Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kato, Masato; Maeda, Koji; Ozawa, Takayuki; Kashimura, Motoaki; Kihara, Yoshiyuki
Journal of Nuclear Science and Technology, 48(4), p.646 - 653, 2011/04
Physical properties and irradiation behavior of minor actinide-bearing MOX were evaluated for the development of advanced fast reactor fuels. The physical properties of the fuels were described as functions of minor actinide content and oxygen-to-metal (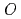 /
/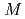 ) ratio, and the effect of minor actinide addition into MOX on those properties was found to be negligibly small. The irradiation tests of fuel pellets having
) ratio, and the effect of minor actinide addition into MOX on those properties was found to be negligibly small. The irradiation tests of fuel pellets having 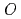 /
/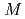 ratios of 1.98 or 1.96 were carried out at high linear heat rate of about 430W/cm. The redistribution of actinide element and oxygen were analyzed by using the evaluated properties, and maximum temperatures of the pellets were estimated. The maximum temperature of the pellets of
ratios of 1.98 or 1.96 were carried out at high linear heat rate of about 430W/cm. The redistribution of actinide element and oxygen were analyzed by using the evaluated properties, and maximum temperatures of the pellets were estimated. The maximum temperature of the pellets of 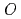 /
/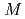 =1.96 was estimated to reach 2680K which was 130K higher than that of
=1.96 was estimated to reach 2680K which was 130K higher than that of 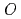 /
/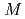 =1.98 pellets. The maximum temperature of the pellet was lower as compared with its melting temperature of higher than 3000K. In the results of post-irradiation examination, no trace of melting was observed.
=1.98 pellets. The maximum temperature of the pellet was lower as compared with its melting temperature of higher than 3000K. In the results of post-irradiation examination, no trace of melting was observed.
 Pu
Pu )O
)O (1.92
(1.92 x
x 2.00) based fuels containing actinides and/or lanthanides
2.00) based fuels containing actinides and/or lanthanidesKomeno, Akira; Kato, Masato; Uno, Hiroki*; Takeuchi, Kentaro; Morimoto, Kyoichi; Kashimura, Motoaki
IOP Conference Series; Materials Science and Engineering, 9, p.012016_1 - 012016_7, 2010/05
Times Cited Count:10 Percentile:94.82(Chemistry, Inorganic & Nuclear)It is expected that the important data for design of fast reactor fuel can be provided by evaluating the relationship between fuel composition and phase separation with reported and new measurement data. According to evaluation with reported data and new measured data, a relationship between fuel composition and phase separation temperature of MOX fuel was indicated. Higher minor actinides-containing MOX had a lower phase separation temperature at O/M ratio region from 1.92 to 1.96.
Kato, Masato; Morimoto, Kyoichi; Komeno, Akira; Nakamichi, Shinya; Kashimura, Motoaki
Proceedings of 10th OECD/NEA Information Exchange Meeting on Actinide and Fission Product Partitioning and Transmutation (CD-ROM), p.201 - 209, 2010/00
Japan Atomic Energy Agency has developed homogeneous MOX fuel containing minor actinide (MA) elements such as Np and Am. To measure physical properties of the fuel is essential for its development, because their data are needed to evaluate irradiation behavior. In this report, the physical properties, melting temperature, thermal conductivity, lattice parameter, oxygen potential and phase separation behavior, were reviewed, and effect of MA content was discussed.
Kato, Masato; Segawa, Tomoomi; Takeuchi, Kentaro; Kashimura, Motoaki; Kihara, Yoshiyuki
Proceedings of International Conference on Advanced Nuclear Fuel Cycle; Sustainable Options & Industrial Perspectives (Global 2009) (CD-ROM), p.2051 - 2058, 2009/09
In the Fast Reactor Cycle technology Development (FaCT) project conducted by Japan Atomic Energy Agency, minor actinide-containing MOX (MA-MOX) fuel has been developed. The fuel is a homogeneous MOX fuel which contains a maximum of 5% MA such as Am and Np. The oxygen-to-metal (O/M) ratio of the fuel is adjusted to less than 1.97 to control fuel and cladding chemical interaction in a high burn-up of 150 GWd/t. In this paper, the thermal properties of raw powder and sintering behavior of oxidized powder were investigated. In addition the O/M ratio adjustment procedure was established.
Kato, Masato; Komeno, Akira; Uno, Hiroki*; Sugata, Hiromasa*; Nakae, Nobuo; Konashi, Kenji*; Kashimura, Motoaki
Journal of Nuclear Materials, 393(1), p.134 - 140, 2009/08
Times Cited Count:44 Percentile:92.68(Materials Science, Multidisciplinary)In plutonium compounds, the lattice parameter increases due to self-radiation damage by  -decay of plutonium isotopes. The lattice parameter change and its thermal recovery in plutonium and uranium mixed dioxide (MOX) were studied. The lattice parameter for samples of MOX powders and pellets that had been left in the air for up to 32 years was measured. The lattice parameter increased and was saturated at about 0.29%. The change in lattice parameter was formulated as a function of self-radiation dose. Three stages in the thermal recovery of the damage were observed in temperature ranges of below 673K, 673-1073K and above 1073K. The activation energies in each recovery stage were estimated to be 0.12 eV, 0.73 eV and 1.2 eV, respectively, and the corresponding mechanism for each stage was considered to be the recovery of the anion Frenkel defect, the cation Frenkel defect and a defect connected with helium, respectively.
-decay of plutonium isotopes. The lattice parameter change and its thermal recovery in plutonium and uranium mixed dioxide (MOX) were studied. The lattice parameter for samples of MOX powders and pellets that had been left in the air for up to 32 years was measured. The lattice parameter increased and was saturated at about 0.29%. The change in lattice parameter was formulated as a function of self-radiation dose. Three stages in the thermal recovery of the damage were observed in temperature ranges of below 673K, 673-1073K and above 1073K. The activation energies in each recovery stage were estimated to be 0.12 eV, 0.73 eV and 1.2 eV, respectively, and the corresponding mechanism for each stage was considered to be the recovery of the anion Frenkel defect, the cation Frenkel defect and a defect connected with helium, respectively.
Kato, Masato; Morimoto, Kyoichi; Tamura, Tetsuya*; Sunaoshi, Takeo*; Konashi, Kenji*; Aono, Shigenori; Kashimura, Motoaki
Journal of Nuclear Materials, 389(3), p.416 - 419, 2009/06
Times Cited Count:12 Percentile:60.72(Materials Science, Multidisciplinary)Plutonium and uranium mixed oxide (MOX) has been developed to use as a core fuel of the fast reactor. The oxygen to metal ratio (O/M) of the MOX fuel is an important parameter to control the FCCI. The oxygen potential and the oxygen diffusion coefficient of the MOX are essential data to understand the oxygen behaviour in MOX. The oxygen potentials of the MOX were measured with accuracy as a function of O/M and temperatures in the previous work. In this work the oxygen chemical diffusion coefficient in (Pu U
U )O
)O and (Pu
and (Pu U
U )O
)O were investigated using thermo gravimetric technique. The kinetics of the reduction processes of (Pu
were investigated using thermo gravimetric technique. The kinetics of the reduction processes of (Pu U
U )O
)O and (Pu
and (Pu U
U )O
)O were measured by TG-DTA method. The oxygen chemical diffusion coefficients have been estimated from the reduction curves. It was concluded that the oxygen chemical diffusion coefficient in (Pu
were measured by TG-DTA method. The oxygen chemical diffusion coefficients have been estimated from the reduction curves. It was concluded that the oxygen chemical diffusion coefficient in (Pu U
U )O
)O is a smaller than that of (Pu
is a smaller than that of (Pu U
U )O
)O .
.
 system
systemNakamichi, Shinya; Kato, Masato; Sunaoshi, Takeo*; Uchida, Teppei; Morimoto, Kyoichi; Kashimura, Motoaki; Kihara, Yoshiyuki
Journal of Nuclear Materials, 389(1), p.191 - 196, 2009/05
Times Cited Count:2 Percentile:17.04(Materials Science, Multidisciplinary)Japan Atomic Energy Agency researchers have developed mixed oxide (MOX) fuels containing minor actinides (MA). These fuels were irradiated for ten minutes in the FBR Joyo in some short-term irradiation tests. The Si-condensed phases were observed at the center of the pellets in the post irradiation examination. Si impurities came to be mixed into the raw materials in the ball milling process, because Si rubber was used as the lining of the milling pot. Content of Si in the pellets was within the specification of the fuel. It is important to investigate the Si state in MOX at high temperatures like the reactor operating temperature of the fuel to evaluate irradiation behavior. In the present work, MOX specimens with mixed SiO impurity were prepared. The ratio of MOX to SiO
impurity were prepared. The ratio of MOX to SiO was controlled at a mol fraction of 3 to 1. The specimens were first heated at 1973K in atmospheres of three different oxygen partial pressures to adjust the O/M ratio. Then these specimens were sealed in a tungsten capsule, and heated at 2273K or 2673K. Compounds consisting of Pu and Si were observed at grain boundaries of the MOX matrix in specimens after heat treatment. These compounds were not observed in grain interior and MOX matrix was not affected significantly by Si impurity. These compounds tended to form in specimens with low O/M ratio and in specimens heated at higher temperatures.
was controlled at a mol fraction of 3 to 1. The specimens were first heated at 1973K in atmospheres of three different oxygen partial pressures to adjust the O/M ratio. Then these specimens were sealed in a tungsten capsule, and heated at 2273K or 2673K. Compounds consisting of Pu and Si were observed at grain boundaries of the MOX matrix in specimens after heat treatment. These compounds were not observed in grain interior and MOX matrix was not affected significantly by Si impurity. These compounds tended to form in specimens with low O/M ratio and in specimens heated at higher temperatures.
Takeuchi, Kentaro; Kato, Masato; Sunaoshi, Takeo*; Aono, Shigenori; Kashimura, Motoaki
Journal of Nuclear Materials, 385(1), p.103 - 107, 2009/03
Times Cited Count:4 Percentile:29.45(Materials Science, Multidisciplinary)The effective thermal conductivities were evaluated by measuring radial temperature distribution in MH-MOX powder. The effective thermal conductivities were very small and they changed significantly with O/M, bulk density and atmosphere gas. The results in this work were analyzed by the model of Hamilton and Crosser and the new model for the effective thermal conductivity for MH-MOX powder was derived as a function of powder properties, thermal conductivity of atmosphere gas and temperature.
Kato, Masato; Morimoto, Kyoichi; Nakamichi, Shinya; Sugata, Hiromasa*; Konashi, Kenji*; Kashimura, Motoaki; Abe, Tomoyuki
Nihon Genshiryoku Gakkai Wabun Rombunshi, 7(4), p.420 - 428, 2008/12
The melting temperatures of MOX for fast reactor fuel were investigated as functions of Pu content, Am content and oxygen-to-metal (O/M) ratio using thermal arrest technique. Rhenium inner was used for the measurement to prevent the reaction between the sample and capsule materials. The solidus temperatures decreased with increasing Pu and Am content and increased with decreasing O/M ratio. It is considered that the maximum temperature in U-Pu-O system varies in hypostoichiometric composition region. The melting temperatures were evaluated by ideal solid solution model in UO -PuO
-PuO -AmO
-AmO -PuO
-PuO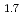 system, and the model was derived for calculating solidus and liquidus temperature. The derived model reproduced the experimental data with
system, and the model was derived for calculating solidus and liquidus temperature. The derived model reproduced the experimental data with  25 K.
25 K.
Kato, Masato; Morimoto, Kyoichi; Sugata, Hiromasa*; Konashi, Kenji*; Kashimura, Motoaki; Abe, Tomoyuki
Journal of Alloys and Compounds, 452(1), p.48 - 53, 2008/03
Times Cited Count:32 Percentile:78.13(Chemistry, Physical)Plutonium and uranium mixed oxide has been developed as a fuel of a fast reactor. The maximum temperature of the fuel pellet is limited within a design criterion to prevent fuel melting. So, the melting points of the mixed oxide have been investigated since the development of fast reactor started. However the measured data are limited. In this work, the melting points of (U1-yPuy)O (y: 0, 0.12, 0.2, 0.3, 0.4) were measured by the thermal arrest method. The evaluated melting point of this study underestimates in case of MOX with high Pu contents of 30% and 40%. The solidus of UO
(y: 0, 0.12, 0.2, 0.3, 0.4) were measured by the thermal arrest method. The evaluated melting point of this study underestimates in case of MOX with high Pu contents of 30% and 40%. The solidus of UO , (Pu
, (Pu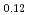 U
U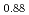 )O
)O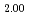 and (Pu
and (Pu U
U )O
)O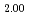 were determined to be 3128K, 3077K and 3052K, respectively. The solidus temperature of hypostoichiometric MOX slightly increased with decreasing O/M.
were determined to be 3128K, 3077K and 3052K, respectively. The solidus temperature of hypostoichiometric MOX slightly increased with decreasing O/M.
 solid solutions
solid solutionsMorimoto, Kyoichi; Kato, Masato; Ogasawara, Masahiro*; Kashimura, Motoaki; Abe, Tomoyuki
Journal of Alloys and Compounds, 452(1), p.54 - 60, 2008/03
Times Cited Count:32 Percentile:78.13(Chemistry, Physical)Plutonium and uranium mixed oxide (MOX) fuel with high Pu content have been developed as a fuel of fast reactor (FR). As the storage time of Pu raw material between reprocessing and fabrication increases, americium content of the fabricated MOX fuel increases up to a few percent. In this work, the thermal conductivity of MOX fuel containing Am was investigated as a part of clarifying the effect of Am content on thermal physical properties. The pellets of (Am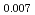 Pu
Pu U)O
U)O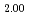 , (Am
, (Am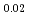 Pu
Pu U)O
U)O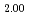 and (Am
and (Am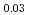 Pu
Pu U)O
U)O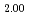 were prepared. The oxygen to metal ratio (O/M ratio) of sintered pellet was adjusted to 2.00. The thermal diffusivity measurement was carried out in the range of temperature from 900 K to 1700 K by the laser flash method, and thermal conductivity of these pellets was evaluated. The heat capacity for evaluating thermal conductivity was derived from heat capacity of UO
were prepared. The oxygen to metal ratio (O/M ratio) of sintered pellet was adjusted to 2.00. The thermal diffusivity measurement was carried out in the range of temperature from 900 K to 1700 K by the laser flash method, and thermal conductivity of these pellets was evaluated. The heat capacity for evaluating thermal conductivity was derived from heat capacity of UO , PuO
, PuO and AmO
and AmO by using the Kopp-Neumann rule.
by using the Kopp-Neumann rule.
 oxide
oxideMorimoto, Kyoichi; Kato, Masato; Ogasawara, Masahiro*; Kashimura, Motoaki
Journal of Nuclear Materials, 374(3), p.378 - 385, 2008/03
Times Cited Count:37 Percentile:89.13(Materials Science, Multidisciplinary)The thermal conductivities of (U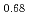 Pu
Pu Am
Am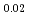 )O
)O solid solutions (x = 0.0 - 0.1) were studied at temperatures from 900 to 1773 K. Thermal conductivities were obtained from the thermal diffusivity measured by laser flash method. The thermal conductivities obtained experimentally up to about 1400K could be expressed by a classical phonon transport model,
solid solutions (x = 0.0 - 0.1) were studied at temperatures from 900 to 1773 K. Thermal conductivities were obtained from the thermal diffusivity measured by laser flash method. The thermal conductivities obtained experimentally up to about 1400K could be expressed by a classical phonon transport model,  = (A+BT)
= (A+BT) , A(x) = 2.89
, A(x) = 2.89 x + 2.24
x + 2.24 10
10 (m K/W) and B(x) = (- 6.70
(m K/W) and B(x) = (- 6.70 x + 2.48)
x + 2.48)  10
10 (m/W). The experimental values of A showed a good agreement with theoretical predictions. The experimental values of B could be fairly expressed by the theoretical prediction in the region x
(m/W). The experimental values of A showed a good agreement with theoretical predictions. The experimental values of B could be fairly expressed by the theoretical prediction in the region x  0.04, but not deviated from the ones in the region x
0.04, but not deviated from the ones in the region x  0.04. Although this reason could not be understood well, it is most likely that the uncertainty in the measurement of melting temperature cause this difference.
0.04. Although this reason could not be understood well, it is most likely that the uncertainty in the measurement of melting temperature cause this difference.
 -PuO
-PuO system
systemKato, Masato; Morimoto, Kyoichi; Sugata, Hiromasa*; Konashi, Kenji*; Kashimura, Motoaki; Abe, Tomoyuki
Journal of Nuclear Materials, 373(1-3), p.237 - 245, 2008/02
Times Cited Count:64 Percentile:96.04(Materials Science, Multidisciplinary)The melting of plutonium and uranium mixed oxide (MOX) containing Pu of more than 30% was investigated using a tungsten capsule and a rhenium inner capsule. In the conventional measurement of MOX in the tungsten capsule, a liquid phase of tungsten and plutonium oxide appeared in the MOX during melting. This liquid phase was found to have an effect on the measurement of melting point. Therefore the rhenium inner capsule was used to avoid the effect. The solidus and liquidus temperatures in the UO -PuO
-PuO system were decided from the MOX data measured using the rhenium capsule, and the effect of the Am content on the solidus temperature was evaluated. The variation of the solidus and liquidus temperatures in the UO
system were decided from the MOX data measured using the rhenium capsule, and the effect of the Am content on the solidus temperature was evaluated. The variation of the solidus and liquidus temperatures in the UO -PuO
-PuO -AmO
-AmO ternary system was represented to an accuracy of
ternary system was represented to an accuracy of  =
= 9K and
9K and  =
= 16K, respectively, by the ideal solution model.
16K, respectively, by the ideal solution model.
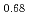 Pu
Pu Am
Am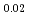 )O
)O in high temperature region
in high temperature regionKomeno, Akira; Morimoto, Kyoichi; Kato, Masato; Kashimura, Motoaki; Ogasawara, Masahiro*; Sunaoshi, Takeo*
Transactions of the American Nuclear Society, 97(1), p.616 - 617, 2007/11
no abstracts in English

Morimoto, Kyoichi; Kato, Masato; Komeno, Akira; Kashimura, Motoaki
Transactions of the American Nuclear Society, 97(1), p.618 - 619, 2007/11
Plutonium and uranium mixed oxide (MOX) fuel with high Pu-content has been developed as a fuel for fast reactors (FRs). Thermal conductivity of the oxide fuel is among the most important properties for design and performance analyses of fuel rods. Among recent reports, there have been none examining of thermal conductivity of MOX fuel containing Am except our studies. In this study, the thermal conductivities of MOX fuel with 30% Pu-content, as obtained by our group, were evaluated as functions of temperature, oxygen-to-metal (O/M) ratio and Am-content.
 Pu
Pu )O
)O with two fcc phases
with two fcc phasesSuzuki, Kiichi; Kato, Masato; Tamura, Tetsuya*; Aono, Shigenori; Kashimura, Motoaki
Journal of Alloys and Compounds, 444-445, p.590 - 593, 2007/10
Times Cited Count:5 Percentile:37.12(Chemistry, Physical)It was reported that sintered MOX pellet of hypostoichiometric composition was oxidized at room temperature in an atmosphere of inert gas and air. The region of two fcc phases exist at room temperature in the (U,Pu)O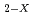 with Pu content of greater than 20%. In this study, the oxidation rate of (U
with Pu content of greater than 20%. In this study, the oxidation rate of (U Pu
Pu )O
)O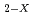 with two fcc phases was investigated to contribute to understanding of the oxidation behavior using thermogravimetric technique. The sintered pellets of (U
with two fcc phases was investigated to contribute to understanding of the oxidation behavior using thermogravimetric technique. The sintered pellets of (U Pu
Pu )O
)O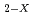 were prepared by mechanical blending method and were sliced into disc-like sample with about 1 mm thick and 85-93% theoretical density. The oxidation rate of the samples were measured at 60, 125 and 150
were prepared by mechanical blending method and were sliced into disc-like sample with about 1 mm thick and 85-93% theoretical density. The oxidation rate of the samples were measured at 60, 125 and 150 C in an atmosphere of Air, N
C in an atmosphere of Air, N and Air/N
and Air/N gas mixture containing moisture of 1 - 700ppm using thermal gravity and differential thermal analysis. The curve of the isothermal oxidation was analyzed by the model of diffusion in a system consisting of two phases. The diffusion model can represent the oxidation curve as a function of time and temperature. In the results of X-ray diffraction measurement, fcc phase with O/M
gas mixture containing moisture of 1 - 700ppm using thermal gravity and differential thermal analysis. The curve of the isothermal oxidation was analyzed by the model of diffusion in a system consisting of two phases. The diffusion model can represent the oxidation curve as a function of time and temperature. In the results of X-ray diffraction measurement, fcc phase with O/M  2.00 was observed to increase by oxidation of sample. These results indicate that the oxidation of the (U
2.00 was observed to increase by oxidation of sample. These results indicate that the oxidation of the (U Pu
Pu )O
)O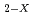 with two fcc phases proceeds by diffusion of the phase with O/M
with two fcc phases proceeds by diffusion of the phase with O/M  2.00 which is formed on the sample surface.
2.00 which is formed on the sample surface.
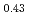 Am
Am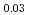 U
U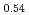 )O
)O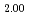
Nakamichi, Shinya; Kato, Masato; Morimoto, Kyoichi; Sugata, Hiromasa*; Kashimura, Motoaki; Abe, Tomoyuki
Transactions of the American Nuclear Society, 96(1), p.191 - 192, 2007/06
JAEA has developed plutonium and uranium mixed oxide (MOX) containing 20-32%Pu content as a fuel of the fast breeder reactor. During irradiation, large temperature gradient in radial direction of a fuel pellet causes redistribution of Pu and U, and the Pu content increases to about 43% at the pellet center. The maximum temperature of the fuel pellet during irradiation is limited within the design criterion to prevent fuel melting. So, it is important to evaluate melting points of MOX containing 43%Pu. In this work, it is confirmed that the MOX with 43%Pu content is not melted by heat treatment just below the melting point which was determined by thermal arrest technique using Re inner capsule. The MOX specimen with 43%Pu content was heated at 2978K for 40s using Re inner capsule. Optical micrograph and XRD results show the specimen was heated at the temperature less than solidus temperature. So it was confirmed that (Pu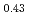 Am
Am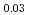 U
U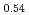 )O
)O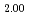 was solid phase at 2978K
was solid phase at 2978K 20K.
20K.
Kato, Masato; Morimoto, Kyoichi; Sugata, Hiromasa*; Konashi, Kenji*; Kashimura, Motoaki; Abe, Tomoyuki
Transactions of the American Nuclear Society, 96(1), p.193 - 194, 2007/06
Melting point of a nuclear fuel is one of the important physical properties for its development, because it limits maximum temperature of the fuel during operation. A rhenium inner capsule was used to prevent the reaction with capsule for measuring melting points of MOX. In this work melting points of MOX with 40% and 46%Pu were investigated as a function of an O/M ratio using Re inner, and the effect of the O/M ratio on the melting points was evaluated. The solidus and liquidus temperatures in (Pu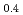 U
U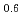 )O
)O and (Pu
and (Pu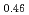 U
U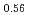 )O
)O were measured by thermal arrest method. It was observed that the melting points in the both samples increased with a decrease of the O/M from 2.00, and their data were 50-100K higher than existing data measured in previous works which were measured with W capsule.
were measured by thermal arrest method. It was observed that the melting points in the both samples increased with a decrease of the O/M from 2.00, and their data were 50-100K higher than existing data measured in previous works which were measured with W capsule.
Kato, Masato; Morimoto, Kyoichi; Komeno, Akira; Nakamichi, Shinya; Kashimura, Motoaki; Abe, Tomoyuki; Uno, Hiroki*; Ogasawara, Masahiro*; Tamura, Tetsuya*; Sugata, Hiromasa*; et al.
JAEA-Technology 2006-049, 32 Pages, 2006/10
Japan Atomic Energy Agency has developed a fast breeder reactor(FBR), and plutonium and uranium mixed oxide (MOX) having low density and 20-30%Pu content has used as a fuel of the FBR, Monju. In plutonium, Americium has been accumulated during long-term storage, and Am content will be increasing up to 2-3% in the MOX. It is essential to evaluate the influence of Am content on physical properties of MOX on the development of FBR in the future. In this study melting points and thermal conductivities which are important data on the fuel design were measured systematically in wide range of composition, and the effects of Am accumulated were evaluated. The solidus temperatures of MOX were measured as a function of Pu content, oxygen to metal ratio(O/M) and Am content using thermal arrest technique. The sample was sealed in a tungsten capsule in vacuum for measuring solidus temperature. In the measurements of MOX with Pu content of more than 30%, a rhenium inner capsule was used to prevent the reaction between MOX and tungsten. In the results, it was confirmed that the melting points of MOX decrease with as an increase of Pu content and increase slightly with a decrease of O/M ratio. The effect of Am content on the fuel design was negligible small in the range of Am content up to 3%. Thermal conductivities of MOX were evaluated from thermal diffusivity measured by laser flash method and heat capacity calculated by Nuemann- Kopp's law. The thermal conductivity of MOX decreased slightly in the temperature of less than 1173K with increasing Am content. The effect of Am accumulated in long-term storage fuel was evaluated from melting points and thermal conductivities measured in this study. It is concluded that the increase of Am in the fuel barely affect the fuel design in the range of less than 3%Am content.
Komeno, Akira; Kato, Masato; Morimoto, Kyoichi; Kashimura, Motoaki; Abe, Tomoyuki; Sugata, Hiromasa*; Shibata, Katsuya*; Uno, Hiroki*; Tamura, Tetsuya*
no journal, ,
no abstracts in English