Hf.
Hf.
 and Pr
and Pr O
O for XANES analysis with redox property
for XANES analysis with redox propertySuzuki, Chikashi; Nishi, Tsuyoshi; Nakada, Masami; Akabori, Mitsuo; Hirata, Masaru; Kaji, Yoshiyuki
International Journal of Quantum Chemistry, 109(12), p.2744 - 2752, 2009/05
Times Cited Count:5 Percentile:32.11(Chemistry, Physical)no abstracts in English
Sasaki, Yuji; Kitatsuji, Yoshihiro; Hirata, Masaru; Kimura, Takaumi; Yoshizuka, Kazuharu*
Proceedings of International Solvent Extraction Conference "Solvent Extraction-Fundamentals to Industrial Applications" (ISEC 2008), p.745 - 750, 2008/09
The multidentate diamides are synthesized and examined for the actinide (An) extractions, bi- and tridentate extractants are focused in this work. The extraction of actinide was performed from 0.1-6M HNO to organic solvents. It was obvious that tetraalkyl-diglycolamide (DGA) derivatives, (methylimino)bis(N,N-dioctylacetamide) (MIDOA) and dimethyl-dioctyl-2-(3-oxapentadecane)-malonamide (DMDOOPDMA) have relatively high D values (D(Pu): more than 70). The following notable results using DGA extractants are obtained, (1) DGA with short alkyl chain give higher D values than those with long alkyl chain, (2) DGA with long alkyl chain have high solubility in n-dodecane. The molecular modeling with computational chemistry was also used to elucidate the effects of structural and electronic properties of the reagents on their different extractabilities.
to organic solvents. It was obvious that tetraalkyl-diglycolamide (DGA) derivatives, (methylimino)bis(N,N-dioctylacetamide) (MIDOA) and dimethyl-dioctyl-2-(3-oxapentadecane)-malonamide (DMDOOPDMA) have relatively high D values (D(Pu): more than 70). The following notable results using DGA extractants are obtained, (1) DGA with short alkyl chain give higher D values than those with long alkyl chain, (2) DGA with long alkyl chain have high solubility in n-dodecane. The molecular modeling with computational chemistry was also used to elucidate the effects of structural and electronic properties of the reagents on their different extractabilities.
Nishi, Tsuyoshi; Nakada, Masami; Ito, Akinori; Suzuki, Chikashi; Hirata, Masaru; Akabori, Mitsuo
Journal of Nuclear Materials, 374(3), p.339 - 343, 2008/03
Times Cited Count:24 Percentile:78.52(Materials Science, Multidisciplinary)EXAFS and XANES analysis were applied in a study of americium dioxide (AmO ) with fluorite structure. EXAFS result for Am-L
) with fluorite structure. EXAFS result for Am-L absorption edge of AmO
absorption edge of AmO was good agreement with the long-ranged structural data from X-ray diffraction analysis. In order to characterize XANES in aspect of the electronic structure, the theoretical assignment for the AmO
was good agreement with the long-ranged structural data from X-ray diffraction analysis. In order to characterize XANES in aspect of the electronic structure, the theoretical assignment for the AmO was performed with the relativistic DV-X
was performed with the relativistic DV-X molecular orbital method. The calculated XANES spectrum well reproduced the experimental spectrum. The theoretical assignment of the XANES spectra is very useful for the development of MA-MOX fuel for the future nuclear fuel cycle.
molecular orbital method. The calculated XANES spectrum well reproduced the experimental spectrum. The theoretical assignment of the XANES spectra is very useful for the development of MA-MOX fuel for the future nuclear fuel cycle.
 molecular orbital method to
molecular orbital method to  -uranium alloys with transition metals
-uranium alloys with transition metalsKurihara, Masayoshi*; Hirata, Masaru; Onoe, Jun*; Nakamatsu, Hirohide*
Progress in Nuclear Energy, 50(2-6), p.549 - 555, 2008/03
Times Cited Count:1 Percentile:9.68(Nuclear Science & Technology)The alloying behavior of 4d and 5d transition metals (TMs) in  -phase solid uranium (U) has been investigated using the discrete-variational Dirac-Fock-Slater (DV-DFS) method. We examined the d-orbital energy (Md value) of these TMs, the orbital overlap population (OOP) between the d-orbital of these TMs and the 5f or 6d orbital of
-phase solid uranium (U) has been investigated using the discrete-variational Dirac-Fock-Slater (DV-DFS) method. We examined the d-orbital energy (Md value) of these TMs, the orbital overlap population (OOP) between the d-orbital of these TMs and the 5f or 6d orbital of  -U, and the effective charge on each atom of the
-U, and the effective charge on each atom of the  -U/TM alloy, and the effect of these parameters on alloying. It was found that the maximum solid solubility of TMs into
-U/TM alloy, and the effect of these parameters on alloying. It was found that the maximum solid solubility of TMs into  -U was exponentially proportional to both the Md and the OOP (U5f-TMd and U6d-TMd). We found a good correlation between the MSS and Md, the effective charge, or OOP.
-U was exponentially proportional to both the Md and the OOP (U5f-TMd and U6d-TMd). We found a good correlation between the MSS and Md, the effective charge, or OOP.
Yamada, Hidenori*; Tamada, Taro; Kosaka, Megumi*; Miyata, Kohei*; Fujiki, Shinya*; Tano, Masaru*; Moriya, Masayuki*; Yamanishi, Mamoru*; Honjo, Eijiro; Tada, Horiko*; et al.
Protein Science, 16(7), p.1389 - 1397, 2007/07
Times Cited Count:41 Percentile:60.11(Biochemistry & Molecular Biology)In an attempt to control protein incorporation in a crystal lattice, a leucine zipper-like hydrophobic interface (comprising four leucine residues) was introduced into a helical region (helix 2) of the human pancreatic ribonuclease 1 (RNase 1) that was predicted to form a suitable crystallization interface. Although crystallization of wild type RNase 1 has not yet been reported, the RNase 1 mutant having four leucines (4L-RNase 1) was successfully crystallized under several different conditions. The crystal structures were subsequently determined by X-ray crystallography by molecular replacement using the structure of bovine RNase A. The overall structure of 4L-RNase 1 is quite similar to that of the bovine RNase A, and the introduced leucine residues formed the designed crystal interface. To further characterize the role of the introduced leucine residues in crystallization of RNase 1, the number of leucines was reduced to three or two (3L- and 2L-RNase 1, respectively). Both mutants crystallized and a similar hydrophobic interface as in 4L-RNase 1 was observed. A related approach to engineer crystal contacts at helix 3 of RNase 1 (N4L-RNase 1) was also evaluated. N4L-RNase 1 also successfully crystallized, and formed the expected hydrophobic packing interface. These results suggest that appropriate introduction of a leucine zipper-like hydrophobic interface can promote intra molecular symmetry for more efficient protein crystallization in crystal lattice engineering efforts.
Sasaki, Yuji; Rapold, P.*; Arisaka, Makoto; Hirata, Masaru; Kimura, Takaumi; Hill, C.*; Cote, G.*
Solvent Extraction and Ion Exchange, 25(2), p.187 - 204, 2007/03
Times Cited Count:154 Percentile:93.47(Chemistry, Multidisciplinary)Extraction of Eu(III) and Am(III) from HNO into the organic solvents using N,N,N',N'-tetraoctyl-diglycolamide (TODGA) was investigated in order to study the detailed extraction reaction. The chemical species: 1:2 for metal:TODGA complex is present in polar diluents. On the other hand, the metal complexes need three or more number of TODGA molecules to remain stable in non-polar diluents. The HNO
into the organic solvents using N,N,N',N'-tetraoctyl-diglycolamide (TODGA) was investigated in order to study the detailed extraction reaction. The chemical species: 1:2 for metal:TODGA complex is present in polar diluents. On the other hand, the metal complexes need three or more number of TODGA molecules to remain stable in non-polar diluents. The HNO concentration dependence on the distribution ratio suggests that HNO
concentration dependence on the distribution ratio suggests that HNO participates in the metal extraction. Infrared spectra indicate that the carbonyl oxygen coordinates with Eu(III), and luminescence lifetimes suggest that there is no water molecule in the inner coordination sphere of Eu-complex extracted.
participates in the metal extraction. Infrared spectra indicate that the carbonyl oxygen coordinates with Eu(III), and luminescence lifetimes suggest that there is no water molecule in the inner coordination sphere of Eu-complex extracted.