Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Beerwerth, R.*; Kaneya, Yusuke*; Makii, Hiroyuki; Mitsukai, Akina*; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; et al.
Journal of the American Chemical Society, 140(44), p.14609 - 14613, 2018/11
Times Cited Count:32 Percentile:69.40(Chemistry, Multidisciplinary)The first ionization potential (IP ) yields information on valence electronic structure of an atom. IP
) yields information on valence electronic structure of an atom. IP values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP
values of heavy actinides beyond einsteinium (Es, Z = 99), however, have not been determined experimentally so far due to the difficulty in obtaining these elements on scales of more than one atom at a time. Recently, we successfully measured IP of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP
of lawrencium (Lr, Z = 103) using a surface ionization method. The result suggests that Lr has a loosely-bound electron in the outermost orbital. In contrast to Lr, nobelium (No, Z = 102) is expected to have the highest IP among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP
among the actinide elements owing to its full-filled 5f and the 7s orbitals. In the present study, we have successfully determined IP values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP
values of No as well as fermium (Fm, Z = 100) and mendelevium (Md, Z = 101) using the surface ionization method. The obtained results indicate that the IP value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
value of heavy actinoids would increase monotonically with filling electrons up in the 5f orbital like heavy lanthanoids.
 SO
SO and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)
and HF/HCl solutions into toluene with Aliquat336; Sulfate and fluoride complex formation of Mo and W towards chemical studies of seaborgium (Sg)Toyoshima, Atsushi; Mitsukai, Akina; Tsukada, Kazuaki; Oe, Kazuhiro*; Haba, Hiromitsu*; Komori, Yukiko*; Murakami, Masashi; Kaneya, Yusuke*; Sato, Daisuke*; Asai, Masato; et al.
Journal of Radioanalytical and Nuclear Chemistry, 317(1), p.421 - 430, 2018/07
Times Cited Count:3 Percentile:25.46(Chemistry, Analytical)We have studied extraction behavior of group-6 elements Mo and W to search for suitable conditions for an on-line extraction experiment of their heavier homolog, seaborgium (Sg). Batch-wise extraction of carrier-free radiotracers  Mo and
Mo and 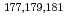 W were carried out from 0.10 - 8.6 M H
W were carried out from 0.10 - 8.6 M H SO
SO and 1.0
and 1.0 10
10 - 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H
- 5.0 M HF/1.0 M HCl into toluene with a quaternary ammonium compound, Aliquat336. Anionic sulfate complexes of Mo and W with charge - 2 were extracted with Aliquat336 from H SO
SO solutions with concentrations of [H
solutions with concentrations of [H SO
SO ]
]  2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
2 M. In HF/1.0 M HCl, oxyfluoro complexes of Mo and W with charge - 1 were interpreted to be formed and extracted with Aliquat336. From these results, favorable conditions for the extraction of Sg are discussed.
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Stora, T.*; Sato, Nozomi*; Kaneya, Yusuke; Tsukada, Kazuaki; D llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
llmann, C. E.*; Eberhardt, K.*; Eliav, E.*; et al.
EPJ Web of Conferences, 131, p.05001_1 - 05001_6, 2016/12
Times Cited Count:0 Percentile:0.00(Chemistry, Inorganic & Nuclear)Ionization efficiency in a surface ionization process depends on the first ionization potential of the atom. Based on the dependence, the ionization potential of the atom can be determined. We measured ionization efficiencies of fermium, einsteinium, mendelevium, and lawrencium by using a newly developed gas-jet coupled surface ion-source. The ionization potential of the elements have not been determined so far due to their low production rates and/or their short half-lives. Based on a relationship between the ionization efficiency and the ionization potential obtained via measurements of short-lived lanthanide isotopes, the ionization potentials of these actinide elements have been successfully determined.
Eichler, R.*; Asai, Masato; Brand, H.*; Chiera, N. M.*; Di Nitto, A.*; Dressler, R.*; D llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
llmann, Ch. E.*; Even, J.*; Fangli, F.*; Goetz, M.*; et al.
EPJ Web of Conferences, 131, p.07005_1 - 07005_7, 2016/12
Times Cited Count:3 Percentile:71.15(Chemistry, Inorganic & Nuclear)In recent years gas-phase chemical studies assisted by physical pre-separation allowed for the productions and investigations of fragile single molecular species of superheavy elements. The latest highlight is the formation of very volatile hexacarbonyl compound of element 106, Sg(CO) . Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO)
. Following this success, second-generation experiments were performed to measure the first bond dissociation energy between the central metal atom and the surrounding ligand. The method using a tubular decomposition reactor was developed and successfully applied to short-lived Mo(CO) , W(CO)
, W(CO) , and Sg(CO)
, and Sg(CO) .
.
 at the single-atom level
at the single-atom levelSteinegger, P.*; Asai, Masato; Dressler, R.*; Eichler, R.*; Kaneya, Yusuke*; Mitsukai, Akina*; Nagame, Yuichiro; Piguet, D.*; Sato, Tetsuya; Sch del, M.; et al.
del, M.; et al.
Journal of Physical Chemistry C, 120(13), p.7122 - 7132, 2016/04
Times Cited Count:28 Percentile:63.01(Chemistry, Physical)A new experimental method "vacuum chromatography" has been developed to measure adsorption enthalpy of superheavy elements, and its feasibility has been examined using short-lived thallium isotopes. The short-lived thallium isotopes were produced at the JAEA tandem accelerator. The thallium ion beam prepared with an on-line isotope separator which ionized and mass-separated the thallium isotopes was injected into an isothermal vacuum chromatography apparatus. A temperature-dependent adsorption property of thallium atom on SiO surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
surface were measured. The adsorption enthalpy of thallium was determined to be 158 kJ/mol. The thallium is a homolog of element 113. Thus, the vacuum chromatography developed in this study enables us to perform chemical experiments for short-lived superheavy elements with half-lives of a order of one second.
 and W(CO)
and W(CO)
Usoltsev, I.*; Eichler, R.*; Wang, Y.*; Even, J.*; Yakushev, A.*; Haba, Hiromitsu*; Asai, Masato; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; et al.
llmann, Ch. E.*; et al.
Radiochimica Acta, 104(3), p.141 - 151, 2016/03
Times Cited Count:34 Percentile:94.30(Chemistry, Inorganic & Nuclear)Conditions of the production and decomposition of hexacarbonyl complexes of short-lived Mo and W isotopes were investigated to study thermal stability of the heaviest group 6 hexacarbonyl complex Sg(CO) . A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO)
. A tubular flow reactor was tested to decompose the hexacarbonyl complexes and to extract the first bond dissociation energies. A silver was found to be the most appropriate reaction surface to study the decomposition of the group 6 hexacarbonyl. It was found that the surface temperature at which the decomposition occurred was correlated to the first bond dissociation energy of Mo(CO) and W(CO)
and W(CO) , indicating that the first bond dissociation energy of Sg(CO)
, indicating that the first bond dissociation energy of Sg(CO) could be determined with this technique.
could be determined with this technique.
Sato, Tetsuya; Asai, Masato; Borschevsky, A.*; Stora, T.*; Sato, Nozomi; Kaneya, Yusuke; Tsukada, Kazuaki; D llmann, Ch. E.*; Eberhardt, K.*; Eliav, E.*; et al.
llmann, Ch. E.*; Eberhardt, K.*; Eliav, E.*; et al.
Nature, 520(7546), p.209 - 211, 2015/04
Times Cited Count:112 Percentile:97.06(Multidisciplinary Sciences)Ionization efficiency in a surface ionization process depends on the first ionization potential of the atom. Based on the dependence, the ionization potential of the atom can be determined. We successfully measured ionization efficiencies of lawrencium (Lr, 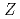 =103) using a gas-jet coupled surface ion-source. The ionization potential of Lr has not been determined owing to its low production rate and its short half-life. Based on a relationship between the ionization efficiency and the ionization potential obtained via measurements of short-lived lanthanide isotopes, the ionization potential of Lr was determined.
=103) using a gas-jet coupled surface ion-source. The ionization potential of Lr has not been determined owing to its low production rate and its short half-life. Based on a relationship between the ionization efficiency and the ionization potential obtained via measurements of short-lived lanthanide isotopes, the ionization potential of Lr was determined.
Even, J.*; Ackermann, D.*; Asai, Masato; Block, M.*; Brand, H.*; Di Nitto, A.*; D llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
llmann, Ch. E.*; Eichler, R.*; Fan, F.*; Haba, Hiromitsu*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(3), p.2457 - 2466, 2015/03
Times Cited Count:15 Percentile:73.95(Chemistry, Analytical)Rapid In situ synthesis of metal carbonyl complexes has been demonstrated using short-lived isotopes produced in nuclear fission and fusion reactions. The short-lived isotopes with high recoil energy directly react with carbon-monoxides and form carbonyl complexes. Only highly volatile complexes were fast transported in a gas stream to counting and chemistry devices. Short-lived Mo, Tc, Ru, Rh, W, Re, Os, and Ir were found to form volatile carbonyl complexes, while no volataile complex of Hf and Ta were detected. This technique has been applied to a chemical investigation of the superheavy element Sg (atomic number 106), and will be applicable to various fields of nuclear science with short-lived transition metal isotopes.
 gas-jet system coupled to a surface-ionization type ion-source in JAEA-ISOL; Towards determination of the first ionization potential of Lr (Z = 103)
gas-jet system coupled to a surface-ionization type ion-source in JAEA-ISOL; Towards determination of the first ionization potential of Lr (Z = 103)Sato, Tetsuya; Asai, Masato; Sato, Nozomi; Tsukada, Kazuaki; Toyoshima, Atsushi; Oe, Kazuhiro*; Miyashita, Sunao*; Kaneya, Yusuke; Osa, Akihiko; Sch del, M.; et al.
del, M.; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1253 - 1257, 2015/02
Times Cited Count:9 Percentile:56.39(Chemistry, Analytical)We have developed a surface-ionization ion-source coupled to the He/CdI gas-jet transport system for the Isotope Separator On-Line (ISOL) at the JAEA tandem accelerator for experimental determination of the first ionization potential of lawrencium (Lr,
gas-jet transport system for the Isotope Separator On-Line (ISOL) at the JAEA tandem accelerator for experimental determination of the first ionization potential of lawrencium (Lr, 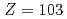 ). We performed to ionize a short-lived Lr isotope and various lanthanide isotopes. We successfully observed mass-separated ions of
). We performed to ionize a short-lived Lr isotope and various lanthanide isotopes. We successfully observed mass-separated ions of  Lr by using our present system at the first time. The first ionization potential of Lr was evaluated based on a correlation between of effective ionization potential and ionization efficiency of short-lived lanthanide isotopes in our system.
Lr by using our present system at the first time. The first ionization potential of Lr was evaluated based on a correlation between of effective ionization potential and ionization efficiency of short-lived lanthanide isotopes in our system.
Oe, Kazuhiro*; Attallah, M. F.*; Asai, Masato; Goto, Naoya*; Gupta, N. S.*; Haba, Hiromitsu*; Huang, M.*; Kanaya, Jumpei*; Kaneya, Yusuke*; Kasamatsu, Yoshitaka*; et al.
Journal of Radioanalytical and Nuclear Chemistry, 303(2), p.1317 - 1320, 2015/02
Times Cited Count:10 Percentile:60.46(Chemistry, Analytical)A new technique for continuous dissolution of nuclear reaction products transported by a gas-jet system was developed for superheavy element (SHE) chemistry. In this technique, a hydrophobic membrane is utilized to separate an aqueous phase from the gas phase. With this technique, the dissolution efficiencies of short-lived radionuclides of 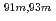 Mo and
Mo and 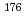 W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
W were measured. Yields of more than 80% were observed for short-lived radionuclides at aqueous-phase flow rates of 0.1-0.4 mL/s. The gas flow-rate had no influence on the dissolution efficiency within the studied flow range of 1.0-2.0 L/min. These results show that this technique is applicable for on-line chemical studies of SHEs in the liquid phase.
Even, J.*; Yakushev, A.*; D llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
llmann, Ch. E.*; Haba, Hiromitsu*; Asai, Masato; Sato, Tetsuya; Brand, H.*; Di Nitto, A.*; Eichler, R.*; Fan, F. L.*; et al.
Science, 345(6203), p.1491 - 1493, 2014/09
Times Cited Count:67 Percentile:82.96(Multidisciplinary Sciences)A new superheavy element complex, a seaborgium carbonyl, has been successfully synthesized, and its adsorption property has been studied using a cryo-thermochromatography and  -detection apparatus COMPACT. Nuclear reaction products of short-lived
-detection apparatus COMPACT. Nuclear reaction products of short-lived  Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of
Sg preseparated with a gas-filled recoil ion separator GARIS at RIKEN were directly injected into a gas cell filled with He/CO mixture gas, and chemical reaction products of volatile carbonyl complexes were trasported to COMPACT. The Sg carbonyl complex detected with COMPACT was found to be very volatile with adsorption enthalpy of  50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO)
50 kJ/mol, from which we have concluded that this complex should be a Sg hexacarbonyl Sg(CO) . This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
. This is the first synthesis of organometallic compounds of transactinide elements for which only simple inorganic comounds have been synthesized so far.
 /Md
/Md reduction potential studied with flow electrolytic chromatography
reduction potential studied with flow electrolytic chromatographyToyoshima, Atsushi; Li, Z.*; Asai, Masato; Sato, Nozomi; Sato, Tetsuya; Kikuchi, Takahiro; Kaneya, Yusuke; Kitatsuji, Yoshihiro; Tsukada, Kazuaki; Nagame, Yuichiro; et al.
Inorganic Chemistry, 52(21), p.12311 - 12313, 2013/11
Times Cited Count:8 Percentile:33.87(Chemistry, Inorganic & Nuclear)The reduction behavior of mendelevium (Md) was studied using a flow electrolytic chromatography apparatus. By applying appropriate potentials on the chromatography column, the more stable Md is reduced to Md
is reduced to Md . The reduction potential of the Md
. The reduction potential of the Md + e
+ e
 Md
Md couple was determined to be -0.16
couple was determined to be -0.16 0.05 V vs. a normal hydrogen electrode.
0.05 V vs. a normal hydrogen electrode.
Sato, Tetsuya; Sato, Nozomi; Asai, Masato; Tsukada, Kazuaki; Toyoshima, Atsushi; Oe, Kazuhiro; Miyashita, Sunao; Sch del, M.; Kaneya, Yusuke*; Nagame, Yuichiro; et al.
del, M.; Kaneya, Yusuke*; Nagame, Yuichiro; et al.
Review of Scientific Instruments, 84(2), p.023304_1 - 023304_5, 2013/02
Times Cited Count:17 Percentile:58.20(Instruments & Instrumentation)We have developed the surface ion-source coupled to the He/CdI gas-jet transport system to measure the ionization potential of Lr atatom-at-a-time conditions. We successfully ionized and mass-separated for the first time
gas-jet transport system to measure the ionization potential of Lr atatom-at-a-time conditions. We successfully ionized and mass-separated for the first time  Lr ions by applying the present ion-source and the ISOL technique. The ionization efficiencies of Lr were estimated to be approximately 42% and 24% at 2600 K on Re and Ta surfaces, respectively. These values were higher than those of Lu in all of ionization condition. The results indicate that the ionization potential of Lr would be lower than that of Lu, 5.4 eV. Therefore, it is concluded that the surface ion-source is a promising apparatus tomeasure the first ionization potential of Lr. Using the present system, determination of the ionization potential of Lr is being performed.
Lr ions by applying the present ion-source and the ISOL technique. The ionization efficiencies of Lr were estimated to be approximately 42% and 24% at 2600 K on Re and Ta surfaces, respectively. These values were higher than those of Lu in all of ionization condition. The results indicate that the ionization potential of Lr would be lower than that of Lu, 5.4 eV. Therefore, it is concluded that the surface ion-source is a promising apparatus tomeasure the first ionization potential of Lr. Using the present system, determination of the ionization potential of Lr is being performed.
 I and
I and 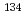 Cs in soil samples and a simplified calculation of cascade summing corrections for volume source
Cs in soil samples and a simplified calculation of cascade summing corrections for volume sourceAsai, Masato; Kaneya, Yusuke*; Sato, Tetsuya; Tsukada, Kazuaki; Oe, Kazuhiro; Sato, Nozomi; Toyoshima, Atsushi
Journal of Nuclear and Radiochemical Sciences (Internet), 12(1), p.5 - 10, 2012/06
To measure radioactivities in soil contaminated by the accident of the Fukushima Daiichi Nuclear Power Plant, efficiency calibration of Ge detectors for  I,
I, 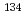 Cs, and
Cs, and  Cs in volume sources was investigated.
Cs in volume sources was investigated.  -ray detection efficiencies for these nuclei were determined precisely using standard soil samples containing
-ray detection efficiencies for these nuclei were determined precisely using standard soil samples containing 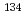 Cs,
Cs,  Cs,
Cs, 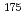 Hf, and
Hf, and  Zr. These standard sources were prepared by admixing radioactive solutions with soil, and point sources were made from the same solutions to determine their radioactivity concentrations. Cascade summing corrections for
Zr. These standard sources were prepared by admixing radioactive solutions with soil, and point sources were made from the same solutions to determine their radioactivity concentrations. Cascade summing corrections for  rays of
rays of 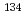 Cs in soil samples were evaluated experimentally. To easily calculate the cascade summing corrections for volume sources, we examined a simplified method using averaged efficiencies, and evaluated its validity through a comparison of the calculated correction factors with the experimental ones.
Cs in soil samples were evaluated experimentally. To easily calculate the cascade summing corrections for volume sources, we examined a simplified method using averaged efficiencies, and evaluated its validity through a comparison of the calculated correction factors with the experimental ones.
Sato, Nozomi; Asai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Toyoshima, Atsushi; Li, Z.*; Kikuchi, Takahiro; Kaneya, Yusuke*; Sch del, M.; Nagame, Yuichiro; et al.
del, M.; Nagame, Yuichiro; et al.
no journal, ,
The first ionization potential (IP) of the last actinide, lawrencium (Lr), is predicted to be lower than those of other actinides. To determine the IP of Lr based on the surface ionization comparison technique, we are developing a surface-ionization-type ion-source coupled to a gas-jet transport system at the Isotope Separator On-Line at the JAEA tandem accelerator facility. In the present study, we measured ionization efficiencies of lanthanides as a function of ion-source temperature to examine the applicability of our apparatus to the IP measurement. It was found that the obtained ionization efficiency of each lanthanide element is affected by an ionization potential as well as a boiling point.
Toyoshima, Atsushi; Li, Z.*; Asai, Masato; Sato, Nozomi; Sato, Tetsuya; Oe, Kazuhiro; Kikuchi, Takahiro; Kaneya, Yusuke*; Kitatsuji, Yoshihiro; Tsukada, Kazuaki; et al.
no journal, ,
Redox potential of mendelevium (Md) was determined with flow electrolytic chromatography. Mendelevium-255 with a half-life of 27 min was produced in the  Cm(
Cm(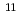 B, 4
B, 4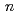 ) reaction at the JAEA tandem accelerator. Reaction products transported by a KCl/He gas-jet method were collected on a chemistry apparatus. After removing KCl with HDEHP column chromatography, elution behavior of Md in 0.10 M HCl was investigated with a flow electrolytic column apparatus at the applied potentials between -0.3 and -0.6 V vs. a Ag/AgCl reference electrode. At -0.3 V, elution behavior of Md was the same as that of
) reaction at the JAEA tandem accelerator. Reaction products transported by a KCl/He gas-jet method were collected on a chemistry apparatus. After removing KCl with HDEHP column chromatography, elution behavior of Md in 0.10 M HCl was investigated with a flow electrolytic column apparatus at the applied potentials between -0.3 and -0.6 V vs. a Ag/AgCl reference electrode. At -0.3 V, elution behavior of Md was the same as that of 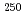 Bk
Bk , showing that the stable Md
, showing that the stable Md was not reduced to the divalent state. At -0.6 V, elution of Md was quite similar to that of Sr
was not reduced to the divalent state. At -0.6 V, elution of Md was quite similar to that of Sr , demonstrating that Md
, demonstrating that Md was successfully reduced to Md
was successfully reduced to Md . From the behavior of Md against the applied potentials, we evaluated its redox potential to be -0.40
. From the behavior of Md against the applied potentials, we evaluated its redox potential to be -0.40 0.03 V.
0.03 V.
Oe, Kazuhiro; Tsukada, Kazuaki; Asai, Masato; Sato, Tetsuya; Toyoshima, Atsushi; Miyashita, Sunao; Nagame, Yuichiro; Sch del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
del, M.; Kaneya, Yusuke*; Lerum, H. V.*; et al.
no journal, ,
We are planning to investigate the redox potentials of element 106, seaborgium, with a combination of the rapid liquid-liquid extraction apparatus SISAK and a flow electrolytic column. In order to combine these two apparatuses, we need to reduce the liquid flow-rate of the SISAK system. In this study, a new degasser, which works with a lower flow rate, was developed for SISAK. It separates the gas-liquid mixture using a hydrophobic Teflon membrane (only the gas can go through the membrane). Using this new degasser, dissolution efficiencies of gas-jet transported products were measured. At the typical flow rate for the SISAK system (0.4 mL/s), a dissolution efficiency of approximately 80% was obtained. A high yield of around 80% was also observed at a flow rate of 0.1 mL/s with a mixer for the gas-liquid mixing.
Sato, Nozomi; Asai, Masato; Tsukada, Kazuaki; Sato, Tetsuya; Toyoshima, Atsushi; Li, Z.*; Oe, Kazuhiro; Kikuchi, Takahiro; Kaneya, Yusuke*; Sch del, M.; et al.
del, M.; et al.
no journal, ,
The first ionization potential (IP) of the last actinide, lawrencium (Lr), is predicted to be lower than those of other actinides. To determine the IP of Lr based on the surface ionization comparison technique, we are developing a surface-ionization-type ion-source coupled to a gas-jet transport system at the Isotope Separator On-Line at the JAEA tandem accelerator facility. In the present study, we measured ionization efficiencies of lanthanides as a function of ion-source temperature to examine the applicability of our apparatus to the IP measurement. It was found that the obtained ionization efficiency of each lanthanide element is affected by an ionization potential as well as a boiling point.
Sato, Tetsuya; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Kasamatsu, Yoshitaka*; Li, Z.; Sato, Nozomi; Kikuchi, Takahiro; Liang, X. H.*; Kaneya, Yusuke*; et al.
no journal, ,
To investigate chemical properties of the superheavy element dubnium (Db, Z = 105), we have so far developed an on-line isothermal gas chromatographic apparatus. In this work, the on-line experiments with group-5 elements, Nb and Ta, using short-lived isotopes 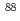 Nb and
Nb and 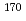 Ta as the homologues of Db were carried out. We determined optimum conditions to produce volatile compounds of the elements. Under the obtained conditions, overall efficiencies of the volatile compounds were measured as a function of the isothermal temperature in chlorinating condition with mixture of nitrogen gas and the SOCl
Ta as the homologues of Db were carried out. We determined optimum conditions to produce volatile compounds of the elements. Under the obtained conditions, overall efficiencies of the volatile compounds were measured as a function of the isothermal temperature in chlorinating condition with mixture of nitrogen gas and the SOCl vapor containing 1% oxygen. Based on the isothermal gas chromatographic behavior of them, it was found that Ta compounds would be less volatile than Nb ones.
vapor containing 1% oxygen. Based on the isothermal gas chromatographic behavior of them, it was found that Ta compounds would be less volatile than Nb ones.
Toyoshima, Atsushi; Oe, Kazuhiro; Asai, Masato; Miyashita, Sunao; Sato, Tetsuya; Sato, Nozomi; Kaneya, Yusuke*; Kitatsuji, Yoshihiro; Tsukada, Kazuaki; Nagame, Yuichiro; et al.
no journal, ,