Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Abe, Takeyasu; Iida, Yoshihisa; Sasamoto, Hiroshi; Ishii, Eiichi
Proceedings of Water-Rock Interaction (WRI-17)/ Applied Isotope Geochemistry (AIG-14) (Internet), 6 Pages, 2023/08
Cation exchange is one of important elementary reactions among water-rock interactions in sedimentary rocks. In order to model non-constant Na-K-Ca-Mg-H exchange selectivity of sedimentary rocks, exchange isotherms and pH dependence on cation exchange capacity of smectite were analyzed following active fraction model. As the result of comparison of exchangeable cationic composition between measured and calculated, selectivity coefficients determined in this study were preferred to those presented by previous studies. Using the coefficients and groundwater hydrochemistry reported previously, expected compositions of exchangeable cations were calculated. The results suggested that pH and total Na-K-Ca-Mg-H concentration of groundwater are important factors to interpret observed compositional variation in exchangeable base cations.
Ohira, Saki; Abe, Takeyasu; Iida, Yoshihisa
Radiochimica Acta, 111(7), p.525 - 531, 2023/07
Times Cited Count:0 Percentile:0.00(Chemistry, Inorganic & Nuclear)The solubility of 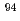 Nb in calcium alkaline solutions is one of the important parameters in safety assessment of intermediate-depth disposal which are assumed to use cementitious materials. Nb solubility and solubility-limiting solid phases of Nb in these systems remain unclear. The oversaturation solubility experiments were performed systematically in the 0.001-0.1 M CaCl
Nb in calcium alkaline solutions is one of the important parameters in safety assessment of intermediate-depth disposal which are assumed to use cementitious materials. Nb solubility and solubility-limiting solid phases of Nb in these systems remain unclear. The oversaturation solubility experiments were performed systematically in the 0.001-0.1 M CaCl solutions under alkali conditions, and the characterization of precipitated solid phase controlling Nb solubility was conducted. The negative dependence of Nb solubilities on pH and Ca concentration was observed in solubility experiments, the molar ratio of Nb to Ca of precipitated solid phase was 0.66. The pH and Ca dependence of Nb solubilities was reproduced by the reaction with Nb aqueous species Nb(OH)
solutions under alkali conditions, and the characterization of precipitated solid phase controlling Nb solubility was conducted. The negative dependence of Nb solubilities on pH and Ca concentration was observed in solubility experiments, the molar ratio of Nb to Ca of precipitated solid phase was 0.66. The pH and Ca dependence of Nb solubilities was reproduced by the reaction with Nb aqueous species Nb(OH)
 and Ca-Nb oxide with the molar ratio of Nb to Ca 0.66, e.g., Ca
and Ca-Nb oxide with the molar ratio of Nb to Ca 0.66, e.g., Ca Nb
Nb O
O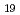 (am).
(am).
Ohira, Saki; Iida, Yoshihisa
Proceedings of Waste Management Symposia 2023 (WM2023) (Internet), 10 Pages, 2023/02
The sorption distribution coefficient ( d) of niobium-94 (Nb-94) on minerals is one of the important parameters in safety assessment of radioactive waste disposal. In a previous study, the
d) of niobium-94 (Nb-94) on minerals is one of the important parameters in safety assessment of radioactive waste disposal. In a previous study, the  d values of Nb under alkali condition in the presence of Ca, were two orders of magnitude higher than those in the presence of Na. In this study, Nb sorption experiments were performed to reexamine the effect of Ca on Nb sorption on clay minerals, and blank tests were performed to check for precipitation formation. The results showed that the Nb sorption onto montmorillonite and illite, did not depend on the Ca concentration, and
d values of Nb under alkali condition in the presence of Ca, were two orders of magnitude higher than those in the presence of Na. In this study, Nb sorption experiments were performed to reexamine the effect of Ca on Nb sorption on clay minerals, and blank tests were performed to check for precipitation formation. The results showed that the Nb sorption onto montmorillonite and illite, did not depend on the Ca concentration, and  d values obtained in the presence of Ca were the same as those in the absence of Ca. A sorption model assuming sorption by complexation on the mineral surface was developed and then calculated using the geochemical calculation code. The model with the surface species X_ONb(OH)
d values obtained in the presence of Ca were the same as those in the absence of Ca. A sorption model assuming sorption by complexation on the mineral surface was developed and then calculated using the geochemical calculation code. The model with the surface species X_ONb(OH) and X_ONb(OH)
and X_ONb(OH)
 represented trends in the data obtained.
represented trends in the data obtained.
Abe, Takeyasu; Iida, Yoshihisa
Journal of Advanced Concrete Technology, 20(3), p.236 - 253, 2022/03
Times Cited Count:3 Percentile:7.07(Construction & Building Technology)This paper is a state-of-the-art report on the performance assessment of cementitious and related materials as components of engineered barrier systems for radioactive waste management. In this paper, (1) the concept of safety functions is reviewed as the engineering background of discussion, (2) an overview of the postclosure performance assessment for Belgian low- and intermediate-level short-lived radioactive waste disposal is provided, and (3) a modeling methodology for engineered barrier systems is analyzed using the concept of "mandala for durability mechanics". According to these works, authors present technical suggestions for technical stakeholders of Japanese low-level radioactive waste disposal.
Shimada, Asako; Taniguchi, Yoshinori; Kakiuchi, Kazuo; Ohira, Saki; Iida, Yoshihisa; Sugiyama, Tomoyuki; Amaya, Masaki; Maruyama, Yu
Scientific Reports (Internet), 12(1), p.2086_1 - 2086_11, 2022/02
Times Cited Count:3 Percentile:28.39(Multidisciplinary Sciences)no abstracts in English
Fujimoto, Shinji*; Tsuchiya, Hiroaki*; Ogawa, Soma*; Iida, Yoshihisa; Taniguchi, Naoki
Materials and Corrosion, 72(1-2), p.333 - 338, 2021/01
Times Cited Count:3 Percentile:13.68(Materials Science, Multidisciplinary)The stress corrosion cracking (SCC) of pure copper in bentonite was examined by a slow strain rate test (SSRT). The bentonite was swollen with pure water or aqueous solutions containing NH of 0.05 M and 0.1 M. The thick corrosion films and particulate deposits were formed on copper surface after SSRT. Typical tarnish rupture type SCC occurred on pure copper in swollen bentonite with and without NH
of 0.05 M and 0.1 M. The thick corrosion films and particulate deposits were formed on copper surface after SSRT. Typical tarnish rupture type SCC occurred on pure copper in swollen bentonite with and without NH . Crack propagation rate was enhanced by NH
. Crack propagation rate was enhanced by NH . It is confirmed thick oxide layer is formed on copper during plastic deformation which resulting in the tarnish crack type SCC. The many particulate deposits observed on the surface might be formed as a results of rapid dissolution of Cu
. It is confirmed thick oxide layer is formed on copper during plastic deformation which resulting in the tarnish crack type SCC. The many particulate deposits observed on the surface might be formed as a results of rapid dissolution of Cu to form porous CuO at locallized deformed sites.
to form porous CuO at locallized deformed sites.
Yamaguchi, Tetsuji; Ohira, Saki; Hemmi, Ko; Barr, L.; Shimada, Asako; Maeda, Toshikatsu; Iida, Yoshihisa
Radiochimica Acta, 108(11), p.873 - 877, 2020/11
Times Cited Count:8 Percentile:54.81(Chemistry, Inorganic & Nuclear)Iida, Yoshihisa; Nakadoi, Yasumasa; Yamaguchi, Tetsuji
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 24(1), p.53 - 64, 2017/06
The information on the contaminated water treatment in Fukushima Daiichi Nuclear Power Station announced by Tokyo Electric Power Co., Inc. (TEPCO) were summarized in terms of the management of the secondary wastes, for the purpose of accumulating technical knowledge for long-term storage of the wastes. Concerns for the long-term soundness of waste storage containers were pointed out as follows, corrosion of stainless steel containers exposed to radiation in the presence of chloride ions, corrosion of stainless steel containers under acidic conditions or in the presence of activated carbon, and radiation degradation of the high-performance container (HIC) in which slurry was stored.
Iida, Yoshihisa; Yamaguchi, Tetsuji; Tanaka, Tadao; Hemmi, Ko
Journal of Nuclear Science and Technology, 53(10), p.1573 - 1584, 2016/10
Times Cited Count:13 Percentile:72.77(Nuclear Science & Technology)The sorption behavior of thorium (Th) onto granitic rock and its major constituent were investigated by batch sorption experiments. Experiments were carried out under variable pH and carbonate concentrations. Distribution coefficients decreased with increased carbonate concentrations and showed the minimal value at pH 9-10. This sorption tendency was likely due to forming the hydroxide-carbonate complexes of Th in the solutions. The order of sorbability for Th was mica  feldspar
feldspar  quartz = granite. The sorption behaviors of Th onto these minerals were analyzed by the triple-layer surface complexation model with the Visual Minteq computer program. The model calculations assuming the inner-sphere surface complexation of Th were able to explain the experimental results reasonably well. It was shown that the sorption behavior of Th onto granite can be explained primarily by the complexation with the surface sites of feldspar.
quartz = granite. The sorption behaviors of Th onto these minerals were analyzed by the triple-layer surface complexation model with the Visual Minteq computer program. The model calculations assuming the inner-sphere surface complexation of Th were able to explain the experimental results reasonably well. It was shown that the sorption behavior of Th onto granite can be explained primarily by the complexation with the surface sites of feldspar.
Iida, Yoshihisa; Barr, L.; Yamaguchi, Tetsuji; Hemmi, Ko
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 23(1), p.3 - 8, 2016/06
Thorium (Th)-229 is one of the important radionuclides for the performance assessment calculations for high-level radioactive waste repositories. The sorption behavior of Th onto montmorillonite and illite were investigated by batch sorption experiments. Experiments were carried out under variable pH and carbonate concentrations. The sorbability of montmorillonite was higher than that of illite. Distribution coefficients, 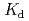 (m
(m kg
kg ), decreased with increased carbonate concentrations and showed the minimal value at around pH 10. The sorption behaviors of Th were analyzed by the non-electrostatic surface complex model with PHREEQC computer program. The model calculations were able to explain the experimental results reasonably well. The decreases of
), decreased with increased carbonate concentrations and showed the minimal value at around pH 10. The sorption behaviors of Th were analyzed by the non-electrostatic surface complex model with PHREEQC computer program. The model calculations were able to explain the experimental results reasonably well. The decreases of 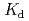 was likely due to the stabilization of aqueous species by hydroxo-carbonate complexations in the solutions.
was likely due to the stabilization of aqueous species by hydroxo-carbonate complexations in the solutions.
Hemmi, Ko; Yamaguchi, Tetsuji; Iida, Yoshihisa
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 22(1), p.3 - 10, 2015/06
Iodine and tin are important elements in performance assessment of geological disposal of radioactive wastes. Sorption experiments of iodine were carried out under varying nitrate concentration with a range of 0 to 5 mol dm at neutral pH range in order to determine the distribution coefficient of iodine was zero or non-zero value. The experimental results with estimated statistical errors showed non-zero values for tuffaceous sandstone except for NaNO
at neutral pH range in order to determine the distribution coefficient of iodine was zero or non-zero value. The experimental results with estimated statistical errors showed non-zero values for tuffaceous sandstone except for NaNO concentration 0.5 mol dm
concentration 0.5 mol dm . Non-zero values were also obtained under NaNO
. Non-zero values were also obtained under NaNO concentrations higher than 0.5 mol dm
concentrations higher than 0.5 mol dm for granodiorite. Sorption experiments of tin were carried out at high pH range in order to check whether the distribution coefficient of tin decreases significantly with pH as a result of formation of anionic hydrolysis species of tin. The distribution coefficients of tin on granodiorite decreased from 9.79
for granodiorite. Sorption experiments of tin were carried out at high pH range in order to check whether the distribution coefficient of tin decreases significantly with pH as a result of formation of anionic hydrolysis species of tin. The distribution coefficients of tin on granodiorite decreased from 9.79 10
10 m
m kg
kg at pH10.4 to 2.46
at pH10.4 to 2.46 10
10 m
m kg
kg at pH12.4. The distribution coefficient of tin on tuffaceous sandstone was about one order of magnitude higher (about 2
at pH12.4. The distribution coefficient of tin on tuffaceous sandstone was about one order of magnitude higher (about 2 10
10 m
m kg
kg ) than that of granodiorite at pH around 12.4.
) than that of granodiorite at pH around 12.4.
Iida, Yoshihisa
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 22(1), p.11 - 12, 2015/06
The summary of NUCE session titled "The current status and issues of the contaminated water treatment system at Fukushima Daiichi Nuclear Power Plant" in 2015 AESJ Spring Meeting is reported.
Yamaguchi, Tetsuji; Takeda, Seiji; Nishimura, Yuki; Iida, Yoshihisa; Tanaka, Tadao
Radiochimica Acta, 102(11), p.999 - 1008, 2014/11
Times Cited Count:1 Percentile:8.00(Chemistry, Inorganic & Nuclear)An attempt was made to select thermodynamic data with uncertainties and to evaluate the solubility of radioelements with uncertainties considering variation in groundwater chemistry. The thermodynamic data was selected by reviewing the JAEA-TDB released in 2012. Data for Nb, Pd and Pa were revised from the viewpoint of the consistency of the data selection process. Data for Se, U and Pa were revised from the viewpoint of the conservativeness. Up-to-date ternary calcium-metal(IV)-OH complexes were adopted for Zr, Th, U, Np and Pu. A Monte Carlo-based probabilistic calculation code, PA-SOL, was used for probabilistic analysis of the solubility.
Sakamaki, Keiko; Kataoka, Masaharu; Maeda, Toshikatsu; Iida, Yoshihisa; Kamoshida, Michio; Yamaguchi, Tetsuji; Tanaka, Tadao
Corrosion Engineering, Science and Technology, 49(6), p.450 - 454, 2014/09
Times Cited Count:1 Percentile:0.00(Materials Science, Multidisciplinary)Corrosion experiments of a carbon steel plate embedded in bentonite mixture were conducted toverify our models assessing Eh evolution induced by corrosion of carbon steel overpack. Theexperimental results showed that the Eh decreased for the first 200 days and was subsequentlystabilised at around -450 mV; corrosion products were identified as magnetite and Fe waspresent mostly as divalent Fe within a 5 mm range from the carbon steel plate. Reactive transportmodelling was performed to assess the Eh evolution in the system using kinetic dissolution modelfor metallic iron and thermodynamic equilibrium models for other chemical reactions and closelyreproduced the experimental results. The models were verified only under the conditionsemployed in this study.
 ) onto iron-containing minerals
) onto iron-containing mineralsIida, Yoshihisa; Yamaguchi, Tetsuji; Tanaka, Tadao
Journal of Nuclear Science and Technology, 51(3), p.305 - 322, 2014/03
Times Cited Count:15 Percentile:71.40(Nuclear Science & Technology)Sorption behaviors of HSe onto iron-containing minerals, magnetite, goethite, ferrous oxide and biotite, were investigated by batch sorption experiments under reducing conditions. The sorption ratios for goethite, as representative examples, show negative dependence on pH and independence of the NaCl concentration. The results indicate that the sorption behavior of HSe
onto iron-containing minerals, magnetite, goethite, ferrous oxide and biotite, were investigated by batch sorption experiments under reducing conditions. The sorption ratios for goethite, as representative examples, show negative dependence on pH and independence of the NaCl concentration. The results indicate that the sorption behavior of HSe is inner sphere surface complexation to ferrol sites. The triple layer model was used in the analysis of the sorption behavior of HSe
is inner sphere surface complexation to ferrol sites. The triple layer model was used in the analysis of the sorption behavior of HSe with Visual Minteq computer program. The intrinsic equilibrium constant was determined by the fitting of model calculations to the experimental results to be 5.5. The value was close to the value of HS
with Visual Minteq computer program. The intrinsic equilibrium constant was determined by the fitting of model calculations to the experimental results to be 5.5. The value was close to the value of HS (5.3) which is chemically analogous to HSe
(5.3) which is chemically analogous to HSe .
.
Sawaguchi, Takuma; Yamaguchi, Tetsuji; Iida, Yoshihisa; Tanaka, Tadao; Kitagawa, Isamu
Clay Minerals, 48(2), p.411 - 422, 2013/05
Times Cited Count:3 Percentile:8.75(Chemistry, Physical)Diffusive transport of Cs in compacted sand-bentonite mixtures was studied by a through diffusion method. Experiments were performed under variable aqueous compositions. Effective diffusivity (
in compacted sand-bentonite mixtures was studied by a through diffusion method. Experiments were performed under variable aqueous compositions. Effective diffusivity (
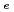 ) values of 5.2E-10
) values of 5.2E-10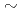 5.9E-9 m
5.9E-9 m s
s were obtained. The variation was somewhat large in the
were obtained. The variation was somewhat large in the 
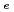 values. Apparent diffusivity (
values. Apparent diffusivity (
 ) values, on the other hand, were 2.0E-12
) values, on the other hand, were 2.0E-12  6.2E-12 m
6.2E-12 m s
s , which shows small variation. The results indicate that, in applying Fick's 1st law of diffusion, diffusive flux is proportional to the apparent concentration gradient of Cs in the sand-bentonite mixture rather than the gradient of Cs concentration in pore water. Since the apparent concentration gradient in sand-bentonite mixtures is nearly equal to the gradient of adsorbed Cs, diffusion of Cs under adsorbed state would be the main mechanism of diffusion of Cs in sand-bentonite mixtures.
, which shows small variation. The results indicate that, in applying Fick's 1st law of diffusion, diffusive flux is proportional to the apparent concentration gradient of Cs in the sand-bentonite mixture rather than the gradient of Cs concentration in pore water. Since the apparent concentration gradient in sand-bentonite mixtures is nearly equal to the gradient of adsorbed Cs, diffusion of Cs under adsorbed state would be the main mechanism of diffusion of Cs in sand-bentonite mixtures.
Otsuka, Ichiro*; Iida, Yoshihisa; Yamaguchi, Tetsuji; Kato, Osamu*; Tateishi, Tsuyoshi*; Tanaka, Tadao
Materials Research Society Symposium Proceedings, Vol.1475, p.507 - 512, 2012/06
We conducted corrosion experiments of carbon wires to identify the cathode reaction with overpack corrosion. Carbon steel wires were immersed in selected aqueous solutions of distilled water, sodium hydrogen carbonate solution and sodium sulfate solution sealed in glass ampoules. The ampoules were kept at 60  C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
C for 150 days. Corrosion products, liquid phase and gas phases were analyzed. The results showed that hydrogen gas generation by reduction of hydrogen ions was dominant cathode reaction. Reduction of sulfate and carbonate anions was not observed. Amorphous ferrous hydroxide and/or iron oxides were identified in all the experiments as the corrosion products. Iron carbonate was also identified in the sodium hydrogen carbonate solution. This result indicates that the hydrogen generation reaction might be the dominant cathode reaction under geological disposal environment and that the Eh of the pore water would reach the equilibrium potential of the hydrogen generation reaction.
Iida, Yoshihisa; Yamaguchi, Tetsuji; Tanaka, Tadao
Journal of Nuclear Science and Technology, 48(8), p.1170 - 1183, 2011/08
Times Cited Count:19 Percentile:78.40(Nuclear Science & Technology)Effective diffusion coefficients of selenium through bentonite sand mixtures under reducing and anaerobic conditions were obtained by the through-diffusion method. Experiments were carried out under variable bentonite content and porewater salinity. The employed bentonite material was Kunigel V1. The dominant diffusion species were determined to be HSe and SeO
and SeO
 under reducing and anaerobic conditions, respectively. The effective diffusion coefficients of selenium species decreased with increasing bentonite content and with decreasing salinity. This tendency is primarily due to anion exclusion which caused by negative surface charge of montmorillonite. A diffusion model for anionic species was developed based on the electric double layer theory and the pore diffusion model, to evaluate diffusion behaviors of HSe
under reducing and anaerobic conditions, respectively. The effective diffusion coefficients of selenium species decreased with increasing bentonite content and with decreasing salinity. This tendency is primarily due to anion exclusion which caused by negative surface charge of montmorillonite. A diffusion model for anionic species was developed based on the electric double layer theory and the pore diffusion model, to evaluate diffusion behaviors of HSe and SeO
and SeO
 in bentonite porewater. The effective diffusion coefficients of selenium species predicted by this model showed good agreement with experimentally measured ones.
in bentonite porewater. The effective diffusion coefficients of selenium species predicted by this model showed good agreement with experimentally measured ones.
Iida, Yoshihisa; Tanaka, Tadao; Yamaguchi, Tetsuji; Nakayama, Shinichi
Journal of Nuclear Science and Technology, 48(2), p.279 - 291, 2011/02
Times Cited Count:14 Percentile:69.66(Nuclear Science & Technology)Batch sorption experiments were carried out to investigate sorption behavior of selenium onto granodiorite, sandy mudstone, tuffaceous sandstone and their major constituent minerals and accessory minerals under reducing conditions. Rocks and groundwater employed in the experiments were sampled with a special care avoiding exposing the rocks and groundwater to the air to minimize their oxidation. The dominant sorbent minerals for selenium were determined to be biotite for granodiorite, and pyrite for sandy mudstone and tuffaceous sandstone at neutral-alkaline pH.
Iida, Yoshihisa; Yamaguchi, Tetsuji; Tanaka, Tadao; Nakayama, Shinichi
Journal of Nuclear Science and Technology, 47(5), p.431 - 438, 2010/05
Times Cited Count:22 Percentile:79.71(Nuclear Science & Technology)Solubility experiments for selenium were performed at high ionic strengths (1.0 and 2.0 M NaCl) under anoxic conditions. Dominant aqueous selenium species were identified as hydrogen selenide, HSe , at pH between 5 and 8, and polyselenide, Se
, at pH between 5 and 8, and polyselenide, Se
 at pH between 9 and 13. A solubility-limiting solid was determined to be amorphous selenium at pH between 5 and 8, and crystalline selenium at pH between 9 and 13. The equilibrium constants of Se(am) + H
at pH between 9 and 13. A solubility-limiting solid was determined to be amorphous selenium at pH between 5 and 8, and crystalline selenium at pH between 9 and 13. The equilibrium constants of Se(am) + H + 2e
+ 2e = HSe
= HSe , logK
, logK = -6.57
= -6.57 0.15, 4Se(cr) + 2e
0.15, 4Se(cr) + 2e = Se
= Se
 logK
logK = -16.67
= -16.67 0.03 and ion interaction coefficients for HSe
0.03 and ion interaction coefficients for HSe and Se
and Se
 versus Na
versus Na ,
,  (HSe
(HSe , Na
, Na ) = -0.01
) = -0.01 0.10 and
0.10 and  (Se
(Se
 , Na
, Na ) = -0.03
) = -0.03 0.02, were determined by specific ion interaction theory. The standard molar free energy of formation of amorphous selenium and polyselenide species were determined to be
0.02, were determined by specific ion interaction theory. The standard molar free energy of formation of amorphous selenium and polyselenide species were determined to be 
 G
G
 (Se(am)) = 6.0
(Se(am)) = 6.0 2.2 kJ mol
2.2 kJ mol ,
, 
 G
G
 (Se
(Se
 ) = 111.64
) = 111.64 1.61 kJ mol
1.61 kJ mol ,
, 
 G
G
 (Se
(Se
 ) = 98.68
) = 98.68 1.02 kJ mol
1.02 kJ mol and
and 
 G
G
 (Se
(Se
 ) = 95.14
) = 95.14 0.17 kJ mol
0.17 kJ mol , respectively.
, respectively.