Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
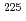 Ac metallofullerene; Toward
Ac metallofullerene; Toward 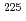 Ac nanogenerator in fullerene
Ac nanogenerator in fullereneAkiyama, Kazuhiko*; Haba, Hiromitsu*; Sueki, Keisuke*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Nagame, Yuichiro; Katada, Motomi*
Chemistry Letters, 38(10), p.978 - 979, 2009/09
Times Cited Count:9 Percentile:36.86(Chemistry, Multidisciplinary)We report on the successful production of a metallofullerene encapsulating the radioactive tracer 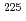 Ac and on its electronic properties studied by radiochromatography. Considering the number of
Ac and on its electronic properties studied by radiochromatography. Considering the number of  -electrons on the fullerene cage estimated from an HPLC retention time on the 5PBB column and the general oxidation state of Ac (+3), a chemical species of the dominant chromatographic peak is suggested to be Ac@C
-electrons on the fullerene cage estimated from an HPLC retention time on the 5PBB column and the general oxidation state of Ac (+3), a chemical species of the dominant chromatographic peak is suggested to be Ac@C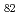 .
.
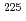 Ac
AcAkiyama, Kazuhiko*; Haba, Hiromitsu*; Tsukada, Kazuaki; Asai, Masato; Toyoshima, Atsushi; Sueki, Keisuke*; Nagame, Yuichiro; Katada, Motomi*
Journal of Radioanalytical and Nuclear Chemistry, 280(2), p.329 - 331, 2009/05
Times Cited Count:7 Percentile:43.76(Chemistry, Analytical)The properties of a metallofullerene that encapsulates 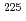 Ac were studied by radio-chromatographic methods. The results of chromatography on a Buckyclutcher column suggest that there are some metallofullerene isomers of AcC
Ac were studied by radio-chromatographic methods. The results of chromatography on a Buckyclutcher column suggest that there are some metallofullerene isomers of AcC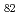 . The identical HPLC retention time of the major AcC
. The identical HPLC retention time of the major AcC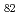 peak with that of La@C
peak with that of La@C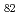 indicates that the structure of one of the AcC
indicates that the structure of one of the AcC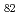 isomers is the same as La@C
isomers is the same as La@C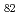 .
.
Akiyama, Kazuhiko; Sueki, Keisuke*; Tsukada, Kazuaki; Yaita, Tsuyoshi; Miyake, Yoko*; Haba, Hiromitsu*; Asai, Masato; Kodama, Takeshi*; Kikuchi, Koichi*; Otsuki, Tsutomu*; et al.
Journal of Nuclear and Radiochemical Sciences, 3(1), p.151 - 154, 2002/06
The oxidation state of actinide elements encapsulated in fullerenes is studied. HPLC elution behavior of actinide-fullerenes is classified into two groups; the elution behavior of the first group, encapsulating U, Np, and Am, is similar to that of the light lanthanide-fullerenes, such as La, Ce, Pr, and Nd, while the behavior of the second group, encapsulating Th and Pa, is quite different from that of any lanthanide-fullerenes. The chemical species in the main HPLC elution peak of each group were identified as M@C82 and M@C84 (M = metal atom) from the mass of the U and Th fullerenes, respectively. The oxidation states of the U and Th atoms in the fullerenes were deduced to be 3+ and 4+, respectively, from the UV/vis/NIR absorption and XANES spectroscopy.
Akiyama, Kazuhiko; Zhao, Y.*; Sueki, Keisuke*; Tsukada, Kazuaki; Haba, Hiromitsu; Nagame, Yuichiro; Kodama, Takeshi*; Suzuki, Shinzo*; Otsuki, Tsutomu*; Sakaguchi, Masahiko*; et al.
Journal of the American Chemical Society, 123(1), p.181 - 182, 2001/01
Times Cited Count:67 Percentile:84.87(Chemistry, Multidisciplinary)no abstracts in English