Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ma, B.*; Teshigawara, Makoto; Wakabayashi, Yasuo*; Yan, M.*; Hashiguchi, Takao*; Yamagata, Yutaka*; Wang, S.*; Ikeda, Yujiro*; Otake, Yoshie*
Nuclear Instruments and Methods in Physics Research A, 995, p.165079_1 - 165079_7, 2021/04
Times Cited Count:2 Percentile:34.88(Instruments & Instrumentation)We have optimized a cold neutron moderator to be operated at the RIKEN accelerator-driven compact neutron source. We selected a safe and easy to manage material, mesitylene, as the RANS cold moderator. An efficient moderator system was designed by studying and optimizing a coupled cold neutron moderator of mesitylene at 20 K with a polyethylene (PE) pre-moderator at room temperature in the slab geometry with Particle and Heavy Ion Transport code System (PHITS) simulations. The parameters of mesitylene and PE thickness, the reflector, and the shielding configuration were studied to increase cold neutron intensities. Consequently, an integrated cold neutron intensity of 1.15 10
10 n/cm
n/cm /
/ A at 2 m from the neutron-producing target was finally achieved, which was 12 times higher than that of the current PE moderator. The results showed attractive application prospect of mesitylene as cold neutron moderator material.
A at 2 m from the neutron-producing target was finally achieved, which was 12 times higher than that of the current PE moderator. The results showed attractive application prospect of mesitylene as cold neutron moderator material.
Onuki, Yoshichika*; Kakihana, Masashi*; Iha, Wataru*; Nakaima, Kenri*; Aoki, Dai*; Nakamura, Ai*; Honda, Fuminori*; Nakashima, Miho*; Amako, Yasushi*; Gochi, Jun*; et al.
JPS Conference Proceedings (Internet), 29, p.012001_1 - 012001_9, 2020/02
 VFeAsO
VFeAsO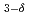 studied by X-ray magnetic circular dichroism
studied by X-ray magnetic circular dichroismHorio, Masafumi*; Takeda, Yukiharu; Namiki, Hiromasa*; Katagiri, Takao*; Wakabayashi, Yuki*; Sakamoto, Shoya*; Nonaka, Yosuke*; Shibata, Goro*; Ikeda, Keisuke*; Saito, Yuji; et al.
Journal of the Physical Society of Japan, 87(10), p.105001_1 - 105001_2, 2018/10
Times Cited Count:2 Percentile:21.2(Physics, Multidisciplinary)Sato, Hiroyuki; Nishida, Akemi; Ohashi, Hirofumi; Muramatsu, Ken*; Muta, Hitoshi*; Itoi, Tatsuya*; Takada, Tsuyoshi*; Hida, Takenori*; Tanabe, Masayuki*; Yamamoto, Tsuyoshi*; et al.
Proceedings of 2017 International Congress on Advances in Nuclear Power Plants (ICAPP 2017) (CD-ROM), 7 Pages, 2017/04
JAEA, in conjunction with Tokyo City University, The University of Tokyo and JGC Corporation, have started development of a PRA method considering the safety and design features of HTGR. The primary objective of the project is to develop a seismic PRA method which enables to provide a reasonably complete identification of accident scenario including a loss of safety function in passive system, structure and components. In addition, we aim to develop a basis for guidance to implement the PRA. This paper provides the overview of the activities including development of a system analysis method for multiple failures, a component failure data using the operation and maintenance experience in the HTTR, seismic fragility evaluation method, and mechanistic source term evaluation method considering failures in core graphite components and reactor building.
Matsuda, Kosuke*; Muramatsu, Ken*; Muta, Hitoshi*; Sato, Hiroyuki; Nishida, Akemi; Ohashi, Hirofumi; Itoi, Tatsuya*; Takada, Tsuyoshi*; Hida, Takenori*; Tanabe, Masayuki*; et al.
Proceedings of 2017 International Congress on Advances in Nuclear Power Plants (ICAPP 2017) (CD-ROM), 7 Pages, 2017/04
This paper proposes a set of procedures for accident sequence analysis in seismic PRAs of HTGRs that can consider the unique accident progression characteristics of HTGRs. Main features of our proposed procedure are as follows: (1) Systematic analysis techniques including Master Logic Diagrams are used to ensure reasonable completeness in identification of initiating events and classification of accident sequences, (2) Information on factors that govern the accident progression and source terms are effectively reflected to the construction of event trees for delineation of accident sequences, and (3) Frequency quantification of seismically-initiated accident sequence frequencies that involve multiplepipe ruptures are made with the use of the Direct Quantification of Fault Trees by Monte Carlo (DQFM) method by a computer code SECOM-DQFM.
Suzuki, Tomoya; Takao, Koichiro*; Kawasaki, Takeshi*; Harada, Masayuki*; Nogami, Masanobu*; Ikeda, Yasuhisa*
Polyhedron, 96, p.102 - 106, 2015/08
Times Cited Count:5 Percentile:41.5(Chemistry, Inorganic & Nuclear)We have determined crystal structures of UO (NO
(NO )
) (
(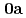 )
) (
(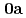 : 2-imidazolidone), UO
: 2-imidazolidone), UO (NO
(NO )
) (
(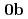 )
) (
(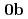 : tetrahydro-2-pyrimidone) and UO
: tetrahydro-2-pyrimidone) and UO (NO
(NO )
) (
(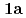 )
) (
(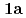 : 1-methyl-2-imidazolidone) by using single crystal X-ray analysis, and examined correlations between melting points (mps) and intermolecular hydrogen bonds (HBs) of UO
: 1-methyl-2-imidazolidone) by using single crystal X-ray analysis, and examined correlations between melting points (mps) and intermolecular hydrogen bonds (HBs) of UO (NO
(NO )
) (CU)
(CU) (CU: cyclic urea derivatives) and UO
(CU: cyclic urea derivatives) and UO (NO
(NO )
) (NRP)
(NRP) (NRP: pyrrolidone derivative).
(NRP: pyrrolidone derivative).
 -alkylated 2-pyrrolidone derivatives
-alkylated 2-pyrrolidone derivativesTakao, Koichiro*; Kawata, Yoshihisa*; Nogami, Masanobu*; Harada, Masayuki*; Morita, Yasuji; Nishimura, Kenji*; Ikeda, Yasuhisa*
Journal of Nuclear Science and Technology, 52(2), p.294 - 298, 2015/02
Times Cited Count:2 Percentile:17.57(Nuclear Science & Technology)Yields of precipitated UO (NO
(NO )
) (NRP)
(NRP) (NRP =
(NRP =  -alkylated 2-pyrrolidone) were precisely determined by considering reduction of the solution volume through the precipitation, which can be estimated from difference in acid concentrations of the liquid phases before and after the precipitation. The studied NRPs were
-alkylated 2-pyrrolidone) were precisely determined by considering reduction of the solution volume through the precipitation, which can be estimated from difference in acid concentrations of the liquid phases before and after the precipitation. The studied NRPs were  -
- -butyl (NBP) and
-butyl (NBP) and  -
- -propyl (NProP) derivatives. In both systems, the precipitation yields precisely determined were always higher than those simply calculated from the ratio of uranium concentrations before and after the precipitation. However, the differences between them are in the range of 0.6% - 2.6%. If such a difference is practically negligible, the volume reduction through the precipitation does not have to be taken into account for simplicity of the analytical manipulation.
-propyl (NProP) derivatives. In both systems, the precipitation yields precisely determined were always higher than those simply calculated from the ratio of uranium concentrations before and after the precipitation. However, the differences between them are in the range of 0.6% - 2.6%. If such a difference is practically negligible, the volume reduction through the precipitation does not have to be taken into account for simplicity of the analytical manipulation.
 ]) solution of [EMI]
]) solution of [EMI] [UO
[UO (NO
(NO )
) ]; First spectrophotometric evidence for existence of [UO
]; First spectrophotometric evidence for existence of [UO (NO
(NO )
) ]
]
Sasaki, Kotoe*; Suzuki, Tomoya*; Arai, Tsuyoshi*; Takao, Koichiro*; Suzuki, Shinichi; Yaita, Tsuyoshi; Ikeda, Yasuhisa*
Chemistry Letters, 43(5), p.670 - 672, 2014/05
Times Cited Count:7 Percentile:25.99(Chemistry, Multidisciplinary)no abstracts in English
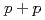 collisions at
collisions at 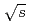 = 200 and 62.4 GeV
= 200 and 62.4 GeVAdare, A.*; Afanasiev, S.*; Aidala, C.*; Ajitanand, N. N.*; Akiba, Yasuyuki*; Al-Bataineh, H.*; Alexander, J.*; Aoki, Kazuya*; Aphecetche, L.*; Armendariz, R.*; et al.
Physical Review C, 83(6), p.064903_1 - 064903_29, 2011/06
Times Cited Count:184 Percentile:99.44(Physics, Nuclear)Transverse momentum distributions and yields for 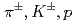 , and
, and 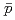 in
in 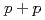 collisions at
collisions at 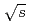 = 200 and 62.4 GeV at midrapidity are measured by the PHENIX experiment at the RHIC. We present the inverse slope parameter, mean transverse momentum, and yield per unit rapidity at each energy, and compare them to other measurements at different
= 200 and 62.4 GeV at midrapidity are measured by the PHENIX experiment at the RHIC. We present the inverse slope parameter, mean transverse momentum, and yield per unit rapidity at each energy, and compare them to other measurements at different 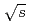 collisions. We also present the scaling properties such as
collisions. We also present the scaling properties such as 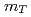 and
and 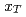 scaling and discuss the mechanism of the particle production in
scaling and discuss the mechanism of the particle production in 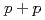 collisions. The measured spectra are compared to next-to-leading order perturbative QCD calculations.
collisions. The measured spectra are compared to next-to-leading order perturbative QCD calculations.
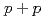 and Au+Au collisions at
and Au+Au collisions at 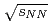 = 200 GeV
= 200 GeVAdare, A.*; Afanasiev, S.*; Aidala, C.*; Ajitanand, N. N.*; Akiba, Yasuyuki*; Al-Bataineh, H.*; Alexander, J.*; Aoki, Kazuya*; Aphecetche, L.*; Aramaki, Y.*; et al.
Physical Review C, 83(4), p.044912_1 - 044912_16, 2011/04
Times Cited Count:8 Percentile:49.7(Physics, Nuclear)Measurements of electrons from the decay of open-heavy-flavor mesons have shown that the yields are suppressed in Au+Au collisions compared to expectations from binary-scaled 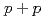 collisions. Here we extend these studies to two particle correlations where one particle is an electron from the decay of a heavy flavor meson and the other is a charged hadron from either the decay of the heavy meson or from jet fragmentation. These measurements provide more detailed information about the interaction between heavy quarks and the quark-gluon matter. We find the away-side-jet shape and yield to be modified in Au+Au collisions compared to
collisions. Here we extend these studies to two particle correlations where one particle is an electron from the decay of a heavy flavor meson and the other is a charged hadron from either the decay of the heavy meson or from jet fragmentation. These measurements provide more detailed information about the interaction between heavy quarks and the quark-gluon matter. We find the away-side-jet shape and yield to be modified in Au+Au collisions compared to 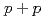 collisions.
collisions.
Suzuki, Tatsuya*; Nomura, Masao*; Fujii, Yasuhiko*; Ikeda, Atsushi; Takaoka, Toru*; Oguma, Koichi*
Nihon Ion Kokan Gakkai-Shi, 21(3), p.328 - 333, 2010/09
Zinc isotope fractionation in an anion exchange resin has been investigated in hydrochloric acid solution by chromatographic technique. It was found that the heavier zinc isotopes were located disproportionately in the solution phase. The isotope fractionation coefficient was varied from the order of 10 to 10
to 10 depending on the hydrochloric acid concentration. The maximum isotope fractionation coefficient was obtained in 1
depending on the hydrochloric acid concentration. The maximum isotope fractionation coefficient was obtained in 1 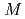 hydrochloric acid, while the distribution coefficient of zinc became maximum at around 2
hydrochloric acid, while the distribution coefficient of zinc became maximum at around 2 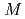 hydrochloric acid. This difference has been further discussed based on the calculated speciation and structural information derived from X-ray absorption spectroscopy.
hydrochloric acid. This difference has been further discussed based on the calculated speciation and structural information derived from X-ray absorption spectroscopy.
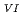 and Pu
and Pu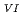
Kim, S.-Y.; Takao, Koichiro*; Haga, Yoshinori; Yamamoto, Etsuji; Kawata, Yoshihisa; Morita, Yasuji; Nishimura, Kenji*; Ikeda, Yasuhisa*
Crystal Growth & Design, 10(5), p.2033 - 2036, 2010/04
Times Cited Count:15 Percentile:77.61(Chemistry, Multidisciplinary)Plutonyl(VI) nitrate complexes with N-cyclohexyl-2-pyrrolidone (NCP) and N-neopentyl-2-pyrrolidone (NNpP) were prepared, and their molecular and crystal structures were determined by single crystal X-ray analysis. The obtained compounds have the similar composition, PuO (NO
(NO )
) (NRP)
(NRP) (NRP = NCP, NNpP), which are analogous to the corresponding U(VI) complexes. Both PuO
(NRP = NCP, NNpP), which are analogous to the corresponding U(VI) complexes. Both PuO (NO
(NO )
) (NRP)
(NRP) complexes show typical structural properties of actinyl(VI) nitrates, i.e., hexagonal-bipyramidal geometry consisting of two NRP molecules and two NO
complexes show typical structural properties of actinyl(VI) nitrates, i.e., hexagonal-bipyramidal geometry consisting of two NRP molecules and two NO
 ions located in trans positions in the equatorial plane of PuO
ions located in trans positions in the equatorial plane of PuO
 moiety. These findings provide one of criteria in selection of suitable NRP as a precipitation agent for the spent nuclear fuel reprocessing based on the precipitation method.
moiety. These findings provide one of criteria in selection of suitable NRP as a precipitation agent for the spent nuclear fuel reprocessing based on the precipitation method.
Ikeda, Atsushi; Tsushima, Satoru*; Takao, Koichiro*; Rossberg, A.*; Funke, H.*; Scheinost, A. C.*; Bernhard, G.*; Yaita, Tsuyoshi; Hennig, C.*
Inorganic Chemistry, 48(24), p.11779 - 11787, 2009/12
Times Cited Count:34 Percentile:79.72(Chemistry, Inorganic & Nuclear)The electrochemical behavior and complex structure of Np carbonato complexes have been investigated in aqueous Na CO
CO and Na
and Na CO
CO /NaOH solutions at different oxidation states by using cyclic voltammetry, X-ray absorption spectroscopy, and density functional theory calculations. The end-member complexes of penta- and hexavalent Np in 1.5 M Na
/NaOH solutions at different oxidation states by using cyclic voltammetry, X-ray absorption spectroscopy, and density functional theory calculations. The end-member complexes of penta- and hexavalent Np in 1.5 M Na CO
CO with pH = 11.7 have been determined as a transdioxo neptunyl tricarbonato complex, [NpO
with pH = 11.7 have been determined as a transdioxo neptunyl tricarbonato complex, [NpO (CO
(CO )
) ]
]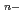 (
( = 5 for Np
= 5 for Np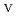 , and 4 for Np
, and 4 for Np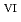 ). In contrast, the electrochemical oxidation of Np
). In contrast, the electrochemical oxidation of Np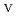 in a highly basic carbonate solution of 2.0M Na
in a highly basic carbonate solution of 2.0M Na CO
CO /1.0M NaOH (pH
/1.0M NaOH (pH  13) yielded a stable heptavalent Np complex of [Np
13) yielded a stable heptavalent Np complex of [Np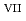 O
O (OH)
(OH) ]
] , indicating that the oxidation reaction from Np
, indicating that the oxidation reaction from Np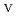 to Np
to Np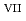 in the carbonate solution involves a drastic structural rearrangement from the transdioxo configuration to a square-planar-tetraoxo configuration, as well as exchanging the coordinating anions from carbonate ions (CO
in the carbonate solution involves a drastic structural rearrangement from the transdioxo configuration to a square-planar-tetraoxo configuration, as well as exchanging the coordinating anions from carbonate ions (CO
 ) to hydroxide ions (OH
) to hydroxide ions (OH ).
).
Morita, Yasuji; Takao, Koichiro*; Kim, S.-Y.; Kawata, Yoshihisa; Harada, Masayuki*; Nogami, Masanobu*; Nishimura, Kenji*; Ikeda, Yasuhisa*
Journal of Nuclear Science and Technology, 46(12), p.1129 - 1136, 2009/12
Times Cited Count:18 Percentile:74.76(Nuclear Science & Technology)A reprocessing system for spent FBR fuels consisting of two precipitation processes has been proposed. In this system, first only U(IV) species are precipitated using pyrrolidone derivative with low hydrophobicity and donicity, and secondly residual U(VI) and Pu(IV, VI) are precipitated simultaneously using pyrrolidone derivative with high precipitation ability. In this study, we have examined precipitation behavior of U(VI), Pu(IV), and Pu(VI) species in nitric acid solutions by using  -
- -propyl-2-pyrrolidone (NProP),
-propyl-2-pyrrolidone (NProP),  -butyl-2-pyrrolidone (NBP),
-butyl-2-pyrrolidone (NBP),  -
-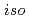 -butyl-2-pyrrolidone (NiBP), or
-butyl-2-pyrrolidone (NiBP), or  -cyclohexyl-2-pyrrolidone (NCP) to select the precipitants for the first precipitation process. As a result, NBP were found to be the most promising precipitant for the first precipitation process.
-cyclohexyl-2-pyrrolidone (NCP) to select the precipitants for the first precipitation process. As a result, NBP were found to be the most promising precipitant for the first precipitation process.
 -Alkyl-2-pyrrolidone derivatives (Alkyl =
-Alkyl-2-pyrrolidone derivatives (Alkyl =  -propyl,
-propyl,  -butyl,
-butyl, 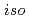 -butyl, and cyclohexyl)
-butyl, and cyclohexyl)Takao, Koichiro*; Noda, Kyoko*; Nogami, Masanobu*; Sugiyama, Yuichi*; Harada, Masayuki*; Morita, Yasuji; Nishimura, Kenji*; Ikeda, Yasuhisa*
Journal of Nuclear Science and Technology, 46(10), p.995 - 999, 2009/10
Times Cited Count:14 Percentile:66.94(Nuclear Science & Technology)We have been developing an advanced reprocessing system for spent FBR fuels based on precipitation method using pyrrolidone derivatives. In the present study, the solubility of UO (NO
(NO )
) (NRP)
(NRP) (NRP =
(NRP =  -alkyl-2-pyrrolidone, alkyl =
-alkyl-2-pyrrolidone, alkyl =  -propyl,
-propyl,  -butyl,
-butyl, 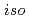 -butyl and cyclohexyl) in aqueous solutions with HNO
-butyl and cyclohexyl) in aqueous solutions with HNO has been examined. As a result, the solubility of each species of UO
has been examined. As a result, the solubility of each species of UO (NO
(NO )
) (NRP)
(NRP) generally decreases with increasing concentrations of HNO
generally decreases with increasing concentrations of HNO and NRP (
and NRP (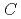 (HNO
(HNO ) and
) and 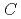 (NRP), respectively). The solubility of UO
(NRP), respectively). The solubility of UO (NO
(NO )
) (NRP)
(NRP) also depends on the type of NRP; a higher hydrophobicity of NRP generally leads to a lower solubility of UO
also depends on the type of NRP; a higher hydrophobicity of NRP generally leads to a lower solubility of UO (NO
(NO )
) (NRP)
(NRP) . The logarithms of effective solubility products (
. The logarithms of effective solubility products (
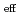 ) of UO
) of UO (NO
(NO )
) (NRP)
(NRP) at different
at different 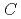 (HNO
(HNO ) values and 293 K were evaluated.
) values and 293 K were evaluated.
Matsukawa, Makoto; Kikuchi, Mitsuru; Fujii, Tsuneyuki; Fujita, Takaaki; Hayashi, Takao; Higashijima, Satoru; Hosogane, Nobuyuki; Ikeda, Yoshitaka; Ide, Shunsuke; Ishida, Shinichi; et al.
Fusion Engineering and Design, 83(7-9), p.795 - 803, 2008/12
Times Cited Count:17 Percentile:72.86(Nuclear Science & Technology)no abstracts in English
 -alkylated 2-pyrrolidone derivatives; Design and optimization of promising precipitant for uranyl ion
-alkylated 2-pyrrolidone derivatives; Design and optimization of promising precipitant for uranyl ionTakao, Koichiro*; Noda, Kyoko*; Morita, Yasuji; Nishimura, Kenji*; Ikeda, Yasuhisa*
Crystal Growth & Design, 8(7), p.2364 - 2376, 2008/07
Times Cited Count:35 Percentile:89.88(Chemistry, Multidisciplinary)Molecular and crystal structures of UO (NO
(NO )
) (NRP)
(NRP) (NRP = N-alkylated 2-pyrrolidone derivative) have been investigated by using single crystal X-ray analysis, IR and Raman spectroscopies. All UO
(NRP = N-alkylated 2-pyrrolidone derivative) have been investigated by using single crystal X-ray analysis, IR and Raman spectroscopies. All UO (NO
(NO )
) (NRP)
(NRP) complexes have typical structural properties of UO
complexes have typical structural properties of UO (NO
(NO )
) (L)
(L) (L = unidentate ligand), i.e., hexagonal-bipyramidal geometry, two NRP and two NO
(L = unidentate ligand), i.e., hexagonal-bipyramidal geometry, two NRP and two NO
 located in trans positions in an equatorial plane of the uranyl ion. Observation of the crystals of the uranyl nitrate complexes with N-n-propyl-2-pyrrolidone and N-iso-propyl-2-pyrrolidone indicate the presence of significant voids in the crystal lattices of these compounds. From this result, an approach for construction of efficient packing of UO
located in trans positions in an equatorial plane of the uranyl ion. Observation of the crystals of the uranyl nitrate complexes with N-n-propyl-2-pyrrolidone and N-iso-propyl-2-pyrrolidone indicate the presence of significant voids in the crystal lattices of these compounds. From this result, an approach for construction of efficient packing of UO (NO
(NO )
) (NRP)
(NRP) was proposed. Consequently, it was found that N-iso-butyl-2-pyrrolidone completely satisfies with the requirement for the efficient packing by filling the voids with the alkyl chain.
was proposed. Consequently, it was found that N-iso-butyl-2-pyrrolidone completely satisfies with the requirement for the efficient packing by filling the voids with the alkyl chain.
Yoshikawa, Hiroshi; Sakaki, Hironao; Sako, Hiroyuki; Takahashi, Hiroki; Shen, G.; Kato, Yuko; Ito, Yuichi; Ikeda, Hiroshi*; Ishiyama, Tatsuya*; Tsuchiya, Hitoshi*; et al.
Proceedings of International Conference on Accelerator and Large Experimental Physics Control Systems (ICALEPCS '07) (CD-ROM), p.62 - 64, 2007/10
J-PARC is a large scale facility of the proton accelerators for the multi-purpose of scientific researches in Japan. This facility consists of three accelerators and three experimental stations. Now, J-PARC is under construction, and LINAC is operated for one year, 3GeV synchrotron has just started the commissioning in this October the 1st. The completion of this facility will be next summer. The control system of accelerators established fundamental performance for the initial commissioning. The most important requirement to the control system of this facility is to minimize the activation of accelerator devices. In this paper, we show that the performances of each layer of this control system have been achieved in the initial stage.
Ikeda, Yasuhisa*; Takao, Koichiro*; Harada, Masayuki*; Morita, Yasuji; Nogami, Masanobu*; Nishimura, Kenji*
Proceedings of International Conference on Advanced Nuclear Fuel Cycles and Systems (Global 2007) (CD-ROM), p.1503 - 1507, 2007/09
We have developed a reprocessing process for spent FBR fuels based on the precipitation method using pyrrolidone derivatives. In previous investigation, N-cyclohexyl-2-pyrrolidone (NCP) is used as a precipitant and a process consisting of selective U precipitation step and U-Pu co-precipitation step was developed. In the present study, in order to examine the applicability of precipitants with lower hydrophobicity than NCP to the selective U precipitation step, we have carried out precipitation experiments of U(VI) by N-butyl-2-pyrrolidone (NBP) and N-propyl-2-pyrrolidone (NProP) and measured decontamination factors of some fission products.
Oigawa, Hiroyuki; Yokoo, Takeshi*; Nishihara, Kenji; Morita, Yasuji; Ikeda, Takao*; Takaki, Naoyuki*
Journal of Nuclear Science and Technology, 44(3), p.398 - 404, 2007/03
Times Cited Count:23 Percentile:81.77(Nuclear Science & Technology)Benefit of implementing Partitioning and Transmutation (P&T) technology was parametrically surveyed in terms of high-level radioactive waste (HLW) disposal by discussing possible reduction of the geological repository area. The results showed that, by recycling minor actinides (MA), the placement area could be reduced by 17-29% in the case of UO -LWR and by 63-85% in the case of MOX-LWR in comparison with the conventional PUREX reprocessing. This significant impact in MOX fuel was caused by the recycle of
-LWR and by 63-85% in the case of MOX-LWR in comparison with the conventional PUREX reprocessing. This significant impact in MOX fuel was caused by the recycle of  Am which was a long-term heat source. Further 70-80% reduction of the placement area in comparison with the MA-recovery case could be expected by partitioning the fission products (FP) into several groups for both fuel types.
Am which was a long-term heat source. Further 70-80% reduction of the placement area in comparison with the MA-recovery case could be expected by partitioning the fission products (FP) into several groups for both fuel types.