Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Uosaki, Kohei*; Morita, Jun*; Katsuzaki, Tomoko*; Takakusagi, Satoru*; Tamura, Kazuhisa; Takahashi, Masamitsu; Mizuki, Junichiro; Kondo, Toshihiro*
Journal of Physical Chemistry C, 115(25), p.12471 - 12482, 2011/06
Times Cited Count:13 Percentile:40.03(Chemistry, Physical)Ag/AgCl reaction at the Ag bilayer, which was underpotentially prepared on a Au(111) surface, was investigated using electrochemical quartz crystal microbalance (EQCM), scanning tunneling microscopy (STM), surface X-ray scattering (SXS), and electrochemical techniques. When the potential was scanned positively from -200 mV, the Cl ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl
ion was adsorbed on the Au(111) electrode surface around 0 mV, and then the phase transition of the adsorbed Cl ion layer from random orientation to (
ion layer from random orientation to (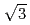

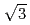 ) structure took place at around +130 mV. The Ag bilayer and Cl
) structure took place at around +130 mV. The Ag bilayer and Cl ions were oxidatively reacted to form the AgCl monolayer with (
ions were oxidatively reacted to form the AgCl monolayer with (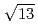

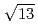 )
) 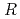 13.9
13.9 structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (
structure around +200 mV, accompanied with the formation of AgCl monocrystalline clusters on the AgCl monolayer surface. The structure of the AgCl monolayer on the Au(111) surface was changed from (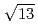

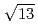 )R13.9
)R13.9 structure to (4
structure to (4 4) structure around +500 mV.
4) structure around +500 mV.