Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Watabe, Hiroshi*; Sato, Tatsuhiko; Yu, K. N.*; Zivkovic, M.*; Krstic, D.*; Nikezic, D.*; Kim, K. M.*; Yamaya, Taiga*; Kawachi, Naoki*; Tanaka, Hiroki*; et al.
Radiation Protection Dosimetry, 200(2), p.130 - 142, 2024/02
Times Cited Count:2 Percentile:43.92(Environmental Sciences)Previously, we have developed DynamicMC for modelling relative movement of ORNL phantom in a radiation field for MCNP. Using this software, 3-dimensional dose distributions in a phantom irradiated by a certain mono-energetic source can be deduced through its graphical user interface (GUI). In this study, we extended DynamicMC to be used in combination with the PHITS by providing it with a higher flexibility for dynamic movement for a less sophisticated anthropomorphic phantom. We anticipate that the present work and the developed open-source tools will be in the interest of nuclear radiation physics community for research and teaching purposes.
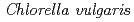 and nano zero-valent iron
and nano zero-valent ironIslam, M. S.*; Maamoun, I.; Falyouna, O.*; Eljamal, O.*; Saha, B. B.*
Journal of Molecular Liquids, 370, p.121005_1 - 121005_11, 2023/01
Times Cited Count:43 Percentile:98.27(Chemistry, Physical) concentration
concentrationOtani, Kyohei; Islam, M. S.*; Sakairi, Masatoshi*; Kaneko, Akira*
Surface and Interface Analysis, 51(12), p.1207 - 1213, 2019/12
Times Cited Count:1 Percentile:1.58(Chemistry, Physical)The corrosion morphology and composition of corrosion products of A3003 formed in model fresh water with different Zn concentrations were investigated by immersion tests combined with surface observations and analysis using an auger electron spectroscope (AES). The cross-sectional AES observations showed that the thickness of the corrosion product layer formed on A3003 decreases with increases in the Zn
concentrations were investigated by immersion tests combined with surface observations and analysis using an auger electron spectroscope (AES). The cross-sectional AES observations showed that the thickness of the corrosion product layer formed on A3003 decreases with increases in the Zn concentration of the model fresh water. A cross-sectional AES point analysis suggested that the corrosion products formed on the A3003 in the Zn
concentration of the model fresh water. A cross-sectional AES point analysis suggested that the corrosion products formed on the A3003 in the Zn containing model fresh water (Zn
containing model fresh water (Zn
 0.1 mM) have a multi-layer structure, and that the inner of Zn-rich layer would have high corrosion protective properties.
0.1 mM) have a multi-layer structure, and that the inner of Zn-rich layer would have high corrosion protective properties.
Islam, M. S.*; Otani, Kyohei; Sakairi, Masatoshi*
ISIJ International, 58(9), p.1616 - 1622, 2018/09
Times Cited Count:7 Percentile:31.22(Metallurgy & Metallurgical Engineering)To elucidate the role of Zn on corrosion of coated steel, the effects of metal cations on the corrosion of carbon steel in the concentrated Cl
on corrosion of coated steel, the effects of metal cations on the corrosion of carbon steel in the concentrated Cl aqueous solutions were studied by immersion tests, surface analysis and electrochemical tests. Among the examined metal cations, Zn
aqueous solutions were studied by immersion tests, surface analysis and electrochemical tests. Among the examined metal cations, Zn showed the significant effect on corrosion inhibition of carbon steel in the Cl
showed the significant effect on corrosion inhibition of carbon steel in the Cl aqueous solution at high concentration. XPS analysis results elucidated that Zn
aqueous solution at high concentration. XPS analysis results elucidated that Zn can remain on the steel surface after immersed in the solutions with Zn
can remain on the steel surface after immersed in the solutions with Zn . EIS measurements showed higher impedance in the solution with Zn
. EIS measurements showed higher impedance in the solution with Zn than other solutions, and the results suggested that Zn
than other solutions, and the results suggested that Zn reduced the defect points in the thin oxide film by forming a metal cation layer. Based on the experimental results, Zn
reduced the defect points in the thin oxide film by forming a metal cation layer. Based on the experimental results, Zn may form a layer on the oxide film that protects the Cl
may form a layer on the oxide film that protects the Cl attack in the solution. The findings demonstrated that the formation of Zn layer on the oxide film is one of the main reason for showing high and longtime corrosion resistance of Zn coated steel substrate.
attack in the solution. The findings demonstrated that the formation of Zn layer on the oxide film is one of the main reason for showing high and longtime corrosion resistance of Zn coated steel substrate.
 aqueous solution
aqueous solutionIslam, M. S.*; Otani, Kyohei; Sakairi, Masatoshi*
Corrosion Science, 140, p.8 - 17, 2018/08
Times Cited Count:17 Percentile:59.10(Materials Science, Multidisciplinary)The corrosion characteristics of SUS304 exposed to 0.5M Cl aqueous solution containing different metal cations were studied with immersion tests, surface analysis and electrochemical tests. The mechanism of corrosion with metal cations was clarified by the XPS analysis results together with the hard and soft acid and base (HSAB) concept and the passive films structure. It is supposed that metal cations with large hardness make a layer by chemical bonding with the passive films. The passive films are protected by the metal cation layer from Cl
aqueous solution containing different metal cations were studied with immersion tests, surface analysis and electrochemical tests. The mechanism of corrosion with metal cations was clarified by the XPS analysis results together with the hard and soft acid and base (HSAB) concept and the passive films structure. It is supposed that metal cations with large hardness make a layer by chemical bonding with the passive films. The passive films are protected by the metal cation layer from Cl attack, and consequently corrosion reactions are inhibited.
attack, and consequently corrosion reactions are inhibited.
 in the radioresistant bacterium
in the radioresistant bacterium 
Islam, M. S.*; Hua, Y.*; Oba, Hirofumi; Sato, Katsuya; Kikuchi, Masahiro; Yanagisawa, Tadashi*; Narumi, Issei
Genes and Genetic Systems, 78(5), p.319 - 327, 2003/10
Times Cited Count:16 Percentile:30.52(Biochemistry & Molecular Biology)no abstracts in English
Islam, M. S.*; Terada, Atsuhiko*; Kinoshita, Hidetaka; Hino, Ryutaro; Monde, Masanori*
JAERI-Tech 2001-044, 49 Pages, 2001/07
no abstracts in English
Sakairi, Masatoshi*; Otani, Kyohei; Islam, M. S.*
no journal, ,
Previous studies showed that zinc ion shows higher corrosion resistant property on corrosion of metals in aqueous environment. It is, however, not fully elucidate role of zinc ions on corrosion inhibition of metals in aqueous environments. In this study, the effects of zinc ion on corrosion behavior of aluminum alloy and steels in aqueous environments. From electrochemical and surface analysis results, zinc ion may forms a very effective shielding film which can block the electrochemical reactions, consequently lowers the corrosion rate.
Islam, M. S.*; Otani, Kyohei; Sakairi, Masatoshi*
no journal, ,
In the previous study, it was founded that Zn significantly inhibited the corrosion of mild steel in Cl
significantly inhibited the corrosion of mild steel in Cl aqueous solution at room temperature. It is still not fully elucidated the corrosion inhibition ability of Zn
aqueous solution at room temperature. It is still not fully elucidated the corrosion inhibition ability of Zn concentration on the steel in Cl
concentration on the steel in Cl aqueous solutions. In the present study, the effects of Zn
aqueous solutions. In the present study, the effects of Zn concentration on the corrosion inhibition of mild steel were investigated by immersion tests. From the immersion tests, it was found that mass change of specimen closely related to the Zn
concentration on the corrosion inhibition of mild steel were investigated by immersion tests. From the immersion tests, it was found that mass change of specimen closely related to the Zn concentration in the solutions. SEM images showed that the numbers of pits are decreasing with increasing the Zn
concentration in the solutions. SEM images showed that the numbers of pits are decreasing with increasing the Zn concentration. XPS and AES results showed that Zn
concentration. XPS and AES results showed that Zn existed as hydroxides by forming a layer with the surface film and the coverage of the layer is increased with increase in Zn
existed as hydroxides by forming a layer with the surface film and the coverage of the layer is increased with increase in Zn concentration.
concentration.
Oue, Atsushi*; Shimizu, Nobuaki*; Tanaka, Atsushi*; Otsuki, Takahiro*; Shinagawa, Masahiko*; Mori, Takahisa*; Saha, M. N.*; Ariful, H. S.*; Islam, S.*; Nakamura, Takako*; et al.
no journal, ,
no abstracts in English