Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Cs solid-state NMR and density functional theory calculations
Cs solid-state NMR and density functional theory calculationsOkubo, Takahiro*; Okamoto, Takuya*; Kawamura, Katsuyuki*; Gu gan, R.*; Deguchi, Kenzo*; Oki, Shinobu*; Shimizu, Tadashi*; Tachi, Yukio; Iwadate, Yasuhiko*
gan, R.*; Deguchi, Kenzo*; Oki, Shinobu*; Shimizu, Tadashi*; Tachi, Yukio; Iwadate, Yasuhiko*
Journal of Physical Chemistry A, 122(48), p.9326 - 9337, 2018/12
Times Cited Count:13 Percentile:60.51(Chemistry, Physical)The structures of Cs adsorption on montmorillonite were investigated by the nuclear magnetic resonance (NMR) spectroscopy. The NMR spectra of Cs adsorbed on montmorillonite samples were measured under different Cs contents and relative humidity levels. NMR parameters were evaluated by the first principle calculations in order to identify the relationship between adsorbed Cs structures and NMR parameters. The comparisons between experimental and theoretical NMR spectra revealed that Cs is preferentially adsorbed at sites near Al for low Cs substituted montmorillonites, and that non-hydrated Cs present in partially Cs substituted samples, even after being hydrated under high relative humidity.
Okubo, Takahiro*; Ibaraki, Moe*; Tachi, Yukio; Iwadate, Yasuhiko*
Applied Clay Science, 123, p.148 - 155, 2016/04
Times Cited Count:29 Percentile:74.44(Chemistry, Physical)The pore distribution of water-saturated compacted clay (Na-montmorillonite at 0.8 and 1.4 g/cm saturated by three salt concentrations) was evaluated using
saturated by three salt concentrations) was evaluated using  H NMR relaxometry and freezing point depression. The populations of interlayer water with four hydrated state and non-interlayer water were calculated from the assumed thresholds. The sample with lower density exhibits higher population of non-interlayer water up to 55%. Low-temperature
H NMR relaxometry and freezing point depression. The populations of interlayer water with four hydrated state and non-interlayer water were calculated from the assumed thresholds. The sample with lower density exhibits higher population of non-interlayer water up to 55%. Low-temperature  H NMR experiments in view of freezing point depression indicated that mesopore water in approximately 4 nm space observed in the calorimetric study was considered as non-interlayer water and the threshold temperature. The result showed that population of non-interlayer water by expected from freezing point depression agreed with
H NMR experiments in view of freezing point depression indicated that mesopore water in approximately 4 nm space observed in the calorimetric study was considered as non-interlayer water and the threshold temperature. The result showed that population of non-interlayer water by expected from freezing point depression agreed with  H NMR relaxometry within 10%. Correlation experiments between longitudinal (
H NMR relaxometry within 10%. Correlation experiments between longitudinal (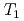 ) and transverse relation times (
) and transverse relation times (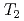 ) at -10
) at -10 C suggested that high-mobility bulk-like water molecules existed at a clay density of 1.4 g/cm
C suggested that high-mobility bulk-like water molecules existed at a clay density of 1.4 g/cm .
.

Okamoto, Yoshihiro; Iwadate, Yasuhiko*; Fukushima, Kazuko*; Matsuura, Haruaki*; Minato, Kazuo
Journal of Physics and Chemistry of Solids, 66(2-4), p.452 - 455, 2005/02
Times Cited Count:6 Percentile:30.12(Chemistry, Multidisciplinary)The structure of molten PbCl was investigated by using X-ray diffrtaction(XRD) and X-ray absorption fine structure(XAFS) analysis techniques. From the fourier transformation analysis if the XRD data, the nearest Pb
was investigated by using X-ray diffrtaction(XRD) and X-ray absorption fine structure(XAFS) analysis techniques. From the fourier transformation analysis if the XRD data, the nearest Pb -Cl
-Cl interaction consists of two kinds of interactions; rigid 4-fold tetrahedron and 2-3 loose coordination. The XAFS result shows a local structure is the 4-fold coordination in the melt.
interaction consists of two kinds of interactions; rigid 4-fold tetrahedron and 2-3 loose coordination. The XAFS result shows a local structure is the 4-fold coordination in the melt.
Okamoto, Yoshihiro; Fukushima, Kazuko*; Iwadate, Yasuhiko*
Journal of Non-Crystalline Solids, 312(314), p.450 - 453, 2002/10
Local structure of molten ZnBr was investigated by both of the Zn and the Br XAFS (X-ray absorption fine structure) measurements above their K-absorption edge at 723 K. The nearest Zn
was investigated by both of the Zn and the Br XAFS (X-ray absorption fine structure) measurements above their K-absorption edge at 723 K. The nearest Zn -Br
-Br distance was determined to be 2.46
distance was determined to be 2.46  for the Zn XAFS and 2.45
for the Zn XAFS and 2.45  for the Br XAFS. The coordination number of Br
for the Br XAFS. The coordination number of Br ion around Zn
ion around Zn was about 4. On the other hand, that of Zn
was about 4. On the other hand, that of Zn ion around Br
ion around Br was about 2. The 1st Br
was about 2. The 1st Br -Br
-Br distance was estimated to be 3.9
distance was estimated to be 3.9  . They shows that a tetrahedral coordinate (ZnBr
. They shows that a tetrahedral coordinate (ZnBr )
) exists and most of the coordinate connects with the next ones through Br
exists and most of the coordinate connects with the next ones through Br ion. The two XAFS functions in the molten ZnBr
ion. The two XAFS functions in the molten ZnBr were reproduced by the combination of the molecular dynamics(MD) simulation and the FEFF7 computations.
were reproduced by the combination of the molecular dynamics(MD) simulation and the FEFF7 computations.
Myochin, Munetaka; Kofuji, Hirohide; Yamana, Hajimu*; Shirai, Osamu*; Yamamura, Tsutomu*; Umesaki, Norimasa*; Matsuura, Haruaki*; Kajinami, Akihiko*; Iwadate, Yasuhiko*; Otori, Norikazu*; et al.
no journal, ,
no abstracts in English
Okubo, Takahiro*; Ibaraki, Moe*; Tachi, Yukio; Iwadate, Yasuhiko*
no journal, ,
1H NMR relaxometry was applied for investigation of pore structure in compacted saturated montmorillonite with different salt concentration. The samples compacted to dry densities of 0.8 and 1.2 g/cm were saturated with pure water, 0.05, 0.10, 0.50, and 1.0 M NaCl solutions. Population of water in different hydrated layer corresponding to 1-, 2-, and 3 hydrated layer was estimated from longitudinal relaxation distributions. The population of water in 1-, 2-, and 3 hydrated layers changed as a function of NaCl concentration. The threshold to discriminate between inter- and intra-layer water is 3 hydrated layer, which is questionable from existence of 4 hydrated layer revealed by modelling of X-ray profile. The amount of water in 1- and intra-layer were negligible small for all conditions at dry density of 1.2 g/cm
were saturated with pure water, 0.05, 0.10, 0.50, and 1.0 M NaCl solutions. Population of water in different hydrated layer corresponding to 1-, 2-, and 3 hydrated layer was estimated from longitudinal relaxation distributions. The population of water in 1-, 2-, and 3 hydrated layers changed as a function of NaCl concentration. The threshold to discriminate between inter- and intra-layer water is 3 hydrated layer, which is questionable from existence of 4 hydrated layer revealed by modelling of X-ray profile. The amount of water in 1- and intra-layer were negligible small for all conditions at dry density of 1.2 g/cm . On the other hand, water in 2-hydrated layer was decreased instead of increment of water in 3-hydrated layer.
. On the other hand, water in 2-hydrated layer was decreased instead of increment of water in 3-hydrated layer.
 Cs solid state NMR, 1; GIPAW prediction of
Cs solid state NMR, 1; GIPAW prediction of  Cs NMR parameters
Cs NMR parametersOkubo, Takahiro*; Iwadate, Yasuhiko*; Tachi, Yukio
no journal, ,
The structures of Cs adsorption on montmorillonite were investigated by the nuclear magnetic resonance (NMR) spectroscopy. NMR parameters were evaluated by the first principle calculations using GIPAW method in order to identify the relationship between adsorbed Cs structures and NMR parameters. It was confirmed that the chemical shift for Cs adsorbed on Al site in tetrahedral sheet was shifted to low magnetic field.
 Cs solid state NMR, 2;
Cs solid state NMR, 2;  Cs MAS and 3Q MAS NMR
Cs MAS and 3Q MAS NMROkamoto, Takuya*; Okubo, Takahiro*; Iwadate, Yasuhiko*; Deguchi, Kenzo*; Shimizu, Tadashi*; Oki, Shinobu*; Tachi, Yukio
no journal, ,
The structures of Cs adsorption on montmorillonite were investigated by the nuclear magnetic resonance (NMR) spectroscopy. The NMR spectra of Cs adsorbed montmorillonite samples with different Cs contents were collected by  Cs Magic Angle Spinning (MAS) NMR and
Cs Magic Angle Spinning (MAS) NMR and  Cs Triple Quantum (3Q) MAS NMR. The adsorbed Cs structures were evaluated based on the correlations between the structural characteristics and the values of the NMR parameters predicted by the first principal calculations.
Cs Triple Quantum (3Q) MAS NMR. The adsorbed Cs structures were evaluated based on the correlations between the structural characteristics and the values of the NMR parameters predicted by the first principal calculations.
Okamoto, Takuya*; Okubo, Takahiro*; Iwadate, Yasuhiko*; Deguchi, Kenzo*; Shimizu, Tadashi*; Oki, Shinobu*; Tachi, Yukio
no journal, ,
The structures of Cs adsorbed on montmorillonite were investigated by  Cs Magic Angle Spinning (MAS) NMR spectroscopy. The NMR spectra of Cs adsorbed on montmorillonite samples were measured under different relative humidity levels and were analyzed to evaluate the change in the structure of hydrated Cs sorbed on montmorillonite.
Cs Magic Angle Spinning (MAS) NMR spectroscopy. The NMR spectra of Cs adsorbed on montmorillonite samples were measured under different relative humidity levels and were analyzed to evaluate the change in the structure of hydrated Cs sorbed on montmorillonite.
Yamazaki, Akio*; Okubo, Takahiro*; Iwadate, Yasuhiko*; Tachi, Yukio
no journal, ,
A structural analysis of water-filled pores in saturated compacted sodium montmorillonite was conducted by 1H NMR spectroscopy. The pore structure including interlayer and intraparticle pores in compacted montmorillonite was characterized by relaxation time and freezing point observed by 1H NMR and their correlations with water-filled pore sizes.
Yamazaki, Akio*; Okubo, Takahiro*; Iwadate, Yasuhiko*; Tachi, Yukio
no journal, ,
A structural analysis of water-filled pores in saturated compacted Ca-type montmorillonite was conducted by 1H NMR spectroscopy. The pore structure including interlayer and intraparticle pores in Ca-type compacted montmorillonite was characterized by relaxation time observed by 1H NMR and their correlations with water-filled pore sizes.
Sakata, Nagisa*; Tachi, Yukio; Iwadate, Yasuhiko*; Okubo, Takahiro*
no journal, ,
Cement-based materials will be used as a component for radioactive waste disposal facility. Understanding water paths in cement-based materials is a key to evaluate their long-term performance. It is well known that 1H transverse relaxation (T2) of water filled pores is related to their porous structure. In this study, 1H T2 distribution analyses were applied to hydration process of cement-based materials, and the relationship between porous structure and mass transport was discussed. Experimental results showed that the main path of water is the grain boundary between cement and silica sand contained in mortar. In addition, C-S-H gel layers in cement and C-S-H gel pore cannot contribute as a path of water.