Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Fukuda, Tatsuya*; Takahashi, Ryo*; Hara, Takuhi*; Ohara, Koji*; Kato, Kazuo*; Matsumura, Daiju; Inaba, Yusuke*; Nakase, Masahiko*; Takeshita, Kenji*
Journal of Nuclear Science and Technology, 58(4), p.399 - 404, 2021/04
Times Cited Count:7 Percentile:55.56(Nuclear Science & Technology)Takeda, Tetsuaki*; Inagaki, Yoshiyuki; Aihara, Jun; Aoki, Takeshi; Fujiwara, Yusuke; Fukaya, Yuji; Goto, Minoru; Ho, H. Q.; Iigaki, Kazuhiko; Imai, Yoshiyuki; et al.
High Temperature Gas-Cooled Reactors; JSME Series in Thermal and Nuclear Power Generation, Vol.5, 464 Pages, 2021/02
As a general overview of the research and development of a High Temperature Gas-cooled Reactor (HTGR) in JAEA, this book describes the achievements by the High Temperature Engineering Test Reactor (HTTR) on the designs, key component technologies such as fuel, reactor internals, high temperature components, etc., and operational experience such as rise-to-power tests, high temperature operation at 950 C, safety demonstration tests, etc. In addition, based on the knowledge of the HTTR, the development of designs and component technologies such as high performance fuel, helium gas turbine and hydrogen production by IS process for commercial HTGRs are described. These results are very useful for the future development of HTGRs. This book is published as one of a series of technical books on fossil fuel and nuclear energy systems by the Power Energy Systems Division of the Japan Society of Mechanical Engineers.
C, safety demonstration tests, etc. In addition, based on the knowledge of the HTTR, the development of designs and component technologies such as high performance fuel, helium gas turbine and hydrogen production by IS process for commercial HTGRs are described. These results are very useful for the future development of HTGRs. This book is published as one of a series of technical books on fossil fuel and nuclear energy systems by the Power Energy Systems Division of the Japan Society of Mechanical Engineers.
Yin, X.; Zhang, L.*; Harigai, Miki*; Wang, X.*; Ning, S.*; Nakase, Masahiko*; Koma, Yoshikazu; Inaba, Yusuke*; Takeshita, Kenji*
Water Research, 177, p.115804_1 - 115804_10, 2020/06
Yin, X.; Zhang, L.*; Meng, C.*; Inaba, Yusuke*; Wang, X.*; Nitta, Ayako; Koma, Yoshikazu; Takeshita, Kenji*
Journal of Hazardous Materials, 387, p.121677_1 - 121677_10, 2020/04
Times Cited Count:13 Percentile:45.76(Engineering, Environmental)Tsutsui, Nao; Ban, Yasutoshi; Suzuki, Hideya*; Nakase, Masahiko*; Ito, Sayumi*; Inaba, Yusuke*; Matsumura, Tatsuro; Takeshita, Kenji*
Analytical Sciences, 36(2), p.241 - 246, 2020/02
Times Cited Count:30 Percentile:83.49(Chemistry, Analytical)To investigate the effective separation of actinides (Ans) from lanthanides (Lns), single-stage batch extraction experiments were performed with a novel extractant, tetradodecyl-1,10-phenanthroline-2,9-diamide (TDdPTDA) with various diluents such as 3-nitrobenzotrifluoride (F-3), nitrobenzene, and  -dodecane for Am, Cm, and Lns. The extraction kinetics with TDdPTDA was rapid enough to perform the actual extraction flow sheet. The slopes of the distribution ratio versus TDdPTDA concentration and the distribution ratio versus nitric acid concentration were similar for F-3 and nitrobenzene systems but different from
-dodecane for Am, Cm, and Lns. The extraction kinetics with TDdPTDA was rapid enough to perform the actual extraction flow sheet. The slopes of the distribution ratio versus TDdPTDA concentration and the distribution ratio versus nitric acid concentration were similar for F-3 and nitrobenzene systems but different from  -dodecane system. These differences were attributed to the characteristics of the diluents. This study reveals high distribution ratios of Am (
-dodecane system. These differences were attributed to the characteristics of the diluents. This study reveals high distribution ratios of Am (
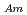 ) and Cm (
) and Cm (
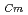 ) for TDdPTDA, with the high separation factors (
) for TDdPTDA, with the high separation factors ( s) of Am from Lns enough for their separation.
s) of Am from Lns enough for their separation.
Mishima, Ria; Inaba, Yusuke*; Tachioka, Sotaro*; Harigai, Miki*; Watanabe, Shinta*; Onoe, Jun*; Nakase, Masahiko*; Matsumura, Tatsuro; Takeshita, Kenji*
Chemistry Letters, 49(1), p.83 - 86, 2020/01
Times Cited Count:6 Percentile:24.27(Chemistry, Multidisciplinary)Separation of platinum group metals (PGMs) from high-level liquid waste generated from the reprocessing of spent nuclear fuels is important to produce good quality vitrified glass for final disposal. A new sorbent, Aluminum hexacyanoferrate (AlHCF), was synthesized and the general sorption behavior of PGMs from concentrated nitric acid was examined. Nitric acid caused substantial elution of AlHCF but the sorption of Pd stabilized the structure. Consequently, Rh was sorbed in the presence of Pd, whereas single Rh sorption caused complete dissolution of AlHCF. Relation between sorbed mount of Pd vs eluted Al and Fe revealed that the elution ratio of Al and Fe was not the same as molar ratio of synthesized AlHCF, indicating the re-sorption of Fe resulted in formation of new structure. The sorption mechanism of PGMs by this new sorbent, AlHCF, not only the simple ion exchange, but also oxidation reduction reaction as well as kinetics play important rule. Understanding the general sorption and dissolution behavior will help improve the sorption performance of PGMs by AlHCF.
Onishi, Takashi; Sekioka, Ken*; Suto, Mitsuo*; Tanaka, Kosuke; Koyama, Shinichi; Inaba, Yusuke*; Takahashi, Hideharu*; Harigai, Miki*; Takeshita, Kenji*
Energy Procedia, 131, p.151 - 156, 2017/12
Times Cited Count:14 Percentile:98.75(Energy & Fuels)no abstracts in English
Koyama, Shinichi; Suto, Mitsuo; Obayashi, Hiroshi; Takeshita, Kenji*; Ogata, Takeshi*; Oaki, Hiroshi*; Inaba, Yusuke*
Separation Science and Technology, 47(14-15), p.2024 - 2028, 2012/11
Times Cited Count:3 Percentile:15.34(Chemistry, Multidisciplinary)A series of separation experiments was performed to study the recovery process for minor actinides (MAs) from the actual spent fuel by using an extraction chromatographic technique. TPPEN gel was used as a new extraction chromatographic agent. Mixed oxide fuel was used as a reference spent fuel to demonstrate the recovery of the MAs. The MOX fuel, including 29.9 wt% plutonium (Pu), was irradiated up to 119 GWd/MTM, and the fuel was then prepared for the extraction experiment. A Mixed solution of MAs and lanthanides (Lns) was prepared. The TPPEN gel was immersed in a 0.01 M NaNO solution, and the pH was adjusted to 4.0. Next, an extraction column was prepared using the TPPEN gel, and the mixed solution of MAs and Lns was passed through the column. The Lns were detected in the eluent after washing with 0.01 M NaNO
solution, and the pH was adjusted to 4.0. Next, an extraction column was prepared using the TPPEN gel, and the mixed solution of MAs and Lns was passed through the column. The Lns were detected in the eluent after washing with 0.01 M NaNO (pH 4.0). For detecting the MAs, the pH of the eluent was changed to 2.0.
(pH 4.0). For detecting the MAs, the pH of the eluent was changed to 2.0.
Inaba, Yusuke*; Tsumagari, Takayuki*; Kida, Tatsuya*; Watanabe, Wataru*; Nakajima, Yasutaka*; Fukuoka, Sachio*; Mori, Atsunori*; Matsumura, Tatsuro; Nakano, Yoshio*; Takeshita, Kenji*
Polymer Journal, 43(7), p.630 - 634, 2011/07
Times Cited Count:11 Percentile:34.06(Polymer Science) -(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing a polymerizable double bond in the substituent structure of the pyridine ring are synthesized and subjected to copolymerization with
-(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing a polymerizable double bond in the substituent structure of the pyridine ring are synthesized and subjected to copolymerization with  -isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA) gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing C3, C4, C10 and branched C3 spacer chains are synthesized and temperature-dependent extraction behavior of cadmium ion is compared. These gels extract Cd(II) ion efficiently from the aqueous solution in the swelling state at 5
-isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA) gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing C3, C4, C10 and branched C3 spacer chains are synthesized and temperature-dependent extraction behavior of cadmium ion is compared. These gels extract Cd(II) ion efficiently from the aqueous solution in the swelling state at 5 C, while little extraction is observed at 45
C, while little extraction is observed at 45 C with shrinking. It is found that poly(TPEN-NIPA) gel of branched C3 spacer (C3b) shows the excellent thermoresponsive extraction performance.
C with shrinking. It is found that poly(TPEN-NIPA) gel of branched C3 spacer (C3b) shows the excellent thermoresponsive extraction performance.
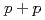 collisions at
collisions at 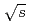 = 200 and 62.4 GeV
= 200 and 62.4 GeVAdare, A.*; Afanasiev, S.*; Aidala, C.*; Ajitanand, N. N.*; Akiba, Yasuyuki*; Al-Bataineh, H.*; Alexander, J.*; Aoki, Kazuya*; Aphecetche, L.*; Armendariz, R.*; et al.
Physical Review C, 83(6), p.064903_1 - 064903_29, 2011/06
Times Cited Count:192 Percentile:99.39(Physics, Nuclear)Transverse momentum distributions and yields for 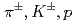 , and
, and 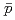 in
in 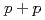 collisions at
collisions at 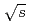 = 200 and 62.4 GeV at midrapidity are measured by the PHENIX experiment at the RHIC. We present the inverse slope parameter, mean transverse momentum, and yield per unit rapidity at each energy, and compare them to other measurements at different
= 200 and 62.4 GeV at midrapidity are measured by the PHENIX experiment at the RHIC. We present the inverse slope parameter, mean transverse momentum, and yield per unit rapidity at each energy, and compare them to other measurements at different 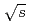 collisions. We also present the scaling properties such as
collisions. We also present the scaling properties such as 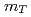 and
and 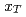 scaling and discuss the mechanism of the particle production in
scaling and discuss the mechanism of the particle production in 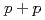 collisions. The measured spectra are compared to next-to-leading order perturbative QCD calculations.
collisions. The measured spectra are compared to next-to-leading order perturbative QCD calculations.
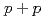 and Au+Au collisions at
and Au+Au collisions at 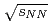 = 200 GeV
= 200 GeVAdare, A.*; Afanasiev, S.*; Aidala, C.*; Ajitanand, N. N.*; Akiba, Yasuyuki*; Al-Bataineh, H.*; Alexander, J.*; Aoki, Kazuya*; Aphecetche, L.*; Aramaki, Y.*; et al.
Physical Review C, 83(4), p.044912_1 - 044912_16, 2011/04
Times Cited Count:10 Percentile:54.72(Physics, Nuclear)Measurements of electrons from the decay of open-heavy-flavor mesons have shown that the yields are suppressed in Au+Au collisions compared to expectations from binary-scaled 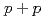 collisions. Here we extend these studies to two particle correlations where one particle is an electron from the decay of a heavy flavor meson and the other is a charged hadron from either the decay of the heavy meson or from jet fragmentation. These measurements provide more detailed information about the interaction between heavy quarks and the quark-gluon matter. We find the away-side-jet shape and yield to be modified in Au+Au collisions compared to
collisions. Here we extend these studies to two particle correlations where one particle is an electron from the decay of a heavy flavor meson and the other is a charged hadron from either the decay of the heavy meson or from jet fragmentation. These measurements provide more detailed information about the interaction between heavy quarks and the quark-gluon matter. We find the away-side-jet shape and yield to be modified in Au+Au collisions compared to 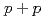 collisions.
collisions.
Matsumura, Tatsuro; Inaba, Yusuke*; Mori, Atsunori*; Takeshita, Kenji*
Journal of Nuclear Science and Technology, 47(2), p.123 - 126, 2010/02
Times Cited Count:13 Percentile:63.45(Nuclear Science & Technology)We are developing a new MA/Ln separation process with TPEN and its derivatives for P&T technology. TPEN is a hexadentate soft-donor ligand that has six soft-donor sites as a kind of podand type molecule and can encapsulate a metal ion. TPEN has good selectivity of Am(III) from Ln(III) and has potential to establish partitioning of MA. From the viewpoint of practical application, the hydrophobicity of TPEN must be improved. Because the dissolution of a slight amount of TPEN (about 10 mol/l) to water will restrict decontamination of Ln in extraction process. In this study, the hydrophobicity of TPEN was improved by introducing alkyl groups and the effect of the introduction of alkyl groups on the separation of Am(III) and Eu(III) was examined. One of them, N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine, TBPEN, showed good selectivity and the maximum separation factor, SF
mol/l) to water will restrict decontamination of Ln in extraction process. In this study, the hydrophobicity of TPEN was improved by introducing alkyl groups and the effect of the introduction of alkyl groups on the separation of Am(III) and Eu(III) was examined. One of them, N,N,N',N'-tetrakis((5-butoxypyridin-2-yl)-methyl)ethylenediamine, TBPEN, showed good selectivity and the maximum separation factor, SF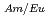 , was 91 at pH 3.02. The hydrophobicity of TBPEN is checked by the distribution data between water and chloroform, and improved successfully. The approach for development of hydrophobic derivatives of TPEN was effective.
, was 91 at pH 3.02. The hydrophobicity of TBPEN is checked by the distribution data between water and chloroform, and improved successfully. The approach for development of hydrophobic derivatives of TPEN was effective.
Fukuoka, Sachio*; Kida, Tatsuya*; Nakajima, Yasutaka*; Tsumagari, Takayuki*; Watanabe, Wataru*; Inaba, Yusuke*; Mori, Atsunori*; Matsumura, Tatsuro; Nakano, Yoshio*; Takeshita, Kenji*
Tetrahedron, 66(9), p.1721 - 1727, 2010/02
Times Cited Count:18 Percentile:47.28(Chemistry, Organic)N,N,N',N'-(tetrakis-2-pyridylmethyl)ethylenediamine (TPEN) derivatives bearing the different number (1-4) of a double bond moiety on the pyridine ring are synthesized and subjected to copolymerization with N-isopropylacrylamide in the presence of AIBN. The obtained poly(TPEN-NIPA)gels show thermo-responsive swelling/shrinking behaviors and are employed for the extraction of cadmium(II) ion from the aqueous solution to examine the relationship of the gel characteristics and the extraction performance. The polymer gels composed of the TPEN derivative bearing three or four double bonds exhibit temperature-dependent change of swelling and shrinking in water. These gels extract CdII ion efficiently from the aqueous solution in the swelling state at 5 C, while little extraction was observed at 45
C, while little extraction was observed at 45 C with shrinking.
C with shrinking.
Matsumura, Tatsuro; Takeshita, Kenji*; Inaba, Yusuke*; Mori, Atsunori*
Proceedings of 10th OECD/NEA Information Exchange Meeting on Actinide and Fission Product Partitioning and Transmutation (CD-ROM), 6 Pages, 2010/00
no abstracts in English
 -tetrakis((5-alkoxypyridin-2-yl)methyl)ethylenediamine, TRPEN, effective ligands for the separation of trivalent minor actinides from lanthanides
-tetrakis((5-alkoxypyridin-2-yl)methyl)ethylenediamine, TRPEN, effective ligands for the separation of trivalent minor actinides from lanthanidesMatsumura, Tatsuro; Inaba, Yusuke*; Koma, Yoshikazu; Morita, Yasuji; Mori, Atsunori*; Takeshita, Kenji*
Proceedings of International Conference on Advanced Nuclear Fuel Cycle; Sustainable Options & Industrial Perspectives (Global 2009) (CD-ROM), p.1107 - 1112, 2009/09
no abstracts in English
Matsumura, Tatsuro; Inaba, Yusuke*; Takeshita, Kenji*; Mori, Atsunori*
Proceedings of International Solvent Extraction Conference "Solvent Extraction-Fundamentals to Industrial Applications" (ISEC 2008), 4 Pages, 2008/09
We are developing a new MA/Ln separation process with derivatives of TPEN (N,N,N',N'-tetrakis(2-pyridylmethyl)ethylenediamine) for P&T technology. TPEN has good selectivity of Am(III) from Ln(III). However, TPEN is soluble slightly (about 10 mol/l) to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize several new hydrophobic derivative of TPEN. One of the ligands, TDdPEN (N,N,N',N'-tetrakis((5-dodecyloxypyridin-2-yl)-methyl)ethylenediamine) showed high selectivity. The maximum separation factor, SF
mol/l) to water. In this study, the hydrophobicity of TPEN is improved by introducing alkyl groups and the selectivity of Am(III) and Eu(III) is examined. We succeeded to synthesize several new hydrophobic derivative of TPEN. One of the ligands, TDdPEN (N,N,N',N'-tetrakis((5-dodecyloxypyridin-2-yl)-methyl)ethylenediamine) showed high selectivity. The maximum separation factor, SF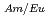 , was over 800. Hydrophobic derivatives of TPEN that have potential of application to the MA/Ln separation process was synthesized successfully.
, was over 800. Hydrophobic derivatives of TPEN that have potential of application to the MA/Ln separation process was synthesized successfully.
Suto, Mitsuo; Obayashi, Hiroshi; Koyama, Shinichi; Sekioka, Ken*; Takeshita, Kenji*; Inaba, Yusuke*; Ogata, Takeshi*; Oaki, Hiroshi*
no journal, ,
no abstracts in English
Suzuki, Shinichi; Akutsu, Kazuhiro; Yaita, Tsuyoshi; Shiwaku, Hideaki; Kobayashi, Toru; Takeshita, Kenji*; Ogata, Takeshi*; Inaba, Yusuke*; Oaki, Hiroshi*; Mori, Atsunori*
no journal, ,
no abstracts in English
Amamoto, Ippei; Kobayashi, Hidekazu; Ayame, Yasuo; Hasegawa, Yoshio*; Utsumi, Kazuo*; Takeshita, Kenji*; Inaba, Yusuke*; Onoe, Jun*; Koshizaka, Akiko*; Kaneta, Yui*
no journal, ,
no abstracts in English
Amamoto, Ippei; Kobayashi, Hidekazu; Ayame, Yasuo; Onishi, Takashi; Inaba, Yusuke*; Utsumi, Kazuo*; Takeshita, Kenji*; Onoe, Jun*; Koshizaka, Akiko*; Hasegawa, Yoshio*
no journal, ,
The development on synthesis of sorbent was carried out to separate the platinum group species such as Ru, Rh and Pd in high-level radioactive liquid waste. After sorption test using developed sorbents, the spherical porous silica impregnated aluminium ferrocyanide was effective to sorb the species of Ru and Pd in the nitric solution. This sorption effect remained stable when tested on actual high-level radioactive liquid waste. It was found that more treated waste could be loaded into the borosilicate glass than untreated waste.