Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Matsumoto, Atsushi; Kamata, Tetsuji*; Takagi, Junichi*; Iwasaki, Kenji*; Yura, Kei
Biophysical Journal, 95(6), p.2895 - 2908, 2008/09
Times Cited Count:19 Percentile:44.72(Biophysics)Protein is activated, but the activation mechanism and generality of the conformational change remain to be elucidated. We performed normal mode analysis of the elastic network model on integrin 
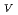

 ectodomain in the bent form and identified key residues which were influential on molecular motions. Iterative normal mode calculations demonstrated that the specific non-bonded interactions involving the key residues work as a snap to keep integrin in the bent form. The importance of the key residues for the conformational change was further verified by mutation experiments. Conservation pattern of amino acid residues among integrin family showed that the characteristic pattern of residues seen around these key residues is found in the limited groups of integrin
ectodomain in the bent form and identified key residues which were influential on molecular motions. Iterative normal mode calculations demonstrated that the specific non-bonded interactions involving the key residues work as a snap to keep integrin in the bent form. The importance of the key residues for the conformational change was further verified by mutation experiments. Conservation pattern of amino acid residues among integrin family showed that the characteristic pattern of residues seen around these key residues is found in the limited groups of integrin  chains.
chains.
Matsumoto, Atsushi; Kamata, Tetsuji*; Iwasaki, Kenji*; Takagi, Junichi*; Yura, Kei
no journal, ,
Integrin, a membrane protein with a huge extracellular domain, participates in cell-cell and cell-extracellular matrix interactions for metazoan. A group of integrins are known to perform a large-scale structural change when they are activated, but their mechanism and generality of the conformational change in other integrins remain to be elucidated. We performed normal mode analysis based on the elastic network models of integrin in the bent form to identify key residues dominating the molecular motions toward activation. We simulated the large conformational change of integrin from the bent to the extended forms by iterative normal mode calculations and demonstrated that specific non-bonded interactions involving the key residues work as a snap to lock integrin in the bent form. The importance of the key residues for the conformational change was verified by mutation experiments.