Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yang, S.*; Katsumura, Yosuke*; Yamashita, Shinichi*; Matsuura, Chihiro*; Hiroishi, Daisuke*; Lertnaisat, P.*; Taguchi, Mitsumasa
Radiation Physics and Chemistry, 123, p.14 - 19, 2016/06
Times Cited Count:2 Percentile:19.71(Chemistry, Physical) -radiolysis of boiling water has been investigated. The G-value of H
-radiolysis of boiling water has been investigated. The G-value of H evolution was found to be very sensitive to the purity of water. In high-purity water, both H
evolution was found to be very sensitive to the purity of water. In high-purity water, both H and O
and O gases were formed in the stoichiometric ratio of 2:1; a negligible amount of H
gases were formed in the stoichiometric ratio of 2:1; a negligible amount of H O
O remained in the liquid phase. The G-values of H
remained in the liquid phase. The G-values of H and O
and O gas evolution depend on the dose rate: lower dose rates produce larger yields. To clarify the importance of the interface between liquid and gas phase for gas evolution, the gas evolution under Ar gas bubbling was measured. A large amount of H
gas evolution depend on the dose rate: lower dose rates produce larger yields. To clarify the importance of the interface between liquid and gas phase for gas evolution, the gas evolution under Ar gas bubbling was measured. A large amount of H was detected, similar to the radiolysis of boiling water. The evolution of gas was enhanced in a 0.5 M NaCl aqueous solution. Deterministic chemical kinetics simulations elucidated the mechanism of radiolysis in boiling water.
was detected, similar to the radiolysis of boiling water. The evolution of gas was enhanced in a 0.5 M NaCl aqueous solution. Deterministic chemical kinetics simulations elucidated the mechanism of radiolysis in boiling water.
 C; Absorption spectra, kinetics and yield of carboxyl radical CO
C; Absorption spectra, kinetics and yield of carboxyl radical CO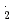

Lin, M.; Katsumura, Yosuke; Muroya, Yusa*; He, H.*; Miyazaki, Toyoaki*; Hiroishi, Daisuke*
Radiation Physics and Chemistry, 77(10-12), p.1208 - 1212, 2008/10
Times Cited Count:24 Percentile:81.47(Chemistry, Physical)Sodium formate neutral aqueous solutions saturated with N O were studied by pulse radiolysis techniques from room temperature to 400
O were studied by pulse radiolysis techniques from room temperature to 400  C. The absorption spectra of CO
C. The absorption spectra of CO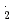
 show a red shift from
show a red shift from 
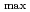 = 235 nm at 25
= 235 nm at 25  C to 280 nm at 400
C to 280 nm at 400  C. The second-order decay of CO
C. The second-order decay of CO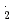
 is not monotonically temperature-dependent: the rate constant (2
is not monotonically temperature-dependent: the rate constant (2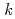 /
/ ) slightly increases with temperature up to 200
) slightly increases with temperature up to 200  C, then decreases to a valley at 300
C, then decreases to a valley at 300  C, and it sharply increases again for temperature
C, and it sharply increases again for temperature  350
350  C.
C. 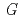

 (CO
(CO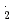
 ) increases with temperature from room temperature to 400
) increases with temperature from room temperature to 400  C. Significant density effect on
C. Significant density effect on 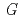

 (CO
(CO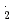
 ) is found at 400
) is found at 400  C. Assumed
C. Assumed  (CO
(CO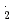
 ) is independent on temperature, at 400
) is independent on temperature, at 400  C, the density dependence of
C, the density dependence of 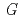 (CO
(CO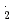
 ) in the presence of N
) in the presence of N O agrees well with that of [
O agrees well with that of [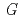 (e
(e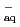 )+
)+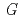 (H
(H )+
)+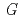 (
( OH)].
OH)].
 produced in water radiolysis
produced in water radiolysisYamashita, Shinichi; Katsumura, Yosuke; Hiroishi, Daisuke*; Murakami, Takeshi*
no journal, ,
Yield of dihydrogen molecule (H ) produced in water radiolysis is important for safety management in nuclear industry including water chemistry in nuclear fission reactors as well as for investigation of very early stages of radiation-induced phenomena, which are closely related to spatial distribution of energy deposition from charged particle to surrounding matter. A new apparatus for online measurements of H
) produced in water radiolysis is important for safety management in nuclear industry including water chemistry in nuclear fission reactors as well as for investigation of very early stages of radiation-induced phenomena, which are closely related to spatial distribution of energy deposition from charged particle to surrounding matter. A new apparatus for online measurements of H produced in water radiolysis, and the apparatus was utilized for
produced in water radiolysis, and the apparatus was utilized for 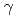 -irradiation. By improving tubing between a sample cell and thermal conductivity detector, which are settled inside and outside the irradiation room, respectively, sensitivity and accuracy of the system has much improved and resultant H
-irradiation. By improving tubing between a sample cell and thermal conductivity detector, which are settled inside and outside the irradiation room, respectively, sensitivity and accuracy of the system has much improved and resultant H yield was almost same as those reported in literatures.
yield was almost same as those reported in literatures.