Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
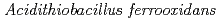
Kanao, Tadayoshi*; Kosaka, Megumi*; Yoshida, Kyoya*; Nakayama, Hisayuki*; Tamada, Taro; Kuroki, Ryota; Yamada, Hidenori; Takada, Jun*; Kamimura, Kazuo*
Acta Crystallographica Section F, 69(6), p.692 - 694, 2013/06
Times Cited Count:10 Percentile:59.68(Biochemical Research Methods)Tetrathionate hydrolase (4THase) from the iron- and sulfur-oxidizing bacterium 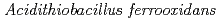 catalyses the disproportionate hydrolysis of tetrathionate to elemental sulfur, thiosulfate and sulfate. The gene encoding 4THase (
catalyses the disproportionate hydrolysis of tetrathionate to elemental sulfur, thiosulfate and sulfate. The gene encoding 4THase (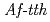 ) was expressed as inclusion bodies in recombinant
) was expressed as inclusion bodies in recombinant 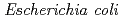 . Recombinant
. Recombinant 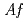 -Tth was activated by refolding under acidic conditions and was then purified to homogeneity. The recombinant protein was crystallized in 20 m
-Tth was activated by refolding under acidic conditions and was then purified to homogeneity. The recombinant protein was crystallized in 20 m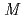 glycine buffer pH 10 containing 50 m
glycine buffer pH 10 containing 50 m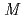 sodium chloride and 33%(
sodium chloride and 33%(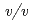 ) PEG 1000 using the hanging-drop vapour-diffusion method. The crystal was a hexagonal cylinder with dimensions of 0.2
) PEG 1000 using the hanging-drop vapour-diffusion method. The crystal was a hexagonal cylinder with dimensions of 0.2  0.05
0.05  0.05 mm. X-ray crystallographic analysis showed that the crystal diffracted to 2.15
0.05 mm. X-ray crystallographic analysis showed that the crystal diffracted to 2.15  resolution and belongs to space group
resolution and belongs to space group 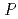 3
3 or
or 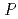 3
3 , with unit-cell parameters
, with unit-cell parameters 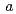 =
= 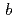 = 92.1,
= 92.1,  = 232.6
= 232.6  .
.
Kosaka, Megumi*; Kanao, Tadayoshi*; Nakayama, Hisayuki*; Yoshida, Kyoya*; Kamimura, Kazuo*; Takada, Jun*; Yamada, Hidenori; Tamada, Taro; Okazaki, Nobuo*; Kuroki, Ryota
no journal, ,
no abstracts in English