Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Yotsuji, Kenji*; Tachi, Yukio; Sakuma, Hiroshi*; Kawamura, Katsuyuki*
Genshiryoku Bakkuendo Kenkyu (CD-ROM), 29(2), p.63 - 81, 2022/12
The understanding of the swelling phenomenon of montmorillonite is essential to predict the physical and chemical behavior of clay-based barriers in radioactive waste disposal systems. This study investigated the key factors controlling crystalline swelling behavior of montmorillonite with different interlayer counter-ions by molecular dynamics (MD) simulations. On the basis of the comparisons between MD simulated and experimental results, the water content in the interlayer in five homoionic (Na , K
, K , Cs
, Cs , Ca
, Ca and Sr
and Sr ) montmorillonite was strongly correlated to the hydration number and the preference of an outer- or inner-sphere complex of each counter-ion. The detailed analysis for these results offer insights that the hydration number is controlled by the hydration free energy, the volume and the distribution of each interlayer counter-ion. The systematic MD simulations with virtually variable parameters clarified that the hydration free energy and the charge of interlayer counter- ions compete as influencing factors, and the control the formation rate of an outer-sphere complex of each counter-ion. The empirical relationships between these key factors will allow essential insights into predicting the swelling behavior of montmorillonite with different interlayer counter-ions.
) montmorillonite was strongly correlated to the hydration number and the preference of an outer- or inner-sphere complex of each counter-ion. The detailed analysis for these results offer insights that the hydration number is controlled by the hydration free energy, the volume and the distribution of each interlayer counter-ion. The systematic MD simulations with virtually variable parameters clarified that the hydration free energy and the charge of interlayer counter- ions compete as influencing factors, and the control the formation rate of an outer-sphere complex of each counter-ion. The empirical relationships between these key factors will allow essential insights into predicting the swelling behavior of montmorillonite with different interlayer counter-ions.
Kawamura, Takaaki*; Fukaya, Yuki; Fukutani, Katsuyuki
Surface Science, 722, p.122098_1 - 122098_8, 2022/08
Times Cited Count:1 Percentile:6.44(Chemistry, Physical)no abstracts in English
Yotsuji, Kenji*; Tachi, Yukio; Sakuma, Hiroshi*; Kawamura, Katsuyuki*
Applied Clay Science, 204, p.106034_1 - 106034_13, 2021/04
Times Cited Count:102 Percentile:99.77(Chemistry, Physical)Kimoto, Kazushi*; Kawamura, Katsuyuki*; Makino, Hitoshi
Journal of Computer Chemistry, Japan, 19(2), p.46 - 49, 2020/00
This study proposes a 2D coarse-grained molecular dynamics (CGMD) method for the compaction simulation of montmorillonite clay. In the CGMD method, a unit structure of a water-hydrated clay molecule is coarse-grained into a particle. Thus, the deformable molecules are modeled as a set of linearly connected coarse-grained particles. As the inter-particle forces, the intra-molecular bonding and inter-molecular van der Waals forces are considered. For simplicity, the intra-molecular bonding is modeled as a linear harmonic oscillator, while the Lenard-Jones potential is used to define the van der Waals force field. With this model, the mechanical compaction of moistured montmorillonite is numerically simulated to find that 4-6 considerably deformed molecules are layered as a result of the compaction. It is alsofound that the simulated XRD pattern agrees to the experiment in terms of the peak angle.
Kawamura, Takaaki*; Fukutani, Katsuyuki
Surface Science, 688, p.7 - 13, 2019/10
Times Cited Count:5 Percentile:22.39(Chemistry, Physical)Yotsuji, Kenji*; Tachi, Yukio; Kawamura, Katsuyuki*; Arima, Tatsumi*; Sakuma, Hiroshi*
Nendo Kagaku, 58(1), p.8 - 25, 2019/00
Molecular dynamics (MD) simulations were conducted to investigate physical properties of water and cations in montmorillonite interlayer nanopores. The swelling behaviors and hydration states were firstly evaluated as functions of interlayer cations and layer charge. The diffusion coefficients of water and cations in interlayer nanopores were decreased in comparison with those in bulk water and came closer to those in bulk water when basal spacing increased. The viscosity coefficients of interlayer water estimated indicated a significant effect of viscoelectricity at 1- and 2-layer hydration states and higher layer charge of montmorillonite. These trends from MD calculations were confirmed to be consistent with existing measured data and previous MD simulation. In addition, model and parameter related to viscoelectric effect used in the diffusion model was refined based on comparative discussion between MD simulations and measurements. The series of MD calculations could provide atomic level understanding for the developments and improvements of the diffusion model for compacted montmorillonite.
 Cs solid-state NMR and density functional theory calculations
Cs solid-state NMR and density functional theory calculationsOkubo, Takahiro*; Okamoto, Takuya*; Kawamura, Katsuyuki*; Gu gan, R.*; Deguchi, Kenzo*; Oki, Shinobu*; Shimizu, Tadashi*; Tachi, Yukio; Iwadate, Yasuhiko*
gan, R.*; Deguchi, Kenzo*; Oki, Shinobu*; Shimizu, Tadashi*; Tachi, Yukio; Iwadate, Yasuhiko*
Journal of Physical Chemistry A, 122(48), p.9326 - 9337, 2018/12
Times Cited Count:15 Percentile:56.66(Chemistry, Physical)The structures of Cs adsorption on montmorillonite were investigated by the nuclear magnetic resonance (NMR) spectroscopy. The NMR spectra of Cs adsorbed on montmorillonite samples were measured under different Cs contents and relative humidity levels. NMR parameters were evaluated by the first principle calculations in order to identify the relationship between adsorbed Cs structures and NMR parameters. The comparisons between experimental and theoretical NMR spectra revealed that Cs is preferentially adsorbed at sites near Al for low Cs substituted montmorillonites, and that non-hydrated Cs present in partially Cs substituted samples, even after being hydrated under high relative humidity.
Sakuma, Hiroshi*; Tachi, Yukio; Yotsuji, Kenji; Suehara, Shigeru*; Arima, Tatsumi*; Fujii, Naoki*; Kawamura, Katsuyuki*; Honda, Akira
Clays and Clay Minerals, 65(4), p.252 - 272, 2017/08
Times Cited Count:2 Percentile:15.19(Chemistry, Physical)Structure and stability of montmorillonite edge faces (110), (010), (100), and (130) of the layer charges y = 0.5 and 0.33 are investigated by the first-principles electronic calculations based on the density functional theory. Stacking and single layer models are tested for understanding the effect of stacking on the stability of montmorillonite edge faces. Most stacking layers stabilize the edge faces by making hydrogen bonds between the layers; therefore, the surface energy of stacking layers is reduced rather than the single layer model. This indicates that the surface energy of edge faces should be estimated depending on the swelling conditions. Lowest surface energies of (010) and (130) edge faces were realized by the presence of Mg ions on the edge faces. These edge faces have a strong adsorption site for cations due to local negative charge of the edges.
Arima, Tatsumi*; Idemitsu, Kazuya*; Inagaki, Yaohiro*; Kawamura, Katsuyuki*; Tachi, Yukio; Yotsuji, Kenji
Progress in Nuclear Energy, 92, p.286 - 297, 2016/09
Times Cited Count:15 Percentile:77.12(Nuclear Science & Technology)Diffusion and adsorption behavior of uranyl (UO ) species is important for the performance assessment of radioactive waste disposal. The diffusion behaviors of UO
) species is important for the performance assessment of radioactive waste disposal. The diffusion behaviors of UO , K
, K , CO
, CO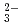 and Cl
and Cl and H
and H O in the aqueous solutions were evaluated by molecular dynamics (MD) calculations. The diffusion coefficient (De) of UO
O in the aqueous solutions were evaluated by molecular dynamics (MD) calculations. The diffusion coefficient (De) of UO is the smallest and is 26% less than the self-diffusion coefficient of H
is the smallest and is 26% less than the self-diffusion coefficient of H O. For the aqueous solution with high concentration of carbonate ions, uranyl carbonate complexes: UO
O. For the aqueous solution with high concentration of carbonate ions, uranyl carbonate complexes: UO CO
CO and UO
and UO (CO
(CO )
) can be observed. For the clay (montmorillonite or illite)-aqueous solution systems, the adsorption and diffusion behaviors of UO
can be observed. For the clay (montmorillonite or illite)-aqueous solution systems, the adsorption and diffusion behaviors of UO and K
and K were evaluated by MD calculations. The distribution coefficients (Kd) increase with the layer charge of clay, and Kd of UO
were evaluated by MD calculations. The distribution coefficients (Kd) increase with the layer charge of clay, and Kd of UO might be smaller than that of K
might be smaller than that of K . Further, their two-dimensional diffusion coefficients were relatively small in the adsorption layer and were extremely small for illite with higher layer charge.
. Further, their two-dimensional diffusion coefficients were relatively small in the adsorption layer and were extremely small for illite with higher layer charge.
Kawamura, Katsuyuki*
JNC TJ8400 2004-028, 69 Pages, 2005/02
Solute and radionuclide transport analysis in bu.er material made of bentonite clay is essential in safety assessment of a geological disposal facility for high-level radioactive waste (HLW). It is keenly required to understand the true physical and chemical process of the transport phenomena and to improve reliability of the safety assessment, since any conventional methods based on experimental models involve di.culty to estimate the robustness for a very long-term behavior. In order to solve this di.culty we start with the molecular dynamics (MD) simulation method for undrstanding the molecular-based fundamental properties such as an ionic state and diffusion characteristics of hydrated smectite clay minerals, and we extend the microscale properties to the macroscale behaviors by applying the multiscale homogenization method. In the study of this year we improved the MD atomic model for the hydrated clay minerals, and a new adsorption-di.usion analysis scheme by the homogenization analysis (HA). In the MD simulation first we improved the interatomic potential model for the smectitic clays. Then the behaviors of hydrated Na-beidellite and its substitution products by Cs and Ca were calculated. Not only the swelling behaviors of the beidellite minerals but also the di.usion characteristics of cations in the interlayer space are calculated. A microscopic image is important to specify micro/macro behavior of bentonite. Last year we observed microstructures of bentonite by using a confocal laser scanning microscope (LSM). Based on the knowlwdge of the local material properties obtained by MD and the microscopic observation we simulated the micro-/macro-behavior of diffusion experiments of the bentonite which included the microscaleadsorption characteristics at the edges of clay minerals.
Kawamura, Katsuyuki*
JNC TJ8400 2004-001, 39 Pages, 2004/03
It is keenly required to understand the true physical and chemical process of the transport phenomena and to improve reliability of the safety assessment. For this perpose, the molecular dynamic (MD) simulation method and the homogenization analysis (HA) were applyed. In this study, MD atomic model sor the hydrated clay minerals were improved, and HA simulation for diffusion experiments of the bentonite including microscale adsorption at the edges of clay mineral were conducted.
Suzuki, Satoru; Kawamura, Katsuyuki*
Journal of Physical Chemistry B, 108, p.13468 - 13474, 2004/00
Times Cited Count:28 Percentile:57.72(Chemistry, Physical)None
Kawamura, Katsuyuki*
JNC TJ8400 2003-011, 55 Pages, 2003/03
Solute and radionuclide transport analysis in buffer material of bentonite day is essential in safety assessment of a geological disposal facility for high-level radioactive waste (HLW). It is keenly required to understand the true physical and chemical process of the transport phenomena and to improve reliability of the safety assessment, since any conventional methods based on experimental models involve difficulty to estimate the level of robustness. For this purpose we start with the molecular dynamics (MD) simulation method to know molecular-based fundamental properties such as an ionic state and diffusion characteristics of hydrated smectite clay minerals, and we intend to extend the microscale properties to the macroscale behaviors by applying the multiscale homogenization method. In the current year study we introduced a detailed study of MD simulation of hydrated clay minerals, and observed microstructures of bentonite by using a confocal laser scanning microscope (LSM). In the MD simulation first a new interatomic potential model was introduced for water molecules. Then the behaviors of hydrated Na-beidellite and its substitution products by Cs and Ca were calculated. Hydrated muscovite and pyrophyllite were also treated. A three-phase system of clay minerals with edges, water and gas is finally was considered to check the behaviors of cations and water molecules near the edges. A microscopic image is important to specify micro/macro behavior of bentonite. We used the LSM (Ar-laser, wave length 488nm) to characterize the microscopic geometry of bentonite. We could observe a 3-D configuration under atmospheric condition to get images of Kunigel V1 (a well-known bentonite in Japan) and Kunipia F (a refined bentonite including 99% of montmorillonite). By using the advantage of LSM we observed swelling properties of bentnite particles under several salt concentration conditions.
Suzuki, Satoru; Sato, Haruo; Ishidera, Takamitsu; Fujii, Naoki*; Kawamura, Katsuyuki*
JNC TN8400 2001-031, 44 Pages, 2002/05
In order to quantify effect of temperature on diffusivity of deuterated water (HDO) in compacted sodium-bentonite, through-diffusion experiments were conducted at elevated tempemture from 298 to 333 K. Kunipia F (Na-montmorillonite content  98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m
98 wt. %; Kunimine Industly Co.) was compacted to a dry density of 0.9 and l.35 Mg/m . Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D
. Since smectite flakes were perpendicularly oriented to a direction of compaction, anisotropy of diffusivity was investigated parallel and normal to the preferred orientation of smectite. Effective diffusion coeficient D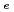 of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D
of HDO was larger for a diffusional direction parallel to the preferred orientation than normal to that for both dry densities. These results well agreed to the previously reported ones for tritiated water. Activation energies of D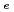 in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
in compacted bentonite increased with increasing dry density in the range of 19 - 25 kJ/mol which was slightly larger than that in bulk water (18 kJ/mol). This relationship can be considered to be due to both the pore structure development and high activation energy of water (18-23 kJ/mol) in the vicinity of smectite surface (within 2 nm) on the basis of molecular dynamics simulations.
Kawamura, Katsuyuki*; Ichikawa, Yasuaki*; Suzuki, Satoru; Shibata, Masahiro; Sato, Haruo; Ueno, Kenichi
JNC TY8400 2002-015, 90 Pages, 2002/03
None
Suzuki, Satoru; Fujishima, Atsushi; Ueno, Kenichi; Ichikawa, Yasuaki*; Kawamura, Katsuyuki*; Fujii, Naoki*; Shibata, Masahiro; Sato, Haruo; Kitayama, Kazumi*
Nendo Kagaku, 41(2), p.43 - 57, 2001/12
None
Suzuki, Satoru; Fujishima, Atsushi; Ueno, Kenichi; Ichikawa, Yasuaki*; Kawamura, Katsuyuki*; Fujii, Naoki*; Shibata, Masahiro; Sato, Haruo
JNC TY8400 2001-003, 66 Pages, 2001/05
no abstracts in English
Suzuki, Satoru; Kawamura, Katsuyuki*
JNC TN8400 2001-005, 41 Pages, 2001/04
A correlation between molecular structure and a vibrational spectrum of interlayer water in Na-smectite was investigated by means of Molecular Dymamics (MDs) simulations. Detailed comparison of simulation results with IR spectroscopic observations for the water-smectite system indicated good agreement. Internal vibrational spectra of water were obtained by the Fourier transformation of velocty auto-correlation function of hydrogen atom. A stretching vibrational spectrum of interlayer water consisted of a broad band with a peak top around 3400cm and a sharp peak around 3650 to 3700cm
and a sharp peak around 3650 to 3700cm . The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O
. The fomer broad band was assigned to O-H vibrations between water molecules as bulk water, while the latter band was attributed to O-H ones oriented to siloxane surface through hydrogen bonding. The hydrogen bond distance, determined as the shortest O-O distance by the radial distribution function (RDF), revealed that hydrogen bond distance between water and siloxane surface (O -O
-O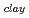
 3.0
3.0  -O
-O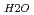 = ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
= ca. 2.8 AA ). These results suggested that interaction between water molecule and siloxane surface weaker than that between water molecules, although they were forced to be oriented.
Yotsuji, Kenji*; Tachi, Yukio; Kawamura, Katsuyuki*
no journal, ,
We used molecular dynamics method to study the diffusivity of water and counterions and the viscosity of interlayer water in montmorillonite clay minerals.
Arima, Tatsumi*; Inagaki, Yaohiro*; Idemitsu, Kazuya*; Kawamura, Katsuyuki*; Yotsuji, Kenji; Tachi, Yukio
no journal, ,
Molecular dynamics simulations of uranyl ion - carbonate ion - clay - water systems were performed to investigate diffusion behavior of uranyl ion in free water and montmorillonite interlayer.